Abstract
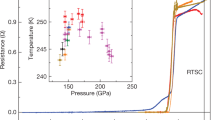
Superconductivity at high temperatures is expected in elements with low atomic numbers, based in part on conventional BCS (Bardeen–Cooper–Schrieffer) theory1. For example, it has been predicted that when hydrogen is compressed to its dense metallic phase (at pressures exceeding 400 GPa), it will become superconducting with a transition temperature above room temperature2. Such pressures are difficult to produce in a laboratory setting, so the predictions are not easily confirmed. Under normal conditions lithium is the lightest metal of all the elements, and may become superconducting at lower pressures3,4; a tentative observation of a superconducting transition in Li has been previously reported5. Here we show that Li becomes superconducting at pressures greater than 30 GPa, with a pressure-dependent transition temperature (Tc) of 20 K at 48 GPa. This is the highest observed Tc of any element; it confirms the expectation that elements with low atomic numbers will have high transition temperatures, and suggests that metallic hydrogen will have a very high Tc. Our results confirm that the earlier tentative claim5 of superconductivity in Li was correct.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bardeen, J., Cooper, L. N. & Schrieffer, J. R. Theory of superconductivity. Phys. Rev. 108, 1175–1204 (1957)
Richardson, C. F. & Ashcroft, N. W. High temperature superconductivity in metallic hydrogen: Electron-electron enhancements. Phys. Rev. Lett. 78, 118–121 (1997)
Allen, P. B. & Cohen, M. L. Pseudopotential calculation of the mass enhancement and superconducting transition temperature of simple metals. Phys. Rev. 187, 525–538 (1969)
Richardson, C. F. & Ashcroft, N. W. Effective electron-electron interactions and the theory of superconductivity. Phys. Rev. B 55, 15130–15145 (1997)
Lin, T. H. & Dunn, K. J. High-pressure and low-temperature study of electrical resistance of lithium. Phys. Rev. B 33, 807–811 (1986)
Neaton, J. B. & Ashcroft, N. W. Pairing in dense lithium. Nature 400, 141–144 (1999)
Hanfland, M., Syassen, K., Christensen, N. E. & Novikov, D. L. New high-pressure phase of lithium. Nature 408, 174–178 (2000)
Christensen, N. E. & Novikov, D. L. Predicted superconductive properties of lithium under pressure. Phys. Rev. Lett. 86, 1861–1864 (2001)
Hanfland, M., Loa, I., Syassen, K., Schwarz, U. & Takemura, K. Equation of state of lithium to 21 GPa. Solid State Commun. 112, 123–127 (1999)
Acknowledgements
We thank H. Goto for technical support in machining the pit-anvils. This work was supported by a Grant-in-Aid for COE (Center of Excellence) Research and Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Shimizu, K., Ishikawa, H., Takao, D. et al. Superconductivity in compressed lithium at 20 K. Nature 419, 597–599 (2002). https://doi.org/10.1038/nature01098
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01098
This article is cited by
-
Ultrasmall single-layered NbSe2 nanotubes flattened within a chemical-driven self-pressurized carbon nanotube
Nature Communications (2024)
-
Spectroscopic evidence for the superconductivity of elemental metal Y under pressure
NPG Asia Materials (2023)
-
Data-driven prediction of complex crystal structures of dense lithium
Nature Communications (2023)
-
The breakdown of both strange metal and superconducting states at a pressure-induced quantum critical point in iron-pnictide superconductors
Nature Communications (2023)
-
Record high Tc element superconductivity achieved in titanium
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.