Abstract
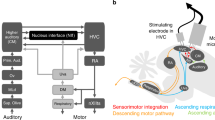
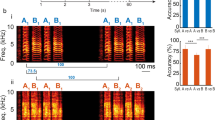
Sequences of motor activity are encoded in many vertebrate brains by complex spatio-temporal patterns of neural activity; however, the neural circuit mechanisms underlying the generation of these pre-motor patterns are poorly understood. In songbirds, one prominent site of pre-motor activity is the forebrain robust nucleus of the archistriatum (RA), which generates stereotyped sequences of spike bursts during song1 and recapitulates these sequences during sleep2. We show that the stereotyped sequences in RA are driven from nucleus HVC (high vocal centre), the principal pre-motor input to RA3,4. Recordings of identified HVC neurons in sleeping and singing birds show that individual HVC neurons projecting onto RA neurons produce bursts sparsely, at a single, precise time during the RA sequence. These HVC neurons burst sequentially with respect to one another. We suggest that at each time in the RA sequence, the ensemble of active RA neurons is driven by a subpopulation of RA-projecting HVC neurons that is active only at that time. As a population, these HVC neurons may form an explicit representation of time in the sequence. Such a sparse representation, a temporal analogue of the ‘grandmother cell’5 concept for object recognition, eliminates the problem of temporal interference during sequence generation and learning attributed to more distributed representations6,7.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Yu, A. C. & Margoliash, D. Temporal hierarchical control of singing in birds. Science 273, 1871–1875 (1996)
Dave, A. S. & Margoliash, D. Song replay during sleep and computational rules for sensorimotor vocal learning. Science 290, 812–816 (2000)
Nottebohm, F., Kelley, D. B. & Paton, J. A. Connections of vocal control nuclei in the canary telencephalon. J. Comp. Neurol. 207, 344–357 (1982)
Scharff, C. & Nottebohm, F. A comparative study of the behavioral deficits following lesions of various parts of the zebra finch song system: implications for vocal learning. J. Neurosci. 11, 2896–2913 (1991)
Barlow, H. The Cognitive Neurosciences 415–435 (MIT Press, Cambridge, Massachusetts, 1995)
Hermann, M., Hertz, J. & Prugel-Bennet, A. Analysis of synfire chains. Network: Comput. Neural Syst. 6(3), 403–414 (1995)
Willshaw, D. J., Buneman, O. P. & Longuet-Higgins, H. C. Non-holographic associative memory. Nature 222, 960–962 (1969)
Konishi, M. The role of auditory feedback in the control of vocalizations in the white-crowned sparrow. Z. Tierpsychol. 22, 770–783 (1965)
Konishi, M. Birdsong: from behavior to neuron. Ann. Rev. Neurosci. 8, 125–170 (1985)
Immelmann, K. in Bird Vocalizations (ed. Hinde, R. A.) 61–74 (Cambridge Univ. Press, New York, 1969)
Fee, M. S., Shraiman, B., Pesaran, B. & Mitra, P. P. The role of nonlinear dynamics of the syrinx in the vocalizations of a songbird. Nature 395, 67–71 (1998)
Vu, E. T., Mazurek, M. E. & Kuo, Y. Identification of a forebrain motor programming network for the learned song of zebra finches. J. Neurosci. 14, 6924–6934 (1994)
Vicario, D. S. Organization of the zebra finch song control system: II. Functional organization of outputs from nucleus robustus archistriatalis. J. Comp. Neurol. 309, 486–494 (1991)
Wild, J. M. Descending projections of the songbird nucleus robustus archistriatalis. J. Comp. Neurol. 338, 225–241 (1993)
Chi, Z. & Margoliash, D. Temporal precision and temporal drift in brain and behavior of zebra finch song. Neuron 32, 899–910 (2001)
Doya, K. & Sejnowski, T. J. in Central Auditory Processing and Neural Modeling (eds Poon, P. W. F. & Brugge, J. F.) 77–88 (Plenum, New York, 1998)
Troyer, T. W. & Doupe, A. J. An associational model of birdsong sensorimotor learning II. Temporal hierarchies and the learning of song sequence. J. Neurophysiol. 84, 1224–1239 (2000)
Mooney, R. Different subthreshold mechanisms underlie song selectivity in identified HVc neurons of the zebra finch. J. Neurosci. 201, 5420–5436 (2000)
Dutar, P., Vu, H. M. & Perkel, D. J. Multiple cell types distinguished by physiological, pharmacological, and anatomic properties in nucleus HVc of the adult zebra finch. J. Neurophysiol. 80, 1828–1838 (1998)
Swadlow, H. Neocortical efferent neurons with very slowly conducting axons: strategies for reliable antidromic identification. J. Neurosci. Meth. 79, 131–141 (1998)
Brillinger, D. R., Bryant, H. L. & Segundo, J. P. Identification of synaptic interactions. Biol. Cybernetics 22, 213–228 (1976)
Doupe, A. J. A neural circuit specialized for vocal learning. Curr. Opin. Neurobiol. 116, 104–111 (1993)
Amari, S.-I. Learning patterns and pattern sequences by self-organizing nets of threshold elements. IEEE Trans. Comput. 1197–1206 (1972)
Kleinfeld, D. & Sompolinsky, H. in Methods in Neuronal Modeling (eds Koch, C. & Segev, I.) 195–246 (MIT Press, Cambridge, Massachusetts, 1989)
Pearlmutter, B. A. Learning state space trajectories in recurrent neural networks. Neural Computat. 1, 263–269 (1989)
Bottjer, S. W., Halsema, K. A., Brown, S. A. & Miesner, E. A. Axonal connections of a forebrain nucleus involved with vocal learning in zebra finches. J. Comp. Neurol. 279, 312–326 (1989)
Mooney, R. Synaptic basis for developmental plasticity in a birdsong nucleus. J. Neurosci. 12, 2464–2477 (1992)
Brainard, M. S. & Doupe, A. J. Interruption of a basal ganglia-forebrain circuit prevents plasticity of learned vocalizations. Nature 404, 762–766 (2000)
Phillips, N. H. & Berger, R. J. Melatonin infusions restore sleep suppressed by continuous bright light in pigeons. Neurosci. Lett. 145, 217–220 (1992)
Fee, M. S. & Leonardo, A. Miniature motorized microdrive and commutator system for chronic neural recordings in small animals. J. Neurosci. Meth. 112, 83–94 (2001)
Acknowledgements
We acknowledge discussions with W. Denk, D. Lee, I. Nebel and S. Seung. We also thank F. Nottelbohm for comments on the manuscript. Recordings of RA neurons in the singing bird were carried out in collaboration with A. Leonardo. This work was supported in part by the National Science Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Hahnloser, R., Kozhevnikov, A. & Fee, M. An ultra-sparse code underliesthe generation of neural sequences in a songbird. Nature 419, 65–70 (2002). https://doi.org/10.1038/nature00974
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature00974
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.