Abstract
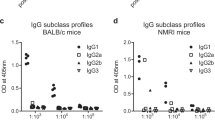
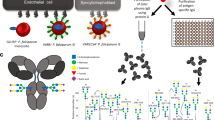
The malaria parasite Plasmodium falciparum infects 5–10% of the world's population and kills two million people annually1. Fatalities are thought to result in part from pathological reactions initiated by a malarial toxin. Glycosylphosphatidylinositol (GPI) originating from the parasite has the properties predicted of a toxin2,3,4,5,6; however, a requirement for toxins in general and GPI in particular in malarial pathogenesis and fatality remains unproven. As anti-toxic vaccines can be highly effective public health tools, we sought to determine whether anti-GPI vaccination could prevent pathology and fatalities in the Plasmodium berghei/rodent model of severe malaria. The P. falciparum GPI glycan of the sequence NH2-CH2-CH2-PO4-(Manα1-2)6Manα1-2Manα1-6Manα1-4GlcNH2α1-6myo-inositol-1,2-cyclic-phosphate was chemically synthesized, conjugated to carriers, and used to immunize mice. Recipients were substantially protected against malarial acidosis, pulmonary oedema, cerebral syndrome and fatality. Anti-GPI antibodies neutralized pro-inflammatory activity by P. falciparum in vitro. Thus, we show that GPI is a significant pro-inflammatory endotoxin of parasitic origin, and that several disease parameters in malarious mice are toxin-dependent. GPI may contribute to pathogenesis and fatalities in humans. Synthetic GPI is therefore a prototype carbohydrate anti-toxic vaccine against malaria.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
World Health Organization World malaria situation 1990. World Health Stat. Q. 45, 257–266 (1992)
Schofield, L. & Hackett, F. Signal transduction in host cells by a glycosylphosphatidylinositol toxin of malaria parasites. J. Exp. Med. 177, 145–153 (1993)
Tachado, S. D. & Schofield, L. Glycosylphosphatidylinositol toxin of Trypanosoma brucei regulates IL-1α and TNF-α expression in macrophages by protein tyrosine kinase mediated signal transduction. Biochem. Biophys. Res. Commun. 205, 984–991 (1994)
Schofield, L. et al. Glycosylphosphatidylinositol toxin of Plasmodium upregulates intercellular adhesion molecule-1, vascular cell adhesion molecule-1, and E-selectin expression in vascular endothelial cells and increases leukocyte and parasite cytoadherence via tyrosine kinase-dependent signal transduction. J. Immunol. 156, 1886–1896 (1996)
Tachado, S. D. et al. Glycosylphosphatidylinositol toxin of plasmodium induces nitric oxide synthase expression in macrophages and vascular endothelial cells by a protein tyrosine kinase-dependent and protein kinase C-dependent signalling pathway. J. Immunol. 156, 1897–1907 (1996)
Tachado, S. D. et al. Signal transduction in macrophages by glycosylphosphatidylinositols of Plasmodium, Trypanosoma and Leishmania: activation of protein tyrosine kinases and protein kinase C by inositolglycan and diacylglycerol moieties. Proc. Natl Acad. Sci. USA 94, 4022–4027 (1997)
Grau, G. E., Taylor, T. E., Molyneux, M. E., Wirima, J. J. & Vassalli, P. Tumor necrosis factor and disease severity in children with falciparum malaria. N. Engl. J. Med. 320, 1586–1591 (1989)
Turner, G. D. H. et al. An immunohistochemical study of the pathology of fatal malaria. Evidence for widespread endothelial activation and a potential role for intercellular adhesion molecule-1 in cerebral sequestration. Am. J. Pathol. 145, 1057–1069 (1994)
Magez, S. et al. The glycosyl-inositol-phosphate and dimyristoylglycerol moieties of the glycosylphosphatidylinositol anchor of the trypanosome variant-specific surface glycoprotein are distinct macrophage-activating factors. J. Immunol. 160, 1949–1956 (1998)
Almeida, I. C. et al. Highly purified glycosylphosphatidylinositols from Trypanosoma cruzi are potent proinflammatory agents. EMBO J. 19, 1476–1485 (2000)
Golgi, C. Sull' infezione malarica. Arch. Sci. med. (Torino) 10, 109–135 (1886)
Dieckmann-Schuppert, A., Bender, S., Odenthal-Schnittler, M., Bause, E. & Schwarz, R. T. Apparent lack of N-glycosylation in the asexual intraerythrocytic stage of Plasmodium falciparum. Eur. J. Biochem. 205, 815–825 (1992)
Dieckmann-Schuppert, A., Bause, E. & Schwarz, R. T. Studies on O-glycans of Plasmodium falciparum-infected human erythrocytes: evidence for O-GlcNAc and O-GlcNAc-transferase in malaria parasites. Eur. J. Biochem. 216, 779–788 (1993)
Berhe, S., Schofield, L., Schwarz, R. T. & Gerold, P. Conservation of structure among glycosylphosphatidyliositol toxins from different geographic isolates of Plasmodium falciparum. Mol. Biochem. Parasitol. 103, 273–278 (1999)
Gowda, D. C., Gupta, P. & Davidson, E. A. Glycosylphosphatidylinositol anchors represent the major carbohydrate modification in proteins of intraerythrocytic stage Plasmodium falciparum. J. Biol. Chem. 272, 6428–6439 (1997)
Gerold, P., Dieckmann-Schuppert, A. & Schwarz, R. T. Glycosylphosphatidylinositols synthesized by asexual erythrocytic stages of the malarial parasite, Plasmodium falciparum. Candidates for plasmodial glycosylphosphatidylinositol membrane anchor precursors and pathogenicity factors. J. Biol. Chem. 269, 2597–2606 (1994)
McConville, M. J. & Ferguson, M. A. J. The structure, biosynthesis and function of glycosylated phosphatidylinositols in the parasitic protozoa and higher eukaryotes. Biochem. J. 294, 305–324 (1993)
White, N. J. & Ho, M. The pathophysiology of malaria. Adv. Parasitol. 31, 83–173 (1992)
Grau, G. E. et al. Tumor necrosis factor (cachectin) as an essential mediator in murine cerebral malaria. Science 237, 1210–1212 (1987)
Grau, G. E. et al. Monoclonal antibody against interferon γ can prevent experimental cerebral malaria and its associated overproduction of tumour necrosis factor. Proc. Natl Acad. Sci. USA 86, 5572–5574 (1989)
Grau, G. E. et al. Late administration of monoclonal antibody to leukocyte function-antigen 1 abrogates incipient murine cerebral malaria. Eur. J. Immunol. 21, 2265–2267 (1991)
Jennings, V. M., Actor, J. K., Lal, A. A. & Hunter, R. L. Cytokine profile suggesting that murine cerebral malaria is an encephalitis. Infect. Immun. 65, 4883–4887 (1997)
Chang, W. L. et al. CD8(+ )-T-cell depletion ameliorates circulatory shock in Plasmodium berghei-infected mice. Infect. Immun. 69, 7341–7348 (2001)
Miller, L. H., Baruch, D. I., Marsh, K. & Doumbo, O. K. The pathogenic basis of malaria. Nature 415, 673–679 (2002)
de Souza, B. J. & Riley, E. M. Cerebral malaria: the contribution of studies in animal models to our understanding of immunopathogenesis. Microbes Infect. 4, 291–300 (2002)
Christophers, S. R. The mechanism of immunity against malaria in communities living under hyperendemic conditions. Ind. J. Med. Res. 12, 273–294 (1924)
Sinton, J. A. A summary of our present knowledge of the mechanism of immunity in malaria. J. Malaria Inst. India 2, 71–83 (1939)
McGregor, I. A., Giles, H. M., Walters, J. H., Davies, A. H. & Pearson, F. A. Effects of heavy and repeated malarial infections on Gambian infants and children. Br. Med. J. 2, 686–692 (1956)
Naik, R. S. et al. Glycosylphosphatidylinositol anchors of Plasmodium falciparum: molecular characterization and naturally elicited antibody response that may provide immunity to malaria pathogenesis. J. Exp. Med. 192, 1563–1576 (2000)
Schofield, F. Selective primary health care: strategies for control of disease in the developing world. XXII. Tetanus: a preventable problem. Rev. Infect. Dis. 8, 144–156 (1986)
Acknowledgements
This work was supported by the UNDP/World Bank/World Health Organization Special Program for Research and Training in Tropical Diseases, the National Institutes of Health, the Human Frontiers of Science Program and the National Health and Medical Research Council. M.C.H. acknowledges a biotechnology training grant fellowship from the NIH. L.S. is an International Research Scholar of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Schofield, L., Hewitt, M., Evans, K. et al. Synthetic GPI as a candidate anti-toxic vaccine in a model of malaria. Nature 418, 785–789 (2002). https://doi.org/10.1038/nature00937
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature00937
This article is cited by
-
Therapeutics through glycobiology: an approach for targeted elimination of malaria
Biologia (2023)
-
Phosphatidylinositol synthesis, its selective salvage, and inter-regulation of anionic phospholipids in Toxoplasma gondii
Communications Biology (2020)
-
Characterization and immune regulation role of an immobilization antigen from Cryptocaryon irritans on groupers
Scientific Reports (2019)
-
IgG antibodies to synthetic GPI are biomarkers of immune-status to both Plasmodium falciparum and Plasmodium vivax malaria in young children
Malaria Journal (2017)
-
TOLLIP gene variant is associated with Plasmodium vivax malaria in the Brazilian Amazon
Malaria Journal (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.