Abstract
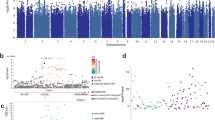
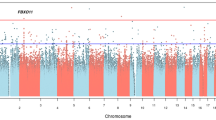
In developed countries, the majority of all violent crime is committed by a small group of antisocial recidivistic offenders, but no genes have been shown to contribute to recidivistic violent offending or severe violent behavior, such as homicide. Our results, from two independent cohorts of Finnish prisoners, revealed that a monoamine oxidase A (MAOA) low-activity genotype (contributing to low dopamine turnover rate) as well as the CDH13 gene (coding for neuronal membrane adhesion protein) are associated with extremely violent behavior (at least 10 committed homicides, attempted homicides or batteries). No substantial signal was observed for either MAOA or CDH13 among non-violent offenders, indicating that findings were specific for violent offending, and not largely attributable to substance abuse or antisocial personality disorder. These results indicate both low monoamine metabolism and neuronal membrane dysfunction as plausible factors in the etiology of extreme criminal violent behavior, and imply that at least about 5–10% of all severe violent crime in Finland is attributable to the aforementioned MAOA and CDH13 genotypes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hamparin DM, Schuster R, Dinstz S, Conrad JP . The Violent Few: A Study of Violent Offenders. Lexington Books: Lexington, MA, 1978.
Tracy PE, Wolfgang ME, Figlio RM . Delinquency in Two Birth Cohorts. Plenum Press: New York, 1990.
Ferguson CJ . Genetic contributions to antisocial personality and behavior: a meta-analytic review from an evolutionary perspective. J Soc Psychol 2010; 150: 160–180.
Mednick SA, Gabrielli WH, Hutchings B . Genetic influences in crime convictions: evidence from and adoption cohort. Science 1984; 224: 891–894.
Hjalmarsson R, Lindquist MJ . The origins of intergenerational associations in crime: Lessons from Swedish adoption data. Labour Econom 2013; 20: 68–81.
Brunner HG, Nelen M, Breakefield XO, Ropers HH, van Oost BA . Abnormal behavior associated with a point mutation in the structural gene for monoamine oxidase A. Science 1993; 262: 578–580.
Caspi A, McClay J, Moffitt TE, Mill J, Martin J, Craig IW et al. Role of genotype in the cycle of violence in maltreated children. Science 2002; 297: 851–854.
Bevilacqua L, Doly S, Kaprio J, Yuan Q, Tikkanen R, Paunio T et al. A population-specific HTR2B stop codon predisposes to severe impulsivity. Nature 2010; 468: 1061–1066.
Aspinwall LG, Brown TR, Tabery J . The double-edged sword: does biomechanism increase or decrease judges' sentencing of psychopaths? Science 2012; 337: 846–849.
Byrd AL, Manuck SB . MAOA, childhood maltreatment, and antisocial behavior: meta-analysis of a gene-environment interaction. Biol Psychiatry 2014; 75: 9–17.
Haberstick BC, Lessema JM, Hewitta JK, Smolena A, Hopferb CJ, Halpernc CT et al. MAOA genotype, childhood maltreatment, and their interaction in the etiology of adult antisocial behaviors. Biol Psychiatry 2014; 75: 25–30.
Kristiansson K, Perola M, Tikkanen E, Kettunen J, Surakka I, Havulinna AS et al. Genome-wide screen for metabolic syndrome susceptibility Loci reveals strong lipid gene contribution but no evidence for common genetic basis for clustering of metabolic syndrome traits. Circ Cardiovasc Genet 2012; 5: 242–249.
Vaara S, Nieminen MS, Lokki ML, Perola M, Pussinen PJ, Allonen J et al. Cohort Profile: The Corogene study. Int J Epidemiol 2012; 41: 1265–1271.
Lehti M . Henkirikoskatsaus 2013. OPTL Verkkokatsauksia 29/2013. Helsinki: Oikeuspoliittinen tutkimuslaitos, 2013. (in Finnish).
Dorfman HM, Meyer-Lindenberg A, Buckholtz JW . Neurobiological mechanisms for impulsive-aggression: The role of MAOA. Curr Top Behav Neurosci 2014, 17: 297–313.
Godar SC, Bortolato M, Castelli MP, Casti A, Casu A, Chen K et al. The aggression and behavioral abnormalities associated with monoamine oxidase A deficiency are rescued by acute inhibition of serotonin reuptake. J Psychiatr Res 2014; 56: 1–9.
Vartiainen H, Tiihonen J, Putkonen A, Koponen H, Virkkunen M, Hakola P, Lehto H . Citalopram, a selective serotonin reuptake inhibitor, in the treatment of aggression in schizophrenia. Acta Psychiatr Scand 1995; 91: 348–351.
Darke S . The toxicology of homicide offenders and victims: a review. Drug Alcohol Rev 2010; 29: 202–215.
Yanowitch R, Coccaro EF . The neurochemistry of human aggression. Adv Genet 2011; 75: 151–169.
Laaksonen E, Koski-Jännes A, Salaspuro M, Ahtinen H, Alho H . A randomized, multicentre, open-label, comparative trial of disulfiram, naltrexone and acamprosate in the treatment of alcohol dependence. Alcohol Alcohol 2008; 43: 53–61.
Tiihonen J, Krupitsky E, Verbitskaya E, Blokhina E, Mamontova O, Föhr J et al. Naltrexone implant for the treatment of polydrug dependence: a randomized controlled trial. Am J Psychiatry 2012; 169: 531–536.
Neale BM, Medland S, Ripke S, Anney RJL, Asherson P, Buitelaar J et al. Case-control genome-wide association study of attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 2010; 49: 906–920.
Sizoo B, van den Brink W, Franke B, Vasquez AA, van Wijngaarden-Cremers P, van der Gaag RJ . Do candidate genes discriminate patients with an autism spectrum disorder from those with attention deficit/hyperactivity disorder and is there an effect of lifetime substance use disorders? World J Biol Psychiatry 2010; 11: 699–708.
Arias-Vásquez A, Altink ME, Rommelse NN, Slaats-Willemse DI, Buschgens CJ, Fliers EA et al. CDH13 is associated with working memory performance in attention deficit/hyperactivity disorder. Genes Brain Behav 2011; 10: 844–851.
Edwards AC, Aliev F, Bierut LJ, Bucholz KK, Edenberg H, Hesselbrock V et al. Genome-wide association study of comorbid depressive syndrome and alcohol dependence. Psychiatr Genet 2012; 22: 31–41.
Kohannim O, Hibar DP, Stein JL, Jahanshad N, Hua X, Rajagopalan P et al. Discovery and replication of gene influences on brain structure using LASSO regression. Front Neurosci 2012; 6: 115.
Hart AB, Engelhardt BE, Wardle MC, Sokoloff G, Stephens M, Harriet de Wit et al. Genome-wide association study of d-amphetamine response in healthy volunteers identifies putative associations, including cadherin 13 (CDH13). PLoS One 2012; 7: e42646.
Lesca G, Rudolf G, Labalme A, Hirsch E, Arzimanoglou A, Genton P et al. Epileptic encephalopathies of the Landau-Kleffner and continuous spike and waves during slow-wave sleep types: genomic dissection makes the link with autism. Epilepsia 2012; 53: 1526–1538.
Mavroconstanti T, Johansson S, Winge I, Knappskog PM, Haavik J . Functional properties of rare missense variants of human CDH13 found in adult attention deficit/hyperactivity disorder (ADHD) patients. PLoS One 2013; 8: e71445.
Rivero O, Sich S, Popp S, Schmitt A, Franke B, Lesch KP . Impact of the ADHD-susceptibility gene CDH13 on development and function of brain networks. Eur Neuropsychopharmacol 2013; 23: 492–507.
GROUP investigators Børglum AD, GROUP investigators Demontis D, GROUP investigators Grove J, GROUP investigators Pallesen J, GROUP investigators Hollegaard MV, GROUP investigators Pedersen CB et al. GROUP investigators Genome-wide study of association and interaction with maternal cytomegalovirus infection suggests new schizophrenia loci. Mol Psychiatry 2014; 19: 325–333.
Mavroconstanti T, Halmøy A, Haavik J . Decreased serum levels of adiponectin in adult attention deficit hyperactivity disorder. Psychiatry Res 2014; 216: 123–130.
Xu W, Cohen-Woods S, Chen Q, Noor A, Knight J, Hosang G et al. Genome-wide association study of bipolar disorder in Canadian and UK populations corroborates disease loci including SYNE1 and CSMD1. BMC Med Genet 2014; 15: 2.
Lundström S, Forsman M, Larsson H, Kerekes N, Serlachius E, Långström N et al. Childhood neurodevelopmental disorders and violent criminality: A sibling control study. J Autism Dev Disord advance online publication, 27 June 2013 (e-pub ahead of print).
Satterfield JH, Faller KJ, Crinella FM, Schell AM, Swanson JM, Homer LD . A 30-year prospective follow-up study of hyperactive boys with conduct problems: adult criminality. J Am Acad Child Adolesc Psychiatry 2007; 46: 601–610.
GTEx. Genotype-Tissue Expression Portalhttp://www.gtexportal.org/home/searchEqtlsaccessed May 27, 2014.
Doyle AE, Faraone SV, McGrath L, Thermenos H, Juelich R, Chaponis J et al. Multivariate association of CDH13 variants and cortical thickness in ADHD. Society of Biological Psychiatry 2014 Annual Meeting, New York, 2014. Biol Psychiatry 2014; 75: 347S.
Young S, Moss D, Sedgwick O, Fridman M, Hodgkins P . A meta-analysis of the prevalence of attention deficit hyperactivity disorder in incarcerated populations. Psychol Med 2014; 7: 1–12.
Paradis S, Harrar DB, Lin Y, Koon AC, Hauser JL, Griffith EC et al. An RNAi-based approach identifies molecules required for glutamatergic and GABAergic synapse development. Neuron 2007; 53: 217–232.
Cloninger CR, Christiansen KO, Reich T, Gottesman II . Implications of sex differences in the prevalences of antisocial personality, alcoholism, and criminality for familial transmission. Arch Gen Psychiatry 1978; 35: 941–951.
Carey G, Gottesman II . Genes and antisocial behavior: perceived versus real threats to jurisprudence. J Law Med Ethics 2006; 34: 342–351.
Tiihonen J, Hakola P . Psychiatric disorders and homicide recidivism. Am J Psychiatry 1994; 151: 436–438.
Acknowledgements
We thank Auli Toivola for her contribution in the Sequenom Mass Array and MAOA VNTR genotyping and Aija Räsänen for secretarial assistance. The study was funded by the Finnish Ministry of Health and Social Affairs through the development fund for Niuvanniemi Hospital, Finland. Hanna M Ollila has received funds from Instrumentarium Science Foundation and Orion-Farmos Research Foundation. Kati Kristiansson has received grant from Orion-Farmos Research Foundation and Academy of Finland (grant number 250207).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Supplementary information
Rights and permissions
About this article
Cite this article
Tiihonen, J., Rautiainen, MR., Ollila, H. et al. Genetic background of extreme violent behavior. Mol Psychiatry 20, 786–792 (2015). https://doi.org/10.1038/mp.2014.130
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2014.130
This article is cited by
-
Targeting monoamine oxidase A: a strategy for inhibiting tumor growth with both immune checkpoint inhibitors and immune modulators
Cancer Immunology, Immunotherapy (2024)
-
Genetische Studien an forensisch Untergebrachten?
Der Nervenarzt (2024)
-
The Evolution of Forensic Genomics: Regulating Massively Parallel Sequencing
Journal of Bioethical Inquiry (2023)
-
Targeting monoamine oxidase A-regulated tumor-associated macrophage polarization for cancer immunotherapy
Nature Communications (2021)
-
Association of serotonin system-related genes with homicidal behavior and criminal aggression in a prison population of Pakistani Origin
Scientific Reports (2021)