Abstract
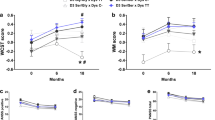
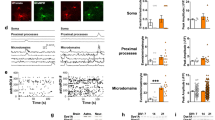
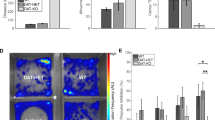
Dysbindin-1 regulates D2-receptor trafficking and is implicated in schizophrenia and related cognitive abnormalities, but whether this molecular effect mediates the clinical manifestations of the disorder is unknown. We explored in dysbindin-1-deficient mice (dys−/−) (1) schizophrenia-related behaviors, (2) molecular and electrophysiological changes in medial prefrontal cortex (mPFC) and (3) the dependence of these on D2-receptor stimulation. Dysbindin-1 disruption altered dopamine-related behaviors and impaired working memory under challenging/stressful conditions. Dys−/− pyramidal neurons in mPFC layers II/III were hyperexcitable at baseline but hypoexcitable following D2 stimulation. Dys−/− were also respectively more and less sensitive to D2 agonist- and antagonist-induced behavioral effects. Dys−/− had reduced expression of Ca2+/calmodulin-dependent protein kinase II (CaMKII) and CaMKKβ in mPFC. Chronic D2 agonist treatment reproduced these changes in protein expression, and some of the dys−/− behavioral effects. These results elucidate dysbindin's modulation of D2-related behavior, cortical activity and mPFC CaMK components, implicating cellular and molecular mechanisms of the association of dysbindin with psychosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Talbot K, Ong WY, Blake DJ, Tang J, Louneva N, Carlson GC et al. Dysbindin-1 and its protein family. In: Javitt D, Kantorowitz J (eds). Handbook of Neurochemistry and Molecular Neurobiology, 3rd edn. Vol. 27 Springer: New York, 2009, pp 107–241.
Chen XW, Feng YQ, Hao CJ, Guo XL, He X, Zhou ZY et al. DTNBP1, a schizophrenia susceptibility gene, affects kinetics of transmitter release. J Cell Biol 2008; 181: 791–801.
Burdick KE, Lencz T, Funke B, Finn CT, Szeszko PR, Kane JM et al. Genetic variation in DTNBP1 influences general cognitive ability. Hum Mol Genet 2006; 15: 1563–1568.
Fallgatter AJ, Herrmann MJ, Hohoff C, Ehlis AC, Jarczok TA, Freitag CM et al. DTNBP1 (dysbindin) gene variants modulate prefrontal brain function in healthy individuals. Neuropsychopharmacology 2006; 31: 2002–2010.
Morris DW, Murphy K, Kenny N, Purcell SM, McGhee KA, Schwaiger S et al. Dysbindin (DTNBP1) and the biogenesis of lysosome-related organelles complex 1 (BLOC-1): main and epistatic gene effects are potential contributors to schizophrenia susceptibility. Biol Psychiatry 2008; 63: 24–31.
Schwab SG, Knapp M, Mondabon S, Hallmayer J, Borrmann-Hassenbach M, Albus M et al. Support for association of schizophrenia with genetic variation in the 6p22.3 gene, dysbindin, in sib-pair families with linkage and in an additional sample of triad families. Am J Hum Genet 2003; 72: 185–190.
Straub RE, Jiang Y, MacLean CJ, Ma Y, Webb BT, Myakishev MV et al. Genetic variation in the 6p22.3 gene DTNBP1, the human ortholog of the mouse dysbindin gene, is associated with schizophrenia. Am J Hum Genet 2002; 71: 337–348.
Talbot K, Eidem WL, Tinsley CL, Benson MA, Thompson EW, Smith RJ et al. Dysbindin-1 is reduced in intrinsic, glutamatergic terminals of the hippocampal formation in schizophrenia. J Clin Invest 2004; 113: 1353–1363.
Weickert CS, Rothmond DA, Hyde TM, Kleinman JE, Straub RE . Reduced DTNBP1 (dysbindin-1) mRNA in the hippocampal formation of schizophrenia patients. Schizophr Res 2008; 98: 105–110.
Weickert CS, Straub RE, McClintock BW, Matsumoto M, Hashimoto R, Hyde TM et al. Human dysbindin (DTNBP1) gene expression in normal brain and in schizophrenic prefrontal cortex and midbrain. Arch Gen Psychiatry 2004; 61: 544–555.
Tang J, LeGros RP, Louneva N, Yeh L, Cohen JW, Hahn CG et al. Dysbindin-1 in dorsolateral prefrontal cortex of schizophrenia cases is reduced in an isoform-specific manner unrelated to dysbindin-1 mRNA expression. Hum Mol Genet 2009; 18: 3851–3863.
Williams NM, Preece A, Morris DW, Spurlock G, Bray NJ, Stephens M et al. Identification in 2 independent samples of a novel schizophrenia risk haplotype of the dystrobrevin binding protein gene (DTNBP1). Arch Gen Psychiatry 2004; 61: 336–344.
Iizuka Y, Sei Y, Weinberger DR, Straub RE . Evidence that the BLOC-1 protein dysbindin modulates dopamine D2 receptor internalization and signaling but not D1 internalization. J Neurosci 2007; 27: 12390–12395.
Ji Y, Yang F, Papaleo F, Wang HX, Gao WJ, Weinberger DR et al. Role of dysbindin in dopamine receptor trafficking and cortical GABA function. Proc Natl Acad Sci USA 2009; 106: 19593–19598.
Laruelle M, Kegeles LS, Abi-Dargham A . Glutamate, dopamine, and schizophrenia: from pathophysiology to treatment. Ann NY Acad Sci 2003; 1003: 138–158.
Goldman-Rakic PS . The cortical dopamine system: role in memory and cognition. Adv Pharmacol (San Diego, Calif) 1998; 42: 707–711.
Kumamoto N, Matsuzaki S, Inoue K, Hattori T, Shimizu S, Hashimoto R et al. Hyperactivation of midbrain dopaminergic system in schizophrenia could be attributed to the down-regulation of dysbindin. Biochem Biophys Res Commun 2006; 345: 904–909.
Li W, Zhang Q, Oiso N, Novak EK, Gautam R, O'Brien EP et al. Hermansky-Pudlak syndrome type 7 (HPS-7) results from mutant dysbindin, a member of the biogenesis of lysosome-related organelles complex 1 (BLOC-1). Nat Genet 2003; 35: 84–89.
Murotani T, Ishizuka T, Hattori S, Hashimoto R, Matsuzaki S, Yamatodani A . High dopamine turnover in the brains of Sandy mice. Neurosci Lett 2007; 421: 47–51.
Jentsch JD, Trantham-Davidson H, Jairl C, Tinsley M, Cannon TD, Lavin A . Dysbindin modulates prefrontal cortical glutamatergic circuits and working memory function in mice. Neuropsychopharmacology 2009; 34: 2601–2608.
Dickman DK, Davis GW . The schizophrenia susceptibility gene dysbindin controls synaptic homeostasis. Science 2009; 326: 1127–1130.
Tang TT, Yang F, Chen BS, Lu Y, Ji Y, Roche KW et al. Dysbindin regulates hippocampal LTP by controlling NMDA receptor surface expression. Proc Natl Acad Sci USA 2009; 106: 21395–21400.
Talbot K . The sandy (sdy) mouse: a dysbindin-1 mutant relevant to schizophrenia research. Prog Brain Res 2009; 179: 87–94.
Runyan JD, Moore AN, Dash PK . A role for prefrontal calcium-sensitive protein phosphatase and kinase activities in working memory. Learn Mem (Cold Spring Harbor, NY) 2005; 12: 103–110.
Gonzalez-Islas C, Hablitz JJ . Dopamine enhances EPSCs in layer II-III pyramidal neurons in rat prefrontal cortex. J Neurosci 2003; 23: 867–875.
Liu XY, Mao LM, Zhang GC, Papasian CJ, Fibuch EE, Lan HX et al. Activity-dependent modulation of limbic dopamine D3 receptors by CaMKII. Neuron 2009; 61: 425–438.
Papaleo F, Crawley JN, Song J, Lipska BK, Pickel J, Weinberger DR et al. Genetic dissection of the role of catechol-O-methyltransferase in cognition and stress reactivity in mice. J Neurosci 2008; 28: 8709–8723.
Takeuchi Y, Fukunaga K, Miyamoto E . Activation of nuclear Ca(2+)/calmodulin-dependent protein kinase II and brain-derived neurotrophic factor gene expression by stimulation of dopamine D2 receptor in transfected NG108-15 cells. J Neurochem 2002; 82: 316–328.
D'Este L, Casini A, Puglisi-Allegra S, Cabib S, Renda TG . Comparative immunohistochemical study of the dopaminergic systems in two inbred mouse strains (C57BL/6J and DBA/2J). J Chem Neuroanat 2007; 33: 67–74.
Takao K, Toyama K, Nakanishi K, Hattori S, Takamura H, Takeda M et al. Impaired long-term memory retention and working memory in sdy mutant mice with a deletion in Dtnbp1, a susceptibility gene for schizophrenia. Mol Brain 2008; 1: 11.
Cox MM, Tucker AM, Tang J, Talbot K, Richer DC, Yeh L et al. Neurobehavioral abnormalities in the dysbindin-1 mutant, sandy, on a C57BL/6J genetic background. Genes Brain Behav 2009; 8: 390–397.
Nguyen PV, Abel T, Kandel ER, Bourtchouladze R . Strain-dependent differences in LTP and hippocampus-dependent memory in inbred mice. Learn Mem 2000; 7: 170–179.
Kellendonk C, Simpson EH, Polan HJ, Malleret G, Vronskaya S, Winiger V et al. Transient and selective overexpression of dopamine D2 receptors in the striatum causes persistent abnormalities in prefrontal cortex functioning. Neuron 2006; 49: 603–615.
Vertes RP . Interactions among the medial prefrontal cortex, hippocampus and midline thalamus in emotional and cognitive processing in the rat. Neuroscience 2006; 142: 1–20.
Vijayraghavan S, Wang M, Birnbaum SG, Williams GV, Arnsten AF . Inverted-U dopamine D1 receptor actions on prefrontal neurons engaged in working memory. Nat Neurosci 2007; 10: 376–384.
Donohoe G, Morris DW, Clarke S, McGhee KA, Schwaiger S, Nangle JM et al. Variance in neurocognitive performance is associated with dysbindin-1 in schizophrenia: a preliminary study. Neuropsychologia 2007; 45: 454–458.
Arnsten AF . Stress impairs prefrontal cortical function in rats and monkeys: role of dopamine D1 and norepinephrine alpha-1 receptor mechanisms. Prog Brain Res 2000; 126: 183–192.
Feenstra MG, Botterblom MH . Rapid sampling of extracellular dopamine in the rat prefrontal cortex during food consumption, handling and exposure to novelty. Brain Res 1996; 742: 17–24.
Koch M . The neurobiology of startle. Prog Neurobiol 1999; 59: 107–128.
Braff DL, Geyer MA, Swerdlow NR . Human studies of prepulse inhibition of startle: normal subjects, patient groups, and pharmacological studies. Psychopharmacology 2001; 156: 234–258.
Arguello PA, Gogos JA . Modeling madness in mice: one piece at a time. Neuron 2006; 52: 179–196.
Hess G, Jacobs KM, Donoghue JP . N-methyl-D-aspartate receptor mediated component of field potentials evoked in horizontal pathways of rat motor cortex. Neuroscience 1994; 61: 225–235.
Miller R . Neural assemblies and laminar interactions in the cerebral cortex. Biol Cybern 1996; 75: 253–261.
Lewis DA, Gonzalez-Burgos G . Intrinsic excitatory connections in the prefrontal cortex and the pathophysiology of schizophrenia. Brain Res Bull 2000; 52: 309–317.
Tseng KY, O'Donnell P . Dopamine-glutamate interactions controlling prefrontal cortical pyramidal cell excitability involve multiple signaling mechanisms. J Neurosci 2004; 24: 5131–5139.
Wang M, Vijayraghavan S, Goldman-Rakic PS . Selective D2 receptor actions on the functional circuitry of working memory. Science 2004; 303: 853–856.
Zhang Y, Bertolino A, Fazio L, Blasi G, Rampino A, Romano R et al. Polymorphisms in human dopamine D2 receptor gene affect gene expression, splicing, and neuronal activity during working memory. Proc Natl Acad Sci USA 2007; 104: 20552–20557.
Druzin MY, Kurzina NP, Malinina EP, Kozlov AP . The effects of local application of D2 selective dopaminergic drugs into the medial prefrontal cortex of rats in a delayed spatial choice task. Behav Brain Res 2000; 109: 99–111.
Nagai T, Kitahara Y, Shiraki A, Hikita T, Taya S, Kaibuchi K et al. Dysfunction of dopamine release in the prefrontal cortex of dysbindin deficient sandy mice: an in vivo microdialysis study. Neurosci Lett 2010; 470: 134–138.
Santana N, Mengod G, Artigas F . Quantitative analysis of the expression of dopamine D1 and D2 receptors in pyramidal and GABAergic neurons of the rat prefrontal cortex. Cereb Cortex 2009; 19: 849–860.
Turrigiano GG, Nelson SB . Homeostatic plasticity in the developing nervous system. Nat Rev Neurosci 2004; 5: 97–107.
Seamans JK, Gorelova N, Durstewitz D, Yang CR . Bidirectional dopamine modulation of GABAergic inhibition in prefrontal cortical pyramidal neurons. J Neurosci 2001; 21: 3628–3638.
Seamans JK, Yang CR . The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog Neurobiol 2004; 74: 1–58.
Le Moine C, Gaspar P . Subpopulations of cortical GABAergic interneurons differ by their expression of D1 and D2 dopamine receptor subtypes. Brain Res Mol Brain Res 1998; 58: 231–236.
Meyer-Lindenberg AS, Olsen RK, Kohn PD, Brown T, Egan MF, Weinberger DR et al. Regionally specific disturbance of dorsolateral prefrontal-hippocampal functional connectivity in schizophrenia. Arch Gen Psychiatry 2005; 62: 379–386.
Lawrie SM, Buechel C, Whalley HC, Frith CD, Friston KJ, Johnstone EC . Reduced frontotemporal functional connectivity in schizophrenia associated with auditory hallucinations. Biol Psychiatry 2002; 51: 1008–1011.
Ford JM, Mathalon DH, Whitfield S, Faustman WO, Roth WT . Reduced communication between frontal and temporal lobes during talking in schizophrenia. Biol Psychiatry 2002; 51: 485–492.
Geyer MA, McIlwain KL, Paylor R . Mouse genetic models for prepulse inhibition: an early review. Mol Psychiatry 2002; 7: 1039–1053.
Weber M, Chang WL, Breier M, Ko D, Swerdlow NR . Heritable strain differences in sensitivity to the startle gating-disruptive effects of D2 but not D3 receptor stimulation. Behav Pharmacol 2008; 19: 786–795.
Halberstadt AL, Geyer MA . Habituation and sensitization of acoustic startle: opposite influences of dopamine D1 and D2-family receptors. Neurobiol Learn Mem 2009; 92: 243–248.
Plappert CF, Pilz PK, Schnitzler HU . Factors governing prepulse inhibition and prepulse facilitation of the acoustic startle response in mice. Behav Brain Res 2004; 152: 403–412.
Chausmer AL, Katz JL . The role of D2-like dopamine receptors in the locomotor stimulant effects of cocaine in mice. Psychopharmacology 2001; 155: 69–77.
Wang Y, Xu R, Sasaoka T, Tonegawa S, Kung MP, Sankoorikal EB . Dopamine D2 long receptor-deficient mice display alterations in striatum-dependent functions. J Neurosci 2000; 20: 8305–8314.
Khan ZU, Koulen P, Rubinstein M, Grandy DK, Goldman-Rakic PS . An astroglia-linked dopamine D2-receptor action in prefrontal cortex. Proc Natl Acad Sci USA 2001; 98: 1964–1969.
Peters M, Mizuno K, Ris L, Angelo M, Godaux E, Giese KP . Loss of Ca2+/calmodulin kinase kinase beta affects the formation of some, but not all, types of hippocampus-dependent long-term memory. J Neurosci 2003; 23: 9752–9760.
Acknowledgements
We thank Q Tian, L Erickson, K Jenkins, J Aney and G Carr for technical assistance. This research was supported by the Intramural Program of the NIH, National Institute of Mental Health.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Supplementary information
Rights and permissions
About this article
Cite this article
Papaleo, F., Yang, F., Garcia, S. et al. Dysbindin-1 modulates prefrontal cortical activity and schizophrenia-like behaviors via dopamine/D2 pathways. Mol Psychiatry 17, 85–98 (2012). https://doi.org/10.1038/mp.2010.106
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2010.106
Keywords
This article is cited by
-
α-Dystrobrevin knockout mice have increased motivation for appetitive reward and altered brain cannabinoid receptor 1 expression
Acta Neuropathologica Communications (2022)
-
Dysbindin-1A modulation of astrocytic dopamine and basal ganglia dependent behaviors relevant to schizophrenia
Molecular Psychiatry (2022)
-
Schizophrenia-associated dysbindin modulates axonal mitochondrial movement in cooperation with p150glued
Molecular Brain (2021)
-
The epistatic interaction between the dopamine D3 receptor and dysbindin-1 modulates higher-order cognitive functions in mice and humans
Molecular Psychiatry (2021)
-
Cortical copper transporter expression in schizophrenia: interactions of risk gene dysbindin-1
Journal of Neural Transmission (2021)