Abstract
Human papilloma virus (HPV) infection causes cancers and their precursors (high-grade squamous intraepithelial lesions) near cervical and anal squamocolumnar junctions. Recently described cervical squamocolumnar junction cells are putative residual embryonic cells near the cervical transformation zone. These cells appear multipotential and share an identical immunophenotype (strongly CK7-positive) with over 90% of high-grade squamous intraepithelial lesions and cervical carcinomas. However, because the number of new cervical cancers discovered yearly world wide is 17-fold that of anal cancer, we posed the hypothesis that this difference in cancer risk reflects differences in the transition zones at the two sites. The microanatomy of the normal anal transformation zone (n=37) and topography and immunophenotype of anal squamous neoplasms (n=97) were studied. A discrete anal transition zone was composed of multilayered CK7-positive/p63-negative superficial columnar cells and an uninterrupted layer of CK7-negative/p63-positive basal cells. The CK7-negative/p63-positive basal cells were continuous with—and identical in appearance to—the basal cells of the mature squamous epithelium. This was in contrast to the cervical squamocolumnar junction, which harbored a single-layered CK7-positive/p63-negative squamocolumnar junction cell population. Of the 97 anal intraepithelial neoplasia/squamous cell carcinomas evaluated, only 27% (26/97) appeared to originate near the anal transition zone and only 23% (22/97) were CK7-positive. This study thus reveals two fundamental differences between the anus and the cervix: (1) the anal transition zone does not harbor a single monolayer of residual undifferentiated embryonic cells and (2) the dominant tumor immunophenotype is in keeping with an origin in metaplastic (CK7-negative) squamous rather than squamocolumnar junction (CK7-positive) epithelium. The implication is that, at birth, the embryonic cells in the anal transition zone have already begun to differentiate, presenting a metaplasia that—similar to vaginal and vulvar epithelium—is less prone to HPV-directed carcinogenesis. This in turn underscores the link between cancer risk and a very small and discrete population of vulnerable squamocolumnar junction cells in the cervix.
Similar content being viewed by others
Main
Human papilloma virus (HPV) infection causes cervical cancer and its precursor lesions (HSIL), specifically at the squamocolumnar junction (squamocolumnar junction) near the transformation zone.1, 2, 3, 4 For decades, this topographical preference for cervical neoplasia has remained unexplained. However, in 2011, a population of residual embryonic cells was discovered at the gastroesophageal squamocolumnar junction that was linked to Barrett’s metaplasia.5 A subsequent study revealed a nearly identical population at the cervical squamocolumnar junction, and these cells were found to share an identical immunophenotype (including strong staining for CK7) with over 90% of high-grade squamous intraepithelial lesions and carcinomas.6 These shared properties coupled with the physical juxtaposition of squamocolumnar junction cells and cervical neoplasia support a carcinogenic sequence that initiates in the cervical squamocolumnar junction cells. In contrast, the mature metaplastic cervical epithelium, and mature squamous epithelium of the ectocervix, vulva, and vagina, are squamocolumnar junction marker-negative, implying carcinogenic HPV infection of non-squamocolumnar junction-type basal keratinocytes. This disparity in the target cell of origin between tumors in the two regions (cervical squamocolumnar junction vs lower genital tract squamous epithelium) has been postulated to explain why the number of new cervical cancers yearly world wide (~500 000) is nearly 20-fold that of vulvo-vaginal carcinomas (~25 000).6, 7
The anorectal junction is presumably another squamocolumnar junction that is similar to the cervix, where the squamous epithelium joins with the rectal mucosa at an ‘anal transition zone’. Epidemiological and molecular studies have shown that HPV is the causative agent of most anal carcinomas, with an estimated population attributable fraction of 88%.8, 9, 10, 11, 12, 13 It is noteworthy that HPV DNA is detected at least as frequently in the anus as in the cervix.14, 15, 16 Furthermore, a history of receptive anal intercourse is not a significant risk factor for anal HPV infection in women.14, 15 Finally, based on the rate of reported anal cancers in the world each year (~28 000), the cervical/anal cancer ratio is about 17:1.7 Thus, despite the high rates of anal HPV exposure, there is an inexplicably low rate of anal cancer in the world relative to the cervix.
Because the incidence rate of anal cancer was so similar to that of the vulva/vagina, we hypothesized that a study of the microanatomy of the cervical and anorectal transition zones might reveal differences to explain their disparate susceptibilities to HPV-related carcinogenesis. The purpose of this study was to (1) characterize the cellular phenotypes in the adult and fetal anal canals, (2) survey a spectrum of anal squamous lesions (low/high-grade squamous intraepithelial lesion, squamous cell carcinoma) for squamocolumnar junction marker expression, and (3) compare the findings with the cervical squamocolumnar junction.
Materials and methods
Rationale, Case Material, and Tissue Classification
The surgical anal canal is composed of three discrete epithelial zones (Figure 1a): (1) colorectal zone (uninterrupted colonic mucosa), (2) anal transitional zone (mixed epithelial type), and (3) squamous zone (uninterrupted squamous epithelium that merges into the perianal skin).17 ‘Normal’ anal canal tissue from adults (six abdominoperineal resections, 26 hemorrhoidectomy) and fetuses (five fetopsies/fetal abortal examinations <24 weeks of gestational age and considered surgical specimens; gestational age ranging from 14–22 weeks, estimated by foot length), in addition to neoplastic anal canal tissue (low grade squamous intraepithelial lesion, n=42; HSIL, n=36; squamous cell carcinoma, n=19), were retrieved from the archives of Brigham and Women's Hospital (Boston, MA, USA) and University of Arkansas Medical Center (Little Rock, AR, USA) with the approval of the Institutional Review Board at the respective institutions. All slides were reviewed (EJY) and categorized into the three zones of the anal canal, as described above. Discrepancies in interpretation were resolved by consensus review (EJY and CPC).
Microanatomy of the anal transition zone. (a) Schematic of the surgical anal canal. (b) Representative fetal anal canal with the squamous, transitional, and colorectal zones and their staining patterns with CK7 and p63. (c) Representative adult anal canal with the squamous and transitional zones and their staining patterns with CK7 and p63. The adult colorectal zone is depicted in Supplementary Figure 2.
Immunohistochemistry
Immunohistochemistry analyses were performed as previously described5, 6, 18 and as routinely performed at Brigham and Women’s Hospital, Department of Pathology. The following primary antibodies were used: CK7 (clone RCK105; Thermo Scientific, Waltham, MA, USA; used at concentration 1:400), p63 (Dako Corp), p16ink4 (Santa Cruz Biotechnology, Santa Cruz, CA, USA), and integrin α6 (Abcam, San Francisco, CA, USA; not routinely used for surgical pathology practice at Brigham and Women’s Hospital). Mouse and rabbit control IgG (Santa Cruz Biotechnology) were used as negative controls. CK7, p63, and p16 antibodies are routinely used for surgical pathology practice at Brigham and Women's Hospital.
Results
Microanatomy of the Anal Transition Zone
A total of 37 anal canals (32 adults and 5 fetuses) were evaluated. There was a discrete anal transitional zone (anal transition zone) epithelium that merged into the colorectal zone proximally and the squamous zone/perianal skin distally. This transitional epithelium was composed of multilayered columnar to cuboidal cells (Figure 1b and c, top row; ranging from one to nine layers) at the surface, with variable mucin production (scant to rare goblet cell metaplasia). Below this was a population of small basal cells that created an uninterrupted, 1–2 cell layers at the stromal interface. Anal glands with epithelium similar to the anal transition zone-type were present within the stroma. In the adult anal transition zone, there were occasional islands of squamous metaplastic epithelium (Supplementary Figure 1A and B) and scattered crypts of colorectal type. These epithelial heterogeneities (eg squamous, colorectal-type cells) were relatively rare in the fetal anal transition zone (one squamous metaplastic island in a 22-week gestational age fetopsy; Supplementary Figure 1C and D); however, the microanatomy of the adult and fetal anal canal (regardless of gestational age) was otherwise remarkably similar. The fetal anal transition zone length correlated with gestational age, increasing in length from 95 cells (14 weeks of gestational age) to 900 cells (22 weeks of gestational age) (mean=349±329; n=5). The length of the adult anal transition zone was difficult to evaluate because of frequent mucosal irregularities (eg mucosal ulceration, squamous metaplasia vs true squamous zone epithelium, etc); however, overall, the adult anal transition zone length varied greatly, ranging from abrupt transition (direct squamous to colorectal epithelial transition; n=4) up to ~700 cell lengths in a longitudinal section of an intact abdominoperineal resection specimen.
The cervical and gastroesophageal squamocolumnar junctions contain residual embryonic cells that are implicated in transition-zone-specific carcinomas and precursor lesions (ie squamous cell carcinoma/high grade squamous intraepithelial lesion in the cervix and esophageal carcinoma/Barrett’s esophagus at the gastroesophageal junction5, 6, 19, 20). These cells strongly express CK7, which is not or weakly expressed in the adjacent squamous and columnar epithelium, respectively. Given that the bladder (with known CK7 expression) and anus share a common embryonic origin,10 we predicted that the anal transition zone epithelium would also be CK7-positive. The multilayered surface epithelium of the anal transition zone (present in 88%; 28 of 32 specimens) stained strongly with CK7 (adult 100%, 28/28; fetal 100%, 5/5; Figure 1b and c and Table 1) but was negative for p63 (0%, 0/28). Scattered CK7-positive anal transition zone cells extended into the adjacent colorectal epithelium and over the surface of the stratified squamous epithelium; however, cells of the squamous and colorectal epithelium did not express CK7 (Figure 1,Supplementary Figure 2). The basal cells underlying the surface anal transition zone cells were p63-positive, CK7-negative, and were present as an uninterrupted layer at the stromal interface in all examined anal canals (100%, 28/28; Figure 1b and c, bottom row and Table 1). This basal cell population emerged at the colorectal/transition zone junction and continued into the squamous/perianal zone as basal keratinocytes. The overall CK7/p63 staining pattern was identical in the adult and fetal anal transition zone.
Anal Intraepithelial Neoplasia and Invasive Squamous Cell Carcinoma Arising from Anal Transition Zone vs Squamous Zone/Perianal Skin
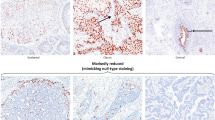
The findings are summarized in Table 2. Ninety-seven (97) total cases of HPV-related precursor lesions (n=78) and invasive squamous cell carcinoma (n=19) arising in the anal canal were evaluated. The lesions were divided into two micro-anatomical categories: (1) anal transition zone (based on the presence of anal transition zone-type epithelium) and (2) squamous zone/perianal skin (based on the presence of uninterrupted squamous epithelium and/or adnexal structures). Given the above criteria, 27% (26/97) of the lesions were present within the anal transition zone, and 73% (71/97) were present within the squamous zone/perianal skin. Of the lesions present in the anal transition zone, 64% (7/11) of low-grade squamous intraepithelial lesions, 100% (8/8) of high-grade squamous intraepithelial lesions, and 86% (6/7) of squamous cell carcinomas expressed patchy to strong/diffuse CK7 (Figure 2, top 3 rows). All CK7-positive lesions were arising from the anal transition zone epithelium. The four CK7-negative low-grade squamous intraepithelial lesions located at the anal transition zone were not arising from the anal transition zone epithelium but from the adjacent squamous epithelium (Supplementary Figure 3). Of the lesions present in the squamous zone/perianal skin, 0% (0/31) of low-grade squamous intraepithelial lesions, 0% (0/28) of high-grade squamous intraepithelial lesions, and 8% (1/12) of squamous cell carcinomas were positive for CK7 (Figure 2, bottom three rows). We speculate that the CK7-negative squamous cell carcinoma present at the anal transition zone and CK7-positive squamous cell carcinoma present at the squamous zone/perianal skin each originated in their respective zones (as expected by their immunophenotype), but were sampled in their nonnative zones into which they invaded/expanded.
Anal intraepithelial neoplasia and anal squamous cell carcinoma arising from the anal transition zone and squamous zone/perianal skin. The top nine panels represent low/high-grade intraepithelial lesions and squamous cell carcinoma arising from the anal transition zone, and their staining patterns for CK7 and p16. The lower nine panels represent low/high-grade intraepithelial lesions and squamous cell carcinoma arising from the squamous zone/perianal skin, and their staining patterns for CK7 and p16.
Discussion
This study has revealed distinct differences between the anus and the cervix with respect to the region where the caudal squamous epithelium undergoes transition to the more cephalad columnar epithelium. Development of this region in the anus entails the emergence of cells comprising a discrete, biphasic anal transition zone epithelium composed of CK7-positive/p63-negative multilayered superficial cells and an underlying CK7-negative/p63-positive population of basal cells. The CK7-positive/p63-negative superficial cells are multilayered with columnar to cuboidal morphology, with variable mucin production, and an apparent capacity for undergoing basal expansion to produce squamous metaplasia (as seen in adult and occasional fetal anal transition zone; Supplementary Figure 2). These cells are limited to the anal transition zone, with scattered cells present in the distal colorectal zone and along the surface of the proximal squamous zone. The CK7-negative/p63-positive basal cells compose the bottom 1–2 cell layers of the anal transition zone and are in direct contact with the basement membrane and anal stroma. These basal cells appear at the distal colorectal zone, continue uninterrupted throughout the anal transition zone, and merge into the squamous zone and perianal skin as basal keratinocytes. The immune-phenotypic findings are in keeping with previous studies that have evaluated the anal canal and its keratin staining patterns.17, 21, 22, 23
As shown in Results, the anal transition zone exhibits some similarity to the cervix transition zone. Both exhibit squamous metaplasia that appears to emerge from the CK7-positive cell population. However, in distinct contrast to the cervix, the anal transition zone does not contain a monolayer of CK7-positive cells.5, 6, 18 Unlike the anal transition zone where the CK7-positive cells define zones of basal cell expansion and squamous metaplasia, the cervical squamocolumnar junction is a single cell-layered epithelium that is directly superimposed on the basement membrane. These two differentiating characteristics seen in the anal transition zone—the multilayered CK7+ cells and the obligatory underlying basal cells—are illustrated in Figure 3.
Schematic representation of the cervical squamocolumnar junction (top panel) vs anal transition zone (lower panel). The cervical squamocolumnar junction cells (CK7-positive) are single layered and are in direct contact with the basement membrane. The anal transition zone cells (CK7-positive) are multilayered and always have an underlying layer of p63-positive basal cells. Blue=CK7-positive junctional cells; red=p63-positive basal cells.
We propose two possible explanations for why the anal transition zone differs from the cervical transition zone. First, a wholesale proliferation of basal cells does not occur with expulsion of the CK7-positive columnar cells, leaving a multilayer population capable of metaplasia without a separate solitary layer of residual embryonic cells clinging to the SC junction.18 An alternative explanation is a collision of CK7-negative/p63-positive basal cells (migrating proximally from the squamous epithelium) with the CK7-positive/p63-negative columnar population, the latter apically displaced to form the bilayered anal transition zone epithelium; this is a developmental model analogous to the formation of the embryonic gastroesophageal junction.19 From our analysis of the fetal anal transition zone, we know that the final form of the biphasic anal transition zone is already established at 14 weeks of gestational age and even earlier (9 weeks of gestational age) according to previous studies.23 A detailed evaluation of the fetuses at earlier gestational ages is necessary for further insight into the mechanism of anorectal junction formation. More practically, however, the question is how these differences in microanatomy of the anal transition zone and cervical squamocolumnar junction influence HPV-related carcinogenic susceptibility.
Of the 97 anal intraepithelial neoplasia/squamous cell carcinomas evaluated, only 22 (23%) were CK7-positive, suggesting that the cells in the anal transition zone are not uniquely vulnerable to carcinogenic HPV transformation. In contrast, nearly all high-grade squamous intraepithelial lesions and squamous cell carcinomas (as well as adenocarcinomas) of the cervix express CK7, as well as other squamocolumnar junction-related biomarkers.6, 18 Two potential differences between basal keratinocytes and squamocolumnar junction cells lie in their proximity to infectious viral particles and their susceptibility to carcinogenesis. Infection of the former requires injury to the epithelium that creates access to basal cells.24 In contrast, the single-layered cervical squamocolumnar junction cells are constantly exposed, making them more physically accessible for HPV infection. In addition to viral access, HPV attachment to the cell surface is an integral step to viral infection. Injury to the squamous epithelium disrupts the integrin α6β4/laminin-5 interaction that anchors basal cells to the basement membrane. Both components are thought to mediate HPV infection: laminin-5 through the attachment and sequestration of the virus at the basement membrane24 and integrin α6β4 as the putative HPV receptor on the cell surface.25, 26 By virtue of the cervical squamocolumnar junction cells having direct contact with the basement membrane, we would expect these cells to express integrins that anchor them to the basement membrane and coincidentally make them more susceptible to HPV infection. Indeed, our preliminary data show that cervical squamocolumnar junction cells and basal cells are enriched for integrin α6 expression, whereas minimal expression is seen in anal transition zone cells and expression is lost in the superficial keratinocytes (Supplementary Figure 4). Therefore, because the cervical squamocolumnar junction cells are (1) single layered and (2) have direct contact with the BM, they may be more susceptible to HPV infection compared with anal transition zone cells. Precisely why the squamocolumnar junction monolayer would be more susceptible to carcinogenesis remains a question requiring further study. However, target cell phenotype as a determinant of risk following HPV infection is a theme repeated throughout the multiple susceptible mucosal sites in the human genital tract and elsewhere.
In summary, the comparative microanatomy of the anal and cervical transition zones exposes distinct topographical differences that may influence cancer outcome. This observation supports the discrepancy in the incidences of cervical and anal carcinoma, despite both lesions arising at an ostensibly similar squamocolumnar junction. However, HPV infection alone does not determine the carcinogenic potential of the lesion, as evidenced by the majority of low-grade squamous intraepithelial lesions that do not progress to high-grade lesions or carcinoma. Further studies investigating the differences in the cellular and molecular biology of the target cells within the cervical and anal squamocolumnar junction are needed to address these additional questions.
References
Bosch FX, Lorincz A, Muñoz N et al. The causal relation between human papillomavirus and cervical cancer. J Clin Pathol 2002;55:244–265.
Ferenczy A, Franco E . Persistent human papillomavirus infection and cervical neoplasia. Lancet Oncol 2002;3:11–16.
Crum CP, Ikenberg H, Richart RM et al. Human papillomavirus type 16 and early cervical neoplasia. N Engl J Med 1984;310:880–883.
Marsh M . Original site of cervical carcinoma; topographical relationship of carcinoma of the cervix to the external os and to the squamocolumnar junction. Obstet Gynecol 1956;7:444–452.
Wang X, Ouyang H, Yamamoto Y et al. Residual embryonic cells as precursors of a Barrett's-like metaplasia. Cell 2011;145:1023–1035.
Herfs M, Yamamoto Y, Laury A et al. A discrete population of squamocolumnar junction cells implicated in the pathogenesis of cervical cancer. Proc Natl Acad Sci USA 2012;109:10516–10521.
Chaturvedi AK . Beyond cervical cancer: burden of other HPV-related cancers among men and women. J Adolesc Health 2010;46:S20–S26.
Frisch M, Glimelius B, van den Brule AJ et al. Sexually transmitted infection as a cause of anal cancer. N Engl J Med 1997;337:1350–1358.
Tilston P . Anal human papillomavirus and anal cancer. J Clin Pathol 1997;50:625–634.
Ryan DP, Compton CC, Mayer RJ . Carcinoma of the anal canal. N Engl J Med 2000;342:792–800.
Clark MA, Hartley A, Geh JI . Cancer of the anal canal. Lancet Oncol 2004;5:149–157.
Parkin DM . The global health burden of infection-associated cancers in the year 2002. Int J Cancer 2006;118:3030–3044.
Forman D, de Martel C, Lacey CJ et al. Global burden of human papillomavirus and related diseases. Vaccine 2012;30:F12–F23.
Palefsky JM, Holly EA, Ralston ML et al. Prevalence and risk factors for anal human papillomavirus infection in human immunodeficiency virus (HIV)-positive and high-risk HIV-negative women. J Infect Dis 2001;183:383–391.
Williams AB, Darragh TM, Vranizan K et al. Anal and cervical human papillomavirus infection and risk of anal and cervical epithelial abnormalities in human immunodeficiency virus-infected women. Obstet Gynecol 1994;83:205–211.
Hernandez BY, McDuffie K, Zhu X et al. Anal human papillomavirus infection in women and its relationship with cervical infection. Cancer Epidemiol Biomark Prev 2005;14:2550–2556.
Fenger C . The anal transitional zone. A method for macroscopic demonstration. Acta Pathol Microbiol Scand [A] 1978;86:225–230.
Herfs M, Vargas SO, Yamamoto Y et al. A novel blueprint for “top down” differentiation defines the cervical squamocolumnar junction during development, reproductive life, and neoplasia. J Pathol 2013;229:460–468.
Xian W, Ho KY, Crum CP et al. Cellular origin of Barrett's esophagus: controversy and therapeutic implications. Gastroenterology 2012;142:1424–1430.
Odze RD . Unraveling the mystery of the gastroesophageal junction: a pathologist’s perspective. Am J Gastroenterol 2005;100:1853–1867.
Williams GR, Talbot IC, Northover JM et al. Keratin expression in the normal anal canal. Histopathology 1995;26:39–44.
Williams GR, Talbot IC, Leigh IM . Keratin expression in anal carcinoma: an immunohistochemical study. Histopathology 1997;30:443–450.
Fritsch H, Zehm S, Illig R et al. New insights into the development and differentiation of the human anorectal epithelia. Are there clinical consequences? Int J Colorectal Dis 2010;25:1231–1242.
Roberts JN, Buck CB, Thompson CD et al. Genital transmission of HPV in a mouse model is potentiated by nonoxynol-9 and inhibited by carrageenan. Nat Med 2007;13:857–861.
Letian T, Tianyu Z . Cellular receptor binding and entry of human papillomavirus. Virol J 2010;7:2.
Huang H-S, Lambert PF . Use of an in vivo animal model for assessing the role of integrin α(6)β(4) and syndecan-1 in early steps in papillomavirus infection. Virology 2012;433:395–400.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Modern Pathology website
Rights and permissions
About this article
Cite this article
Yang, E., Quick, M., Hanamornroongruang, S. et al. Microanatomy of the cervical and anorectal squamocolumnar junctions: a proposed model for anatomical differences in HPV-related cancer risk. Mod Pathol 28, 994–1000 (2015). https://doi.org/10.1038/modpathol.2015.54
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2015.54
This article is cited by
-
Isolation and Characterization of Stem Cells from the Anal Canal Transition Zone in Pigs
Digestive Diseases and Sciences (2023)
-
Human papillomaviruses: diversity, infection and host interactions
Nature Reviews Microbiology (2022)
-
Epithelial expression of Gata4 and Sox2 regulates specification of the squamous–columnar junction via MAPK/ERK signaling in mice
Nature Communications (2021)
-
Opposing Wnt signals regulate cervical squamocolumnar homeostasis and emergence of metaplasia
Nature Cell Biology (2021)