Abstract
Fibrocytes contribute to the fibrotic changes most frequently observed in forms of asthma where inflammation is driven by T helper type 2 (Th2) cells. The mechanisms that regulate the profibrotic function of asthmatic fibrocytes are largely unknown. We isolated circulating fibrocytes from patients with allergen-exacerbated asthma, who showed the presence of fibrocytes, together with elevated concentrations of interleukin (IL)-4 and IL-13 and slightly increased concentrations of the Th17 cell-derived IL-17A, in induced sputum. Fibrocytes stimulated with IL-4 and IL-13 produced high levels of collagenous and non-collagenous matrix components and low levels of proinflammatory cytokines. Conversely, fibrocytes stimulated with IL-17A proliferated and released proinflammatory factors that may promote neutrophil recruitment and airway hyperresponsiveness. IL-17A also indirectly increased α-smooth muscle actin but not collagen expression in fibrocytes. Thus, fibrocytes may proliferate and express a predominant profibrotic or proinflammatory phenotype in asthmatic airways depending on the local concentrations of Th2- and Th17-derived cytokines.
Similar content being viewed by others
Introduction
Asthma is a heterogeneous and multifactorial disorder characterized by recurrent episodes of airflow obstruction, which are frequently triggered by exposure to environmental allergens or viral infections in predisposed individuals.1 Functional and histopathology features include dysfunction of the bronchial epithelium, exaggerated bronchoconstrictor response to a wide variety of stimuli (airway hyperresponsiveness), chronic airway inflammation, and structural changes of the bronchial wall.1, 2, 3, 4, 5 The mechanisms involved in the pathogenesis of structural alterations such as the thickening of the lamina reticularis (also known as reticular basement membrane) and increased smooth muscle mass are under active investigation because these changes are scarcely modified by current therapeutic options and may lead to irreversible airway narrowing2, 3 and accelerated loss of lung function.1
The thickening of the lamina reticularis is more frequently observed in forms of asthma where the inflammatory infiltrate is predominantly composed of T helper type 2 (Th2) cells and eosinophils, such as allergic asthma.6, 7, 8 It is particularly evident in patients with severe disease and persistent airflow obstruction.3, 6, 7, 9, 10 This thickening of the subepithelial reticular sheet reflects an increased deposition of collagen types I (COL1), III (COL3), and V (COL5).9, 11, 12 The non-fibrillar components of the lamina reticularis are also altered in asthma, with excessive deposition of extracellular matrix (ECM) molecules such as hyaluronan (HA), versican (VCAN), perlecan (heparan sulfate proteoglycan 2, HSPG2), and tenascin-C (TNC).11, 12, 13 These changes in the composition of the lamina reticularis are collectively termed “subepithelial fibrosis” and are thought to be caused by the abnormal accumulation of activated fibroblast- and myofibroblast-like cells beneath the asthmatic bronchial epithelium.2, 3, 9, 10, 11, 14 The origin of such cells is still debated, but there is increasing evidence that a substantial proportion of these cells exhibit the phenotypic characteristics of fibrocytes.15
The fibrocyte is a CD34+ cell with mixed leukocytic and mesenchymal features, coexpressing the pan-leukocyte marker CD45, the CD45RO isoform, CD11b, CD13, and COL1 gene and protein.15, 16, 17 Mature fibrocytes constitutively produce fibronectin, COL3, COL5, COL6, VCAN, HSPG2, and HA.18, 19 They have antigen-presenting activity and may modulate ongoing inflammatory responses through the release of a number of cytokines, chemokines, and growth factors.15, 16, 17 Fibrocytes coexpressing CD34 and COL1 mRNA or protein were initially detected in the bronchial mucosa of patients with mild allergic asthma, and their density in the subepithelial zone markedly increased within 24 h following the exposure to the clinically relevant allergen, during the allergen-induced asthmatic response.19 An appreciable proportion of these fibrocytes actively synthesizing COL1 also expressed the contractile protein α-smooth muscle actin (α-SMA).19 The proportion of COL1+ fibrocytes that also showed α-SMA immunoreactivity at 24 h following allergen exposure in this study19 was similar to that reported for subepithelial fibroblast-like cells in similarly allergen-exposed asthmatics.20 Subsequent studies21, 22 demonstrated the presence of CD34+COL1+ fibrocytes, coexpressing CD45RO and/or α-SMA, in the bronchial mucosa of patients with various levels of disease severity. In corticosteroid-naive allergic asthmatics, fibrocyte density in the lamina propria correlated with the thickness of the lamina reticularis.21 In patients with mild-to-severe refractory asthma, fibrocyte infiltration of the lamina propria strongly correlated with asthma severity, as classified on the basis of the treatment required to achieve disease control.22 The infiltrate was more prominent in those who had severe asthma refractory to treatment, and involved the underlying airway smooth muscle bundle in most patients.22 These studies also showed that there are very few or no fibrocytes in the bronchial mucosa of healthy individuals.
The functional characteristics of asthmatic fibrocytes and the mechanisms that regulate their profibrotic function are largely unknown. Moreover, it remains to be elucidated if these cells can modulate the inflammatory response, thereby affecting the asthma phenotype and response to treatment. In the present study, we isolated high numbers of mature fibrocytes from the peripheral blood of patients with allergen-exacerbated asthma, who showed appreciable numbers of fibrocytes in their airways, together with a marked increase in the release of the Th2-derived cytokines interleukin (IL)-4 and IL-13 and a less marked increase in the release of the Th17-derived cytokine IL-17A (also known as IL-17), as assessed by analysis of induced sputum. The asthmatic fibrocytes produced high levels of the major ECM components of the thickened lamina reticularis upon stimulation with IL-4 and IL-13, while they proliferated and released large amounts of factors that may promote neutrophil recruitment and increase bronchial smooth muscle contractility upon stimulation with IL-17A. With these observations, we provide the first evidence that the profibrotic and proinflammatory function of fibrocytes in asthma may be affected by the relative concentrations of Th2- and Th17-derived cytokines, which may vary at the tissue site in different clinical subphenotypes of the disease.6, 7, 8, 23, 24, 25
Results
High numbers of mature fibrocytes can be isolated from the peripheral blood of patients with allergen-exacerbated asthma
Twelve atopic asthmatics were recruited during an unscheduled visit for a moderate exacerbation of their disease, not requiring hospital admission or the use of oral corticosteroids according to standard criteria.26 All patients reported that their asthma exacerbation was triggered by exposure to the allergens to which they were sensitized, which had already caused moderate to severe worsening of their symptoms, also requiring emergency room visits, in the previous years. None had suffered from a recent respiratory infection. Sputum induction and collection of venous blood samples were performed on the day of the unscheduled visit and the clinical characteristics of these patients on that visit are reported in Table 1. Eight non-atopic, healthy individuals (four females; age range: 19–34 years) were recruited to serve as controls. Venous blood samples were collected into heparinized blood collection tubes and aliquots of the leukocyte-enriched fractions of whole blood were separately processed to enumerate circulating fibrocytes and to isolate these cells for functional evaluation.
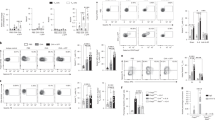
The numbers of circulating mature fibrocytes were determined on cytospin preparations of total peripheral blood leukocytes. For this analysis, cells coexpressing CD34 and COL1 or CD34 and the mRNA encoding the α chain of COL1 (COL1A1), were identified by using double immunofluorescence or double immunohistochemical and in-situ hybridization techniques analogous to those previously used for enumeration of fibrocytes in the bronchial mucosa of patients with allergen-induced asthma exacerbations.19 A similar procedure was adopted to enumerate fibrocytes in cytospins of cells isolated from induced sputum. The number of CD34+COL1+ cells in the peripheral blood of asthmatic patients ranged from 12.2 to 33.4 × 104 per ml (median: 20.2 × 104 per ml). These mature fibrocytes accounted for 88.9–96.4% of the CD34+ cells expressing COL1A1 mRNA. By contrast, the number of CD34+COL1+ cells in the peripheral blood of healthy controls was very low, ranging from 0.14 to 0.82 × 104 per ml (median: 0.37 × 104 per ml). The circulating mature fibrocytes in these subjects accounted for 72.8–89.3% of the CD34+ cells expressing COL1A1 mRNA. The differences between asthmatics and healthy controls were highly statistically significant (P<0.001) and similar to those observed when the numbers of circulating fibrocytes were expressed as percentage of total leukocytes (Figure 1). Analysis of the induced sputum confirmed previous findings19 that patients with an allergen-exacerbated asthma have appreciable numbers of fibrocytes migrating toward the bronchial epithelium (Table 2). In comparison with the healthy controls, the asthmatic subjects also showed sputum eosinophilia, a significant increase in the absolute numbers of lymphocytes and neutrophils, a marked increase in the levels of sputum IL-4 and IL-13 and a less marked, but significant increase, in the sputum concentrations of IL-17A (Table 2).
Comparison of the frequency of CD34+COL1+ (a) and CD34+COL1A1 mRNA+ fibrocytes (b) in the peripheral blood of healthy controls and asthmatic patients. The percentage of cells coexpressing CD34 and COL1 or CD34 and COL1A1 mRNA was calculated on cytospins of total peripheral blood leukocytes. The horizontal lines indicate the medians. ***P<0.001 vs. healthy controls by the Mann–Whitney test.
To obtain a pure population of circulating fibrocytes for phenotypic analysis and in-vitro testing, CD34+ cells were initially separated from total peripheral blood leukocytes by positive immunomagnetic selection. The resulting population was composed of ≥83.4% CD34+CD45+ cells, and the percentage of CD34+CD45+COL1+ fibrocytes ranged from 68.7 to 80.9%, as assessed by flow cytometry (Figure 2a). To further enrich the population of fibrocytes, the adherent fraction of the isolated cells was incubated for 5 days in culture medium supplemented with 20% autologous serum. During this period, the cells uniformly acquired the typical elongated, spindle-like shape that characterizes cultured fibrocytes15, 16, 17, 19, 21, 22 (Figure 2b). On day 5, COL1 immunoreactivity was detected in more than 96% of the harvested cells (Figure 2c). These fibrocytes were tested for the expression of the receptors for IL4, IL-13, and IL-17A by flow cytometry. IL-4 and IL-13 share a common receptor, the type II IL-4 receptor (IL-4Rα/IL-13Rα1),27, 28 and IL-13 signaling through IL-4Rα appears to have a clinically relevant role in allergen-induced asthma.27 In comparison with normal fibrocytes, asthmatic fibrocytes expressed higher levels of IL-4Rα, IL-13Rα1, and the high-affinity receptor for IL-17A, IL-17RA (Figure 2d). The response of the asthmatic fibrocytes to stimulation with IL-4, IL-13, and IL-17A was therefore assessed in the experiments described below, using serum-free media and concentrations of recombinant human cytokines that had been estimated as optimal in preliminary studies (5 ng ml−1 for IL-4 and IL-13, 15 ng ml−1 for IL-17A). The numbers of fibrocytes recovered from the peripheral blood of healthy controls were too low for a similar functional analysis, which was purposely conducted without prior expansion of circulating fibrocytes in long-term cultures as this procedure might alter the phenotypic characteristics of the cells.
Isolation and phenotypic analysis of circulating fibrocytes. CD34+ cells were isolated from the peripheral blood by positive immunomagnetic selection and cultured for 5 days to obtain a pure population of mature fibrocytes. The non-adherent cells were removed after the first 48 h and the adherent cells were reincubated for the next 3 days in fresh medium. Flow cytometric analysis of the isolated cells was performed soon after the immunomagnetic selection (a) and on day 5 (c). The horizontal and vertical lines in the representative dot plots mark fluorescence intensity greater than that observed with isotype-matched controls for the anti-CD45, anti-CD34, and anti-COL1 antibodies. The morphology of the cells in culture was monitored under an inverted microscope and the microphotographs in panel b show representative images in phase-contrast mode taken at the indicated points in time. The expression of the type I interleukin (IL)-4 receptor subunits (IL-4Rα/common γ-chain (γc)), type II IL-4 receptor subunits (IL-4Rα/IL-13Rα1), IL-13Rα2, and IL-17RA on the membrane of control and asthmatic fibrocytes were evaluated by flow cytometry on day 5 (d). The relative level of expression was quantified by subtracting the median fluorescence intensity (MFI) of cells stained with each specific antibody from the MFI of cells stained with the corresponding isotype control and dividing the obtained value by the MFI of unstained cells. Data are expressed as the means and s.d. *P<0.05 vs. control fibrocytes; **P<0.01 vs. control fibrocytes by the unpaired Student's t-test; n=5.
IL-4 and IL-13 increase the production of ECM molecules and profibrotic products in asthmatic fibrocytes
Stimulation of asthmatic fibrocytes with IL-4 or IL-13 similarly induced increased expression of the mRNAs encoding the α1 chain of COL3 (COL3A1), the α2 chain of COL5 (COL5A2), TNC, and the core proteins of VCAN and HSPG2 (Figure 3a). The release of fibrillar collagens, including COL1, COL3, and COL5, was also significantly enhanced in cultures of fibrocytes stimulated with either cytokines, as compared with cultures of non-stimulated cells (Figure 3b). Furthermore, either IL-4 or IL-13 induced a substantial increase in the production of HA and TNC in fibrocytes (Figure 3c) and augmented the expression of α-SMA in these cells (Figure 3d).
Changes in the expression of extracellular matrix (ECM) molecules and α-smooth muscle actin (α-SMA) upon stimulation of asthmatic fibrocytes with interleukin (IL)-4 or IL-13 for up to 72 h. The expression of the mRNA for the indicated ECM molecules (a) was evaluated at 24 h by real-time reverse transcriptase-PCR. The level of expression of the mRNA encoding each target gene was normalized to the level of expression of the mRNA encoding the reference gene, ubiquitin C. The amounts of released fibrillar collagens ((fCOLs) (b), hyaluronan (HA) and tenascin-C (TNC) (c) were measured in the supernatants at 24 h (b) or at 48 h (c) by using colorimetric (b) or ELISA (c) assays. (d) The expression of α-SMA was evaluated by western blot analysis of cell lysates at 72 h, using the loading control, β-tubulin, for signal normalization and quantification of the relative densitometric unit. The blots are representative of three independent experiments with cells from different donors. The data are expressed as the means and s.d. *P<0.05 vs. medium alone; **P<0.01 vs. medium alone by the analysis of variance followed by the Dunnett's post-hoc test ; n=5 in panels a, b and c; n=3 in panel d. HSPG2, heparan sulfate proteoglycan 2; VCAN, versican.
Asthmatic fibrocytes did not constitutively release detectable levels of IL-6, IL-11, and leukemia inhibitory factor (LIF, also known as adipogenesis inhibitory factor), but produced relatively large amounts of IL-11 and detectable amounts of IL-6 and LIF when stimulated with IL-4 or IL-13 (Table 3). Moreover, the conditioned medium from fibrocytes exposed to IL-4 and IL-13 contained augmented levels of the bioactive form of transforming growth factor (TGF)-β1 (Table 3) and the increase in the concentrations of bioactive TGF-β1 was particularly high in response to IL-13 stimulation (Table 3). Conversely, either IL-4 or IL-13 significantly decreased the production of the CXC chemokine ligand (CXCL) CXCL8 (old acronym: IL-8) by fibrocytes (Table 3). Either cytokines also reduced to undetectable levels the amounts of released CXCL1 (old acronym: GROα) and tumor necrosis factor-α (TNF-α), which these cells constitutively produced (Table 3).
IL-17A induces asthmatic fibrocytes to acquire a proinflammatory phenotype
IL-17A is one of the major cytokines produced by Th17 lymphocytes in asthmatic airways and may mainly contribute to the recruitment of neutrophils23, 24, 25 Stimulation of fibrocytes from asthmatic donors with IL-17A did not induce significant changes in the levels of expression of the mRNAs for COL3A1 and COL5A2 or in the levels of expression of the genes encoding the core proteins of VCAN and HSPG2 (Figure 4a). Moreover, fibrocytes stimulated with IL-17A showed only a modest increase in the production of HA and TNC (Figure 4b). The levels of total TGF-β1 were slightly higher in the conditioned media from IL-17A-stimulated fibrocytes, in comparison with the levels detected in cultures of non-stimulated cells, but the concentrations of the bioactive form of TGF-β1 were not significantly increased (Table 4).
Changes in the expression of extracellular matrix (ECM) molecules upon stimulation of asthmatic fibrocytes with interleukin (IL)-17A for up to 48 h. The expression of the mRNA for the indicated ECM molecules (a) was evaluated at 24 h by real-time reverse transcriptase-PCR. The level of expression of the mRNA encoding each target gene was normalized to the level of expression of the mRNA encoding the reference gene, ubiquitin C. The amounts of released hyaluronan (HA) and tenascin-C (TNC) (b) were measured in the supernatants at 48 h by ELISA. The data are expressed as the means and s.d. *P<0.05 vs. medium alone by the paired Student's t-test; n=5. HSPG2, heparan sulfate proteoglycan 2; VCAN, versican.
Although IL-17A induced the production of IL-6, IL-11, and LIF from asthmatic fibrocytes, its effects were different from those observed after stimulation with IL-4 or IL-13 (Table 3) because IL-17A exposure was associated with the production of large amounts of IL-6 and LIF and much lower amounts of IL-11 (Table 4). Differing from IL-4 and IL-13 (Table 3), IL-17A induced a marked increase in the release of CXCL1, CXCL8, and TNF-α from asthmatic fibrocytes (Table 4). Interestingly, fibrocytes cultured in the presence of IL-17A showed a substantial increase in the expression of α-SMA gene and protein, and this effect was almost completely inhibited by a neutralizing antibody against CXCL8 (Figure 5).
Changes in the expression of α-smooth muscle actin (α-SMA) gene and protein upon stimulation with interleukin (IL)-17A, and effects of CXC chemokine ligand 8 (CXCL8) neutralization. Asthmatic fibrocytes were cultured for 72 h in serum-free medium alone or in medium supplemented with IL-17A, in the presence or absence of a neutralizing mouse anti-human CXCL8 IgG1 or control IgG1. The expression of α-SMA mRNA (a) was evaluated by real-time reverse transcriptase-PCR. The levels of expression of the mRNA encoding the target gene under each experimental condition were normalized to the level of expression of the mRNA encoding the reference gene, ubiquitin C. The expression of the protein (b) was evaluated by western blot analysis of cell lysates using the loading control, β-tubulin, for signal normalization and quantification of the relative densitometric unit. The blots are representative of four independent experiments with cells from different donors. Data are expressed as the means and s.d. **P<0.01 for all comparisons by the analysis of variance followed by the Dunnett's or the Tuckey's post-hoc tests; n=4.
In contrast to IL-4 and IL-13, IL-17A significantly increases the proliferation of asthmatic fibrocytes
The ability of fibrocytes to proliferate in response to stimulation with IL-4, IL-13, or IL-17A was assessed by direct cell enumeration after an incubation period of 72 h. Neither IL-4 nor IL-13 exerted a significant mitogenic effect on asthmatic fibrocytes in comparison with medium alone (Figure 6a). By contrast, the mean cell number after stimulation with IL-17A for 72 h was about 2.1-fold higher than in control cultures incubated with medium alone for the same period of time (12.18 vs. 5.83 × 103 cells per well; Figure 6a). In some experiments, cell division was monitored by the PKH-26 dye dilution technique as previously reported.19 Analysis of the changes in fluorescence intensities of the cells labeled with PKH-26 by flow cytometry showed that most of the fibrocytes stimulated with IL-17A for 72 h had undergone at least one division (Figures 6b,c). Cell viability was ≥98.2% in all experiments.
Proliferative responses to stimulation with interleukin (IL)-4, IL-13, or IL-17A. Asthmatic fibrocytes were plated at the density of 5 × 103 cells per well in serum-free medium and incubated for 72 h with medium alone or with medium supplemented with IL-4, IL-13, or IL-17A before enumeration by hemocytometry (a). For analysis of cell division, fibrocytes were labeled with the fluorescent dye PKH-26 before stimulation. The fluorescence of PKH-26 is reduced by 50% during each cell division. A small aliquot of the labeled cells was fixed and analyzed by flow cytometry at time point 0, together with an aliquot of unlabeled cells as negative control. The remaining labeled cells were incubated for 72 h with medium alone or with medium supplemented with IL-4, IL-13, or IL-17A. Their fluorescence was then analyzed by flow cytometry to determine the percentage of cells that had undergone at least one division (proliferative fraction) on the basis of the extent of the shift to the left of PKH-26 fluorescence intensities at 72 h vs. time point 0. Representative histograms are shown in panel b and the results of the quantitative analysis in c are expressed as the mean proliferative fractions and s.d. **P<0.01 vs. medium alone by analysis of variance followed by the Dunnett's post-hoc test; n=7 in panel a; n=3 in panel c.
Discussion
A substantial proportion of the mature fibrocytes that populate the airways of individuals with severe or refractory asthma22 may be recruited from the circulation because appreciable numbers of CD34+COL1+ cells have been detected in the peripheral blood of patients with current symptoms and airflow obstruction.22, 29 This possibility is supported by the results of our previous study19 where intravenously injected mature fibrocytes were tracked to the inflamed airway wall in an animal model of chronic allergic asthma. In the same study,19 we observed the presence of increased numbers of fibrocytes in the bronchial mucosa of patients who were experiencing an allergen-induced asthmatic response. The majority of these cells were located near the area of COL1 deposition beneath the basement membrane and on the average accounted for 42.6% of the cells actively synthesizing COL1. Here, we confirm the presence of fibrocytes in the airways of patients with allergen-exacerbated asthma and provide evidence that a pure population of mature fibrocytes can be successfully isolated from the peripheral blood of these patients to evaluate their functional characteristics and interaction with other cells involved in the pathogenesis of asthma.
It is already known that bronchial epithelial cells from asthmatic patients are a major source of factors that can induce the migration and local differentiation of fibrocytes.15, 16 The bronchial smooth muscle also produces chemoattractants for fibrocytes.22 These observations may explain why fibrocytes predominantly accumulate in the subepithelial zone19, 21 of the asthmatic airways and can even infiltrate the underlying airway smooth muscle bundle in patients with severe disease.22 However, Th2 and Th17 cells are major components of the subepithelial inflammatory infiltrate in most forms of asthma,7, 23, 30, 31, 32 and increased levels of Th2 and Th17 cell-derived cytokines were also detected in the sputum of the asthmatic patients recruited in this study. Therefore, the main objective of this study was to evaluate how these cytokines may potentially affect the function of fibrocytes once they have migrated to the bronchial mucosa of asthmatic patients.
It is widely recognized that IL-4 and IL-13 participate in the pathogenesis of subepithelial fibrosis in asthma.32, 33, 34 We stimulated mature fibrocytes from asthmatic patients with IL-4 and IL-13 and evaluated their ability to affect the production of ECM components and the release of cytokines and growth factors. We found that either cytokines induced fibrocytes to acquire a predominant profibrotic phenotype, characterized by decreased release of the proinflammatory cytokines CXCL1, CXCL8, and TNF-α and increased expression of COL3, COL5, VCAN, HSPG2, HA, and TNC. Moreover, IL-13 promoted the release of large amounts of TGF-β1 and its spontaneous activation, which is one of the major mechanisms through which IL-13 may exert its profibrotic function in vivo.35 IL-4 also increased the contents of bioactive TGF-β1 in the conditioned medium of asthmatic fibrocytes, albeit to a lesser extent.
The ECM molecules produced by fibrocytes in response to IL-4 and IL-13 are those most frequently detected in the thickened lamina reticularis of the bronchial mucosa of asthmatic patients.9, 11, 12, 13, 14 Considering that subepithelial fibrosis is particularly prominent in forms of asthma characterized by Th2 cell-driven chronic inflammation,6, 7, 8 our findings support our hypothesis that fibrocytes may contribute to the development of this structural alteration.15, 16 IL-4 and IL-13 may not be able to expand the population of mature fibrocytes at tissue site because we did not observe any significant mitogenic effect of these cytokines in vitro. However, previous observations indicate that IL-4 and IL-13 may also promote the local differentiation of mature fibrocytes from their monocyte-like precursor cells,36 which are likely present in the inflamed airways of asthmatic individuals.15, 16
IL-17A is one of the major cytokines produced by Th17 lymphocytes in asthma.37, 38 This cytokine has been implicated in the development of corticosteroid-resistant neutrophilic airway inflammation in difficult-to-treat asthma and in asthma predominantly exacerbated by viral infections.23, 24, 25, 37, 38 However, allergic and non-allergic patients with mild or moderate asthma also show increased expression of IL-17A in their airways38, 39 and eosinophils may represent an important source of IL-17A in addition to Th17 cells.39 We observed appreciable concentrations of IL-17A in the induced sputum of the patients recruited in this study, together with elevated numbers of lymphocytes and eosinophils and a significant increase in the absolute number of neutrophils. Stimulation of asthmatic fibrocytes with IL-17A resulted in the release of large amounts of CXCL1, CXCL8, and TNF-α. CXCL1 and CXCL8 are considered to contribute to the recruitment of neutrophils in asthmatic airways.40 Inhaled TNF-α causes airway neutrophilia and airway hyperresponsiveness even in normal individuals and excessive release of this cytokine in the asthmatic airways may also amplify ongoing mucosal inflammation through the activation of nuclear factor-κB.40 Upon stimulation of fibrocytes with IL-17A, the marked increase in the release of TNF-α, CXCL1, and CXCL8 was associated with little changes in the production of collagenous and non-collagenous ECM molecules. Similar results were previously reported for primary cultures of asthmatic bronchial fibroblasts stimulated with this cytokine.39 Although IL-17A induced a slight increase in the release of TGF-β1 from asthmatic fibrocytes, the concentration of the TGF-β1 form with profibrotic activity did not significantly change in cultures of fibrocytes stimulated with IL-17A. Another profibrotic factor that can promote subepithelial fibrosis in vivo, such as IL-11,41 was released in larger amounts from fibrocytes stimulated with IL-4 and IL-13 than from cells stimulated with IL-17A.
In contrast to IL-4 and IL-13, IL-17A significantly increased the proliferation of asthmatic fibrocytes, suggesting that IL-17A may promote the accumulation of mature fibrocytes in the bronchial mucosa of asthmatic individuals through this mechanism. The increased proliferation of fibrocytes resulting from stimulation with IL-17A was associated with the release of large amounts of IL-6 and LIF. Although stimulation of fibrocytes with IL-4 or IL-13 also resulted in the release of IL-6 and LIF from fibrocytes, the amounts of released cytokines were much lower than those detected in cultures of IL-17A-stimulated cells. These cytokines may have contributed to the mitogenic effect of IL-17A because they are known to influence the proliferation of certain subsets of fibroblasts and immature myofibroblasts through direct and indirect mechanisms.41, 42, 43 However, this possibility was not specifically addressed in this study. IL-6 and LIF also exhibit other biological activities that may be relevant to the pathogenesis of asthma.41, 42 IL-6 is known to influence neutrophil recruitment through multiple mechanisms and to exert a key role in the transition from acute to chronic inflammation.41 It also favors an imbalance between Th2 cells and regulatory T cells at tissue sites similar to that observed in asthma.41 LIF may increase bronchial smooth muscle contractility in asthmatic individuals, thereby contributing to airway hyperresponsiveness.41, 44 Thus, interaction of fibrocytes and IL-17A-producing cells in asthmatic airways may also set in motion a chain of events potentially involved in the persistence of structural changes and airway hyperresponsiveness after resolution of an acute inflammatory exacerbation of the disease, particularly in a microenvironment dominated by Th2 lymphocytes.
An additional important result of this study is that CXCL8 may promote an increase in the expression of α-SMA in mature fibrocytes. Interestingly, it has been recently reported that hepatocytes expressing the hepatitis C virus core protein can induce α-SMA expression, without increasing COL1 production, in hepatic stellate cells through the release of CXCL8.45 In asthmatic airways, this chemokine is produced in large amounts by bronchial epithelial cells,46, 47 which represent a major source of other factors involved in the differentiation of fibrocytes into myofibroblast-like cells, particularly TGF-β and endothelin-1.15, 19, 47 Therefore, our novel observation also adds support to the hypothesis that the asthmatic bronchial epithelium has a key role in modulating the phenotype of fibrocytes at the tissue site.15, 16
In conclusion, our study provides the first evidence that IL-4, IL-13, and IL-17A differentially affect the profibrotic and proinflammatory function of fibrocytes isolated from the peripheral blood of patients with allergen-exacerbated asthma. These cells express an asthma-specific profibrotic phenotype when stimulated with IL-4 and IL-13, while they proliferate and acquire a proinflammatory phenotype, which may facilitates neutrophil recruitment and airway hyperresponsiveness, on stimulation with IL-17A. Our findings suggest that fibrocytes may be involved in the development of subepithelial fibrosis and contribute to determine the pattern of infiltrating leukocytes to various extents in asthma, depending on the relative concentrations of Th2 cell-derived cytokines and IL-17A present in the microenvironment.
Methods
Fibrocyte donors. Fibrocyte donors were 12 asthmatic patients and 8 healthy individuals who provided informed consent after protocol approval by the appropriate review board. At the time of peripheral blood cell sampling, asthmatic patients were suffering from a moderate exacerbation of their disease, according to the following standard criteria: worsening of symptoms requiring more frequent use of short acting bronchodilators during the day, or disturbing sleep, or causing early morning wakening, for at least 48–72 h; prebronchodilator forced expiratory volume in one second <80% of the personal best; differential eosinophils count in induced sputum >3%.26, 48 Scoring of symptoms and spirometry were performed as previously described.48 The healthy volunteers had no history of respiratory and/or allergic diseases and their lung function was normal. None of the fibrocyte donors was a current smoker or had a past smoking history of ≥10 pack-years. None of them had suffered from respiratory or systemic infections in the previous 2 months.
Sputum induction and sputum processing were performed according to standard procedures.48, 49 The contents of IL-4, IL-13, and IL-17A in the sputum fluid phase were measured with Abcam ELISA kits (Abcam, Cambridge, UK). Fibrocytes coexpressing CD34 and COL1A1 mRNA were enumerated in cytospins of sputum cells by adopting the procedure described below for enumeration of fibrocytes in cytospins of total circulating leukocytes.
Enumeration of circulating fibrocytes. Blood was processed to obtain the leukocyte-rich fraction as previously described.50 Briefly, the blood was centrifuged at 500 g and the buffy coat was removed. Residual erythrocytes were lysed with Buffer EL (Quiagen, Düsseldorf, Germany) for 15 min at 4 °C and total leukocytes were collected by centrifugation. The leukocytes were washed in Tris-buffered saline, counted and cytocentrifuged onto Superfrost Plus slides (BDH Chemicals, Poole, UK). Before staining, the cells were fixed in HOPE solution (Polysciences, Warrington, PA) and blocked with 20% human serum in Tris-buffered saline for 15 min at room temperature. The slides were then incubated with a fluorescein isothiocyanate-conjugated mouse anti-human CD34 antibody (clone 8G12, BD Biosciences, San Jose, CA). For intracellular staining of COL1, cells were permeabilized with 0.3% saponin solution (Sigma-Aldrich, St Louis, MO) in Tris-buffered saline and sequentially incubated with a rabbit anti-human COL1 (α2 chain) (Novus Biologicals, Littletone, CO), a biotinylated goat anti-rabbit secondary antibody (Novus Biologicals), and rhodamine red X-conjugated streptavidin (Jackson ImmunoResearch, Bar Harbor, ME). Cell nuclei were stained blue using 4′,6-diamidino-2-phenylindole (Vector Laboratories, Burlingame, CA). Isotype-matched mouse antibodies and normal goat IgG were used as negative controls. Cells coexpressing CD34 and COL1A1 mRNA were identified by double immunohistochemistry and in-situ hybridization, using a digoxigenin-labeled antisense probe for detection of COL1A1 mRNA, as previously described.19
At least 800 nucleated cells were examined on duplicate slides, using an Eclipse 80i microscope (Nikon, Tokyo, Japan) for immunofluorescence staining and an Olympus BH-2 microscope (Olympus, New York, NY) for immunohystochemical staining and in-situ hybridization. The percentage of positively stained cells was calculated after subtraction of the percentage of non-specifically stained cells detected in control slides. This value was multiplied by the total number of leukocytes per ml of blood to estimate the absolute number of cells per ml of blood.
Purification of circulating fibrocytes from asthmatic patients. CD34+ cells were isolated from the whole blood by immunomagnetic selection using the mouse class II anti-CD34 antibody and reagents of a commercially available cell separation kit (Catalog no. 18086, Stemcell Technologies, Vancouver, BC) according to the manufacturer's protocol. The purity of the resulting cell population was assessed by flow cytometry, using a phycoerythrin-conjugated mouse class III anti-human CD34 (clone 8G12, BD Biosciences). The isolated cells were also double stained for CD45 and COL1. Following incubation with a phycoerythrin-conjugated mouse anti-human CD45 IgG1 (clone HI30, BD Biosciences), cells were fixed and permeabilized with Cytofix/Cytoperm kit (BD Biosciences/Pharmingen, San Jose, CA) and sequentially incubated with a rat antibody against the N-terminus of human intracellular COL1 precursor protein (clone M58; Chemicon International, Temecula, CA) and a fluorescein isothiocyanate-conjugated goat anti-rat secondary antibody (Invitrogen, Carlsbad, CA). Analysis of the cells stained with these antibodies or the appropriate controls was performed using a FACSCalibur flow cytometry system (BD Biosciences) and the associated CellQuest software, as previously described.18
The cells isolated by immunomagnetic selection were treated with chymopapain (Sigma-Aldrich), 1.5 U per 105 cells, for 10 min at 37 °C to break the antigen–antibody bonds. After removal of the magnetic particles, the cells were seeded into the uncoated wells of 96-well culture plates (5 × 104 cells per well) and incubated for 48 h in RPMI 1640 medium, supplemented with 10 mM N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid (HEPES), 1% nonessential amino acids, 1% sodium pyruvate, 2 mM L-glutamine, 100 U per ml penicillin, 0.1 mg ml−1 streptomycin (all from Sigma-Aldrich), and 20% autologous serum, at 37 °C in a humidified atmosphere (5% CO2 in air). The non-adherent cells were then gently aspirated and the adherent cells were reincubated for 3 days in fresh medium. Cell morphology was monitored under an inverted microscope and the purity of mature fibrocytes was checked at the end of the incubation period by flow cytometry, as reported above. The expression of IL-17RA, types I and II IL-4 receptor subunits and IL-R1α2 was also examined by flow cytometry, using specific antibodies from R&D Systems (Minneapolis, MN).
Stimulation of fibrocytes in culture. In most experiments, the purified mature fibrocytes were seeded into the flat-bottom wells of 96-well plates at the density of 2 × 104 cells per well and stimulated with recombinant human IL-4 (5 ng ml−1; molecular mass 14.9 kDa), IL-13 (5 ng ml−1; molecular mass 12.5 kDa), or IL-17A (15 ng ml−1; molecular mass 31 kDa; Peprotech, Rocky Hill, NJ) for up to 72 h in serum-free RPMI 1640 medium (200 μl per well), containing 500 μg ml−1 bovine serum albumin, 10 mM HEPES, 1% nonessential amino acids, 1% sodium pyruvate, 2 mM L-glutamine, 100 U per ml penicillin, and 0.1 mg ml−1 streptomycin. When cells were tested for the release of collagenous proteins, the culture medium was also supplemented with 50 μg ml−1 ascorbic acid (Sigma-Aldrich) as previously described.18 To evaluate the mitogenic effects of IL-4, IL-13, or IL-17A, fibrocytes were plated at the density of 5 × 103 cells per well and stimulated for 72 h before enumeration by hemocytometry. The proliferative rate was estimated by monitoring the changes in fluorescence intensity of cells stained with PKH-26, as previously reported.19 In all these experiments, cells incubated in the absence of the tested cytokines were used as controls. For blocking experiments, fibrocytes were preincubated for 1 h with medium containing 10 μg ml−1 of a neutralizing mouse anti-human CXCL8 IgG1 (MAB208, R&D Systems) or control IgG1. Cells were then stimulated with IL-17A in the continuing presence of anti-CXCL8 or control IgG1.
Gene expression analysis. This analysis was performed by real-time reverse transcriptase-PCR. A two-step reverse transcriptase-PCR was conducted using a SYBR green-based cells-to-CT kit (Applied Biosystems/Ambion, Austin, TX) for small cell samples according to the manufacturer's instruction and using cell lysates from identical numbers of cultured fibrocytes in each experiment. Specific primers for the target genes COL3A1, COL5A2, VCAN, and HSPG2 were those used in a previous study.18 The primer pairs for the TNC gene and the reference gene, ubiquitin C, were retrieved from the literature,47 and those for the α-SMA gene from the PrimerBank (ID: 213688378b1). The level of expression of the mRNA encoding each target gene was normalized to the level of expression of the mRNA encoding the reference gene.
Quantitation of released products. HA release was quantified by using a commercially available assay kit (Hyaluronan DuoSet, R&D Systems) as previously reported.18 The production of fibrillar collagens was measured with the QuickZyme Collagen Assay kit (Quickzyme, Leiden, The Netherlands). Analysis of TNC release was performed by a sandwich ELISA, using a previously described procedure.19 The capture antibody was a mouse anti-human TNC IgG1 (clone T2H5, AbD Serotec/Morphosys, Oxford, UK); the detection antibody was a biotinylated goat anti-human TNC IgG (BAF3358, R&D Systems) and the standard was a native human TNC (AbD Serotec/Morphosys). The levels of released CXCL1, CXCL8, TNF-α, IL-6, IL-11, and LIF were quantified with ELISA kits from R&D Systems. The concentrations of total and spontaneously activated TGF-β1 in the conditioned media were also measured by ELISA (R&D Systems), using acid-treated and -untreated samples according to the manufacturer's instructions.
Western blot analysis of α-SMA expression. Western blot analysis of cellular lysates was performed as previously described.18 The mouse monoclonal antibodies against human α-SMA and the loading control, β-tubulin, were from Sigma-Aldrich and Abcam, respectively. Densitometric analysis was performed with the ImageQuanta TL (Amersham Biosciences, Piscataway, NJ), using the loading control for normalization of the signal.
Statistical analysis. Statistical analysis was performed with GraphPad Prism version 5 for Windows (GraphPad Software, San Diego, CA).
References
National Asthma Education and Prevention Program. Expert panel report 3 (EPR-3): guidelines for the diagnosis and management of asthma – summary report 2007. J. Allergy Clin. Immunol. 120 (5 Suppl), S94–S138 (2007).
Jeffery, P.K. Remodeling in asthma and chronic obstructive lung disease. Am. J. Respir. Crit. Care Med. 164, S28–S38 (2001).
Benayoun, L., Druilhe, A., Dombret, M.C., Aubier, M. & Pretolani, M. Airway structural alterations selectively associated with severe asthma. Am. J. Respir. Crit Care Med. 167, 1360–1368 (2003).
Cohn, L., Elias, J.A. & Chupp, G.L. Asthma: mechanisms of disease persistence and progression. Annu. Rev. Immunol. 22, 789–815 (2004).
Saglani, S. et al. Ultrastructure of the reticular basement membrane in asthmatic adults, children and infants. Eur. Respir. J. 28, 505–512 (2006).
Wenzel, S.E. et al. Evidence that severe asthma can be divided pathologically into two inflammatory subtypes with distinct physiologic and clinical characteristics. Am. J. Respir. Crit. Care Med. 160, 1001–1008 (1999).
Woodruff, P.G. et al. T-helper type-2-driven inflammation defines major subphenotypes of asthma. Am. J. Respir. Crit. Care Med. 180, 388–395 (2009).
Berry, M. et al. Pathological features and inhaled corticosteroid response of eosinophilic and non-eosinophilic asthma. Thorax 62, 1043–1049 (2007).
Roche, W.R., Beasley, R., Williams, J.H. & Holgate, S.T. Subepithelial fibrosis in the bronchi of asthmatics. Lancet 1, 520–524 (1989).
Boulet, L.-P. et al. Bronchial subepithelial fibrosis correlates with airway responsiveness to methacholine. Chest 112, 45–52 (1997).
Roberts, C.R. Is asthma a fibrotic disease? Chest 107, 111S–117S (1995).
Wilson, J.W. & Li, X. The measurement of reticular basement membrane and submucosal collagen in the asthmatic airway. Clin. Exp. Cytol. 65, 109–126 (2002).
Pini, L. et al. Differences in proteoglycans deposition in the airways of moderate and severe asthmatics. Eur. Respir. J. 29, 71–77 (2007).
Brewster, C.E.P., Howarth, P.H., Djukanovic, R., Wilson, J., Holgate, S.T. & Roche, W.R. Myofibroblasts and subepithelial fibrosis in bronchial asthma. Am. J. Respir. Cell Mol. Biol. 3, 507–511 (1990).
Mattoli, S., Bellini, A. & Schmidt, M. The role of a human hematopoietic mesenchymal progenitor in wound healing and fibrotic diseases and implications for therapy. Curr. Stem Cell Res. Ther. 4, 266–280 (2009).
Bellini, A. & Mattoli, S. The role of the fibrocyte, a bone marrow-derived mesenchymal progenitor, in reactive and reparative fibroses. Lab. Invest. 87, 858–870 (2007).
Reilkoff, R.A., Bucala, R. & Herzog, E.L. Fibrocytes: emerging effector cells in chronic inflammation. Nat. Rev. Immunol. 11, 427–435 (2011).
Bianchetti, L., Barczyk, M., Cardoso, J., Schmidt, M., Bellini, A. & Mattoli, S. Extracellular matrix remodelling properties of fibrocytes. J. Cell. Mol. Med (2011)DOI: 10.1111/j.1582-4934.2011.01344.x.
Schmidt, M., Sun, G., Stacey, M.A., Mori, L. & Mattoli, S. Identification of circulating fibrocytes as precursors of bronchial myofibroblasts in asthma. J. Immunol. 171, 380–389 (2003).
Kariyawasam, H.H., Aizen, M., Barkans, J., Robinson, D.S. & Kay, A.B. Remodeling and airway hyperesponsiveness but not cellular inflammation persist after allergen challenge in asthma. Am. J. Respir. Crit. Care Med. 175, 896–904 (2007).
Nihlberg, K., Larsen, K., Hultgårdh-Nilsson, A., Malmström, A., Bjermer, L. & Westergren-Thorsson, G. Tissue fibrocytes in patients with mild asthma: a possible link to thickness of reticular basement membrane? Respir. Res. 7, 50 (2006).
Saunders, R. et al. Fibrocyte localization to the airway smooth muscle is a feature of asthma. J. Allergy Clin. Immunol. 123, 376–384 (2009).
Alcorn, J.F., Crowe, C.R. & Kolls, J.K. TH17 cells in asthma and COPD. Annu. Rev. Physiol. 72, 495–516 (2010).
Wiehler, S. & Proud, D. Interleukin-17A modulates human airway epithelial responses to human rhinovirus infection. Am. J. Physiol. Lung Cell. Mol. Physiol. 293, L505–515 (2007).
Shin, T.-S. et al. Role of inducible nitric oxide synthase on the development of virus-associated asthma exacerbation which is dependent on Th1 and Th17 cell responses. Exp. Mol. Med. 42, 721–730 (2010).
Reddel, H.K. et al. An official american thoracic society/european respiratory society statement: asthma control and exacerbations. Am. J. Respir. Crit. Care Med. 180, 59–99 (2009).
Khurana Hershey, G.K. IL-13 receptors and signalling pathways: an evolving web. J. Allergy Clin. Immunol. 111, 677–690 (2003).
Elias, J.A. & Lee, C.G. IL-13 in asthma. The successful integration of lessons from mice and humans. Am. J. Respir. Crit. Care Med. 183, 957–958 (2011).
Wang, C.-H. et al. Increased circulating fibrocytes in asthma with chronic airflow obstruction. Am. J. Respir. Crit. Care Med. 178, 583–591 (2008).
Heijink, I.H. & van Oosterhout, A.J. Targeting T cells for asthma. Curr. Opin. Pharmacol. 5, 227–231 (2005).
Kuipers, H. & Lambrecht, B.N. The interplay of dendritic cells, Th2 cells and regulatory T cells in asthma. Curr. Opin. Immunol. 16, 702–708 (2004).
Kay, A.B. The role of T lymphocytes in asthma. Chem. Immunol. Allergy 91, 59–75 (2006).
Komai, M. et al. Role of Th2 responses in the development of allergen-induced airway remodelling in a murine model of allergic asthma. Br. J. Pharmacol. 138, 912–920 (2003).
Wills-Karp, M. Interleukin-13 in asthma pathogenesis. Immunol. Rev. 202, 175–190 (2004).
Lee, C.G. et al. Interleukin-13 induces tissue fibrosis by selectively stimulating and activating transforming growth factor beta(1). J. Exp. Med. 194, 809–821 (2001).
Shao, D.D., Suresh, R., Vakil, V., Gomer, R.H. & Pilling, D. Pivotal advance: Th-1 cytokines inhibit, and Th-2 cytokines promote fibrocyte differentiation. J. Leukoc. Biol. 83, 1323–1333 (2008).
Zhao, Y., Yang, J., Gao, Y. & Guo, W. Th17 immunity in patients with allergic asthma. Int. Arch. Allergy Immunol. 151, 297–307 (2010).
Bullens, D.M. et al. IL-17 mRNA in sputum of asthmatic patients: linking T cell driven inflammation and granulocyte influx? Respir. Res. 7, 135 (2006).
Molet, S. et al. IL-17 is increased in asthmatic airways and induces human bronchial fibroblasts to produce cytokines. J. Allergy Clin. Immunol. 108, 430–438 (2001).
Barnes, P.J. The cytokine network in asthma and chronic obstructive pulmonary disease. J. Clin. Invest. 118, 3546–3556 (2008).
Silver, J.S. & Hunter, C.A. gp130 at the nexus of inflammation, autoimmunity, and cancer. J. Leukoc. Biol. 88, 1145–1156 (2010).
Knight, D. Leukaemia inhibitory factor (LIF): a cytokine of emerging importance in chronic airway inflammation. Pulm. Pharmacol. Ther. 14, 169–176 (2001).
Moodley, Y.P. et al. Fibroblasts isolated from normal lungs and those with idiopatic pulmonary fibrosis differ in interleukin-6/gp130-mediated cell signaling and proliferation. Am. J. Pathol. 163, 345–354 (2003).
Fayon, M. et al. Increased secretion of leukemia inhibitory factor by immature airway smooth muscle cells enhances intracellular signaling and airway contractility. Am. J. Physiol. Lung. Cell. Mol. Physiol. 291, L244–L251 (2006).
Clément, S., Pascarella, S., Conzelmann, S., Gonelle-Gispert, C., Guillox, K. & Negro, F. The hepatitis C virus core protein indirectly induces alpha-smooth muscle actin expression in hepatic stellate cells via interleukin-8. J. Hepatol. 52, 635–643 (2010).
Marini, M., Vittori, E., Hollemborg, J. & Mattoli, S. Expression of the potent inflammatory cytokines, granulocyte-macrophage - colony-stimulating factor and interleukin-6 and interleukin-8, in bronchial epithelial cells of patients with asthma. J Allergy Clin. Immunol. 89, 1001–1009 (1992).
Pégorier, S., Arouche, N., Dombret, M.-C., Aubier, M. & Pretolani, M. Augmented epithelial endothelin-1 expression in refractory asthma. J. Allergy Clin. Immunol. 120, 1301–1307 (2007).
Jayaram, L. et al. Determining asthma treatment by monitoring sputum cell counts: effect on exacerbations. Eur. Respir. J. 27, 483–494 (2006).
Hastie, A.T. et al. Analysis of asthma severity phenotypes and inflammatory proteins in subjects stratified by sputum granulocytes. J. Allergy. Clin. Immunol. 125, 1028–1036 (2010).
Feezor, R.J. et al. Whole blood and leukocyte RNA isolation for gene expression analyses. Physiol. Genomics 19, 247–254 (2004).
Acknowledgements
This study was support by the international FibroGENE and CellNet projects.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
SM is a founding shareholder and board member of AVAIL GmbH. The other authors have no potential conflicts of interest.
Rights and permissions
About this article
Cite this article
Bellini, A., Marini, M., Bianchetti, L. et al. Interleukin (IL)-4, IL-13, and IL-17A differentially affect the profibrotic and proinflammatory functions of fibrocytes from asthmatic patients. Mucosal Immunol 5, 140–149 (2012). https://doi.org/10.1038/mi.2011.60
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mi.2011.60
This article is cited by
-
IL-25 induces airway remodeling in asthma by orchestrating the phenotypic changes of epithelial cell and fibrocyte
Respiratory Research (2023)
-
Skin applications of cannabidiol: sources, effects, delivery systems, marketed formulations and safety
Phytochemistry Reviews (2023)
-
Effects of the serine protease inhibitor rBmTI-A in an experimental mouse model of chronic allergic pulmonary inflammation
Scientific Reports (2019)
-
Acupuncture Regulates the Balance of CD4+ T Cell Subtypes in Experimental Asthma Mice
Chinese Journal of Integrative Medicine (2019)
-
ILC2s in infectious diseases and organ-specific fibrosis
Seminars in Immunopathology (2018)