Abstract
Sleep is regulated by two mechanisms: the homeostatic process and the circadian clock. Light affects sleep and alertness by entraining the circadian clock, and acutely inducing sleep/alertness, in a manner mediated by intrinsically photosensitive retinal ganglion cells. Because intrinsically photosensitive retinal ganglion cells are believed to be minimally sensitive to red light, which is widely used for illumination to reduce the photic disturbance to nocturnal animals during the dark phase. However, the appropriate intensity of the red light is unknown. In the present study, we recorded electroencephalograms and electromyograms of freely moving mice to investigate the effects of red light emitted by light-emitting diodes at different intensities and for different durations on the sleep–wake behavior of mice. White light was used as a control. Unexpectedly, red light exerted potent sleep-inducing effects and changed the sleep architecture in terms of the duration and number of sleep episodes, the stage transition, and the EEG power density when the intensity was >20 lx. Subsequently, we lowered the light intensity and demonstrated that red light at or below 10 lx did not affect sleep–wake behavior. White light markedly induced sleep and disrupted sleep architecture even at an intensity as low as 10 lx. Our findings highlight the importance of limiting the intensity of red light (⩽10 lx) to avoid optical influence in nocturnal behavioral experiments, particularly in the field of sleep and circadian research.
Similar content being viewed by others
Introduction
Homeostatic processes and circadian rhythms have been reported to be key mechanisms in the sleep–wake regulation of mammals1, 2. Prolonged wakefulness may enhance the homeostatic sleep tendency, whereas the circadian rhythm controls the timing of sleep across the light and dark cycle3.
Light is the most potent cue in entraining the circadian rhythm4. In nocturnal animals, the effects of light have been studied intensively in the context of circadian biology5. Therefore, light can be used for image-forming vision, which is crucial for sighted animals to detect and distinguish surrounding objects, as well as for non-image-forming processes such as the entrainment of the circadian rhythm6, the alertness in diurnal animals and humans7 and the acute induction of sleep in nocturnal animals8, 9, 10.
Melanopsin signaling is thought to be critical in the influence of light on sleep; intrinsically photosensitive retinal ganglion cells (ipRGCs) convey signals to the brain11, 12, 13. The ipRGCs expressing melanopsin are maximally sensitive to blue light and minimally sensitive to red light14, 15, 16. Owing to the wavelength selectivity of melanopsin within the retina, blue wavelength light easily activates the ipRGCs, and the signals are transduced to the brain, whereas red wavelength light is relatively difficult to process. Therefore, nocturnal behavior experiments are usually performed in environments with red light to avoid the effects of light.
When experiments are carried out during the dark phase, red illumination is widely used for drug administration or other manipulations. Because there are no accurate rules or recommendations indicating the most appropriate intensity of red light for use in experiments, different laboratories often generate data with large variations, which might be attributed to the different intensities of red light that were applied. Thus, it is imperative to explore the appropriate intensity of red light that has minimal effects on sleep or alertness but offers sufficient illumination for experimenters to see their surroundings during the dark phase.
Light-emitting diode (LED) lights are commonly used and have the highest efficacy among all current optical sources17; therefore, we used LEDs as a light source. Here, we found that red light altered the amount and the architecture of sleep and wakefulness in mice at intensities of 20 lx or higher during the dark period. When the intensity of red light was lowered to 10 lx, there was no effect on the amount and architecture of non-rapid eye movement (non-REM, NREM) sleep and REM sleep in the dark phase.
Materials and methods
Animals
Male SPF C57BL/6J mice at an age of 8–10 weeks were obtained from the Laboratory Animal Center, Chinese Academy of Sciences (Shanghai, China). The mice were housed in a soundproofed and insulated recording room at an ambient temperature of 22±0.5 °C, with a relative humidity of 60±2%. A 12-h light/dark cycle (white lights on at 07:00, illumination intensity≈100 lx, before photic experiments) was automatically controlled. Food and water were available ad libitum. Experimental protocols were approved by the Medical Experimental Animal Administrative Committee of Shanghai, in accordance with the Guidelines of the NIH in the United States regarding the care and use of animals for experimental procedures. Every effort was made to minimize the number of animals for experiments and any pain or discomfort experienced by the animals.
Protocol
Experiment 1
To assess the effects of short exposures of white and red light on sleep in the dark phase, mice from each group were exposed to a 1 h/1 h light/dark (L/D) cycle from ZT12–22 in an environment of white and red light with the same intensity of 100 lx (n=9), 30 lx (n=5–7), 20 lx (n=5, 6) or 10 lx (n=6, 7). Then, mice were exposed to a 12 h/12 h L (100 lx)/D cycle; the subsequent 24 h were recorded. Moreover, darkness exposure in the entire dark phase was used as a control.
Experiment 2
To examine whether long exposures to white and red light at 10 lx would affect sleep and wakefulness, mice (n=5–8) were exposed to 100 lx white light for 12 h, followed by white light, red light at an intensity of 10 lx or darkness for 12 h.
Surgery, sleep data acquisition and analysis
Mice were implanted with electrodes for EEG and electromyogram (EMG) recordings under chloral hydrate anesthesia at 360 mg kg−1 (i.p.). The implant18, consisting of two stainless steel screws (1 mm in diameter) as EEG electrodes, was inserted through the skull (+1.0 mm anteroposterior; −1.5 mm mediolateral from bregma or lambda) according to the mouse brain atlas19. Two insulated stainless steel wires, used as the EMG electrodes, were bilaterally placed into both trapezius muscles. The electrodes were attached to a mini-connector and fixed to the skull with dental cement.
After a 10-day recovery period, the mice were individually housed in transparent barrels and habituated to the recording cable for 3–4 days before recording. The simultaneous recordings for EEG and EMG were carried out with a slip ring that let the mice move freely. The recording started at 07:00, that is, the beginning of the light period.
As previously described18, 20, 21, 22, cortical signals were amplified and filtered (0.5–30 Hz for EEG, 20–200 Hz for EMG), then digitized at a sampling rate of 128 Hz and recorded with SleepSign software (Kissei Comtec, Nagano, Japan). The sleep–wake states were automatically classified in 4-s periods as wakefulness, NREM sleep or REM sleep with the SleepSign software, on the basis of published standard criteria18, 20, 21, 22, 23, 24. The automatically defined wake or sleep stages were then checked visually and corrected if necessary.
EEG power spectra were calculated using fast Fourier transform for the frequency range 0–25 Hz, with a resolution of 0.25 Hz22. The power of each 0.25-Hz bin was first averaged across the sleep stages individually and then normalized as a group by calculation of the percentage of each bin from the total power (0–24.75 Hz) of the individual animal25.
Lighting design
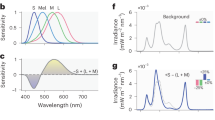
Separate white and red, dimmable LED lamps (LET-50068 3 W, QianHan Lighting, Shanghai, China) placed above each individual animal cage were used for all light experiments. The spectra of the white light (Figure 1a) and red light (Figure 1b) were detected with a spectroradiometer (CS2000, Konica-Minolta, Tokyo, Japan). The spectra of the LED lamps met standards similar to those reported in a previous study26. Silicon controlled dimming (model 280, DELIXI, Changsha, Hunan, China) was used to control the light intensities of the LED lamps. The intensity of the light was determined at the bottom center of each barrel by using a lightmeter (HT-1300, HCJYET, Guangzhou, Guangdong, China).
Effects of 100, 30 and 20 lx white light and red light pulses on NREM sleep and REM sleep during the dark phase. (a, b) The normalized electroluminescence spectra (a.u., arbitrary unit) of white (a) and red (b) LED sources. (c) Time course changes in NREM sleep and REM sleep in mice, which are exposed to 100 lx white light in the day and a 1 h/1 h L (100 lx white or red light)/D cycle or constant darkness at night. Each cycle represents the hourly mean±SEM of NREM sleep and REM sleep. Black, white and red circles indicate the profiles of continuous darkness, white light and red light treatments, respectively. The filled and open bars on the x axes indicate light-off and light-on treatments, respectively, at night. *P<0.05, **P<0.01 indicate significant differences between white light and continuous darkness. #P<0.05, ##P<0.01 indicate significant differences between red light and continuous darkness. Data were assessed via two-way ANOVA followed by a Bonferroni test. (d) Total time spent in NREM sleep and REM sleep of the groups exposed to 100, 30 and 20 lx white or red light, as well as continuous darkness, during 5-h light-on phase. Black, white and red bars show the profiles of continuous darkness, white or red light treatments, respectively. Values are means±SEM (continuous darkness n=7; 100 lx, n=9; 30 lx, n=5-7; 20 lx, n=5, 6). *P<0.05, **P<0.01 compared with continuous darkness, assessed via one-way ANOVA followed by a Bonferroni test.
Statistical analysis
All data are expressed as means±SEM. Comparisons of sleep amounts, as well as number of sleep–wake events, duration and transition of sleep–wake events among the groups exposed to white light, red light and darkness, were performed using one-way analysis of variance (ANOVA) followed by a Bonferroni post hoc test. Comparisons of time course changes in the amounts of sleep among the groups exposed to white light, red light and darkness were assessed using two-way ANOVA followed by a Bonferroni post hoc test. Comparisons of the EEG power density between groups exposed to white or red light and darkness were evaluated using the non-paired, two-tailed Student’s t-test. Comparisons of time course changes in the amounts of sleep between the day following 1 h/1 h L/D white or red light exposure and the baseline were assessed with two-way ANOVA followed by a Bonferroni post hoc test. Prism 5.01 (GraphPad Software, San Diego, CA, USA) was used for all statistical analyses. In all cases, P<0.05 was considered to be significant.
Results and discussion
Acute white and red light at an intensity of 100, 30 or 20 lx induced marked NREM sleep and REM sleep in the dark phase
To assess the effects of acute white and red light at an intensity of 100, 30 or 20 lx on sleep in the dark phase, we examined the sleep–wake profiles of mice exposed to 1 h/1 h L/D cycles from ZT12–22. Darkness exposure during the entire dark phase served as a control. Unexpectedly, acute red light at 100 lx induced marked NREM sleep and REM sleep oscillations, as white light did (Figure 1c). The total time spent in NREM sleep and REM sleep for the 5-h light-on period in each group was calculated. Both white and red light at an intensity of 100, 30 or 20 lx increased NREM sleep and REM sleep significantly, as compared with darkness (Figure 1d). Thus, similarly to white light, acute red light at an intensity of 20 lx or higher induced potent sleep in the dark phase.
Acute white and red light at an intensity of 100, 30, or 20 lx disrupted sleep–wake architecture and EEG power density during the dark phase
To better understand the sleep–wake profile resulting from exposure to acute white and red light at an intensity of 100, 30, or 20 lx, we determined the number and mean duration of episodes, as well as the stage transition number among NREM sleep, REM sleep and wakefulness. Both acute white light and red light at 100 lx, compared with darkness, increased the episode number of REM sleep and the stage transitions from NREM sleep to REM sleep and decreased the mean duration of wakefulness, while acute exposure of red light but not white light increased the stage transitions from REM sleep to wakefulness (Figure 2a and 2d). Acute white and red light at 30 lx decreased the mean duration of wakefulness, whereas red light increased the stage transitions from wakefulness to NREM sleep (Figure 2b and 2e). Both acute white light and red light at 20 lx increased the episode number of REM sleep and the stage transitions (from NREM sleep to REM sleep, from REM sleep to wakefulness), and decreased the mean duration of wakefulness without affecting other sleep–wake parameters (Figure 2c and 2f). However, there was no difference between the white light and red light groups regarding sleep architecture. Moreover, acute 1 h/1 h L/D exposure of white but not red light at an intensity of 100 lx significantly decreased the EEG power density of NREM sleep in the frequency range of 5.75–7.5 Hz, as compared with continuous darkness (Figure 2g), whereas neither acute white light nor red light at 100 lx altered the delta power activity of NREM sleep, as previously reported8. When the light intensity was lowered to 30 or 20 lx, only acute red light at 20 lx decreased the power in the frequency range of 0–1.25 Hz, with an increase in the frequencies of 12.5–24.75 Hz during NREM sleep, as compared with continuous darkness (Figure 2h and 2i). Thus, both acute white light and red light at intensities of 20 lx or higher significantly changed the sleep–wake architecture and EEG power density during the dark phase, thereby indicating that exposure to red light at 20 lx or higher disturbs sleep–wake behaviors at night and that illumination for nocturnal experiments should use low intensities of red light.
Episode number, mean duration (a–c), stage transition (d–f) and EEG power density of NREM sleep (g–i) during 5-h light-on phase. Black, white and red bars show the profiles of continuous darkness, white and red light treatments, respectively. Values are means±SEM (100 lx, n=9; 30 lx, n=5–7; 20 lx, n=5, 6). *P<0.05, **P<0.01 compared with continuous darkness, assessed via one-way ANOVA followed by a Bonferroni test. Blue and red horizontal bars indicate the location of a statistically significant difference (P<0.05, two-tailed unpaired t-test) between white or red light and continuous darkness, respectively. R, REM sleep; S, NREM sleep; W, wake.
Acute red light at an intensity of 10 lx did not influence the amount of NREM sleep or REM sleep in the dark phase
To explore the intensity of the red light without disturbing the sleep–wake behavior in the dark phase, we lowered the light intensity to 10 lx and examined the sleep profiles of the mice exposed to 1 h/1 h L/D cycles from ZT12–22. Darkness exposure during the entire dark phase served as the control. Acute white light at 10 lx, compared with darkness, increased the amount of NREM sleep but not REM sleep significantly, thus suggesting that REM sleep may be less sensitive than NREM sleep to acute white light pulses. However, acute red light at 10 lx, compared with darkness, did not alter NREM sleep or REM sleep (Figure 3a and 3b).
Effects of 10 lx white or red light pulses on NREM sleep and REM sleep during the dark phase. (a) Time course changes of NREM sleep and REM sleep in mice exposed to 100 lx white light in the day and a 1 h/1 h L (10 lx white or red light)/D cycle or continuous darkness at night. Each cycle represents the hourly mean±SEM of NREM sleep and REM sleep. Black, white and red circles indicate the profiles of continuous darkness, white light and red light treatments, respectively. The horizontal filled and open bars on the x axes indicate light-off and light-on treatments, respectively, at night. *P<0.05, **P<0.01 indicate significant differences between white light and continuous darkness. ##P<0.01 indicates significant differences between white light and red light. n.s. indicates no significant difference between red light and continuous darkness. Data shown are assessed via two-way ANOVA followed by a Bonferroni test. (b) Total time spent in NREM sleep and REM sleep of the groups of 10 lx white or red light, as well as continuous darkness, during 5-h light-on phase. Black, white and red bars show the profiles of continuous darkness, white light and red light treatments, respectively. Values are means±SEM (constant darkness n=7; 10 lx, n=6, 7). *P<0.05 compared with continuous darkness, assessed via one-way ANOVA followed by a Bonferroni test.
Acute white light but not red light at an intensity of 10 lx disrupted the sleep–wake architecture and EEG power density during the dark phase
Next, we determined the episode number, mean duration and transition number of NREM sleep, REM sleep and wakefulness in mice exposed to white or red light at an intensity of 10 lx or darkness. Acute white light at 10 lx, as compared with darkness/red light at 10 lx, decreased the mean duration of wakefulness and, as compared with red light at 10 lx, increased the stage transitions (from NREM sleep to wakefulness, from wakefulness to NREM sleep) and the episode number of wakefulness and NREM sleep (Figure 4a and 4b). Moreover, acute 1 h/1 h L/D exposure of white light at 10 lx decreased the power activity of NREM sleep in the frequency range of 0–1.5 Hz, with an increase in the frequencies of 8.75–24.75 Hz, as compared with continuous darkness (Figure 4c). However, there were no differences in the sleep–wake architecture and power density between red light at 10 lx and darkness. These results indicate that acute red light at 10 lx does not affect sleep–wake behavior during the dark phase.
Episode number, mean duration (a), stage transition (b) and EEG power density of NREM sleep (c) during 5-h light-on phase. Black, white and red bars show the profiles of continuous darkness, white and red light (10 lx) treatments, respectively. Values are means±SEM (n=6, 7). **P<0.01 indicates significant differences between two groups. Data were assessed via one-way ANOVA followed by a Bonferroni test. Blue horizontal bars indicate the location of a statistically significant difference (P<0.05, two-tailed unpaired t-test) between white light and continuous darkness. R, REM sleep; S, NREM sleep; W, wake.
Acute white light but not red light at an intensity of 10 lx disturbed sleep–wake profiles on the following day
To assess the after effects of acute white and red light at 10 lx, we examined the sleep–wake profile on the second day after 1 h/1 h L/D exposure. We found that white light at 10 lx caused an increase in both NREM and REM sleep for 2 h when the mice were moved to the dark phase after cessation of 1 h/1 h L/D exposure (Figure 5a and 5b). However, red light at 10 lx did not affect the sleep–wake profiles on the second day, as compared with the control group (Figure 5c and 5d). These data indicated that 1 h/1 h L/D exposure of white light but not red light at 10 lx disturbed sleep–wake profiles on the second day.
NREM sleep and REM sleep in mice after cessation of exposure to 1 h/1 h white light (WL) or red light (RL) at 10 lx/D. (a) Time course changes of NREM sleep and REM sleep in mice after ending exposure to 1 h/1 h WL at 10 lx/D. Each cycle represents the hourly mean±SEM of NREM sleep and REM sleep. White and blue circles indicate the profiles of the baseline day and the day after cessation of exposure to 1 h/1 h WL at 10 lx/D, respectively. The horizontal open and filled bars on the x axes indicate the 12-h dark and 12-h light periods, respectively. *P<0.05, **P<0.01 compared with baseline, assessed via two-way ANOVA followed by a Bonferroni test. (b) Total time spent in NREM sleep and REM sleep during the first 2 h of the dark phase after cessation of 1 h/1 h WL at 10 lx/D. (c) Time course changes of NREM sleep and REM sleep in mice after cessation of exposure to 1 h/1 h RL at 10 lx/D. Each cycle represents the hourly mean±SEM of NREM sleep and REM sleep. White and red circles indicate the profiles of the baseline day and the day after ending exposure to 1 h/1 h RL at 10 lx/D, respectively. The horizontal open and filled bars on the x axes indicate the 12-h dark and 12-h light periods, respectively. Data shown are assessed via two-way ANOVA followed by a Bonferroni test. (d) Total time spent in NREM sleep and REM sleep during the entire day, 12-h light phase and 12-h dark phase of the baseline day and the day after cessation of exposure to 1 h/1 h RL at 10 lx/D, respectively. Values are means±SEM (n=4). No significant difference compared with the baseline, assessed via a two-tailed paired t-test.
Red light exposure at an intensity of 10 lx during the entire dark phase did not affect the amount of NREM or REM sleep or the sleep–wake architecture but altered EEG power density
To investigate the effects of white and red light at 10 lx during the entire dark period on sleep and wakefulness, we examined the sleep–wake profiles of the mice exposed to 12-h white and red light at an intensity of 10 lx. Darkness exposure during the entire dark phase served as the control. We found that 10 lx white light exposure during the entire dark period significantly increased NREM and REM sleep for 2 h and that there was an increased tendency toward sleep in the subsequent hour, but this effect was not statistically significant when compared with the entire dark group (Figure 6a). When the sleep amount during the 3 h after light exposure was calculated, the total amounts of NREM and REM sleep were also increased (Figure 6b). Moreover, white light at 10 lx disturbed the sleep–wake architecture. White light at 10 lx increased the stage transitions (from NREM sleep to wakefulness, from wakefulness to NREM sleep, from NREM sleep to REM sleep and from REM sleep to wakefulness), the episode number for all stages, and the mean duration of REM sleep, as compared with darkness/red light at 10 lx (Figure 6c and 6d). However, mice exposed to red light at 10 lx exhibited the same sleep–wake profiles as mice that stayed in darkness (Figure 6a and 6b). These findings clearly indicated that dim red light does not affect the sleep amount, whereas dim white light does. Furthermore, there were no differences between the groups of 10 lx red light and darkness in terms of episode number and mean duration, and stage transition number among NREM sleep, REM sleep and wakefulness (Figure 6c and 6d). These results demonstrated that 10 lx red light exposure during the entire dark period did not affect the amount or the architecture of sleep and wakefulness. Moreover, 12-h exposure of white and red light at 10 lx decreased the delta power activity of NREM sleep in the frequency range of 0.75–2.75 and 1.0–1.25 Hz within the first 3 h, as well as 0.5–2.25 and 0.5–1.0 Hz within the entire 12 h during the dark phase, respectively, compared with darkness. However, white light at 10 lx increased the power in the frequency ranges of 4.25–6 and 7.75–24.75 Hz within the first 3 h and of 4–5.25 and 7.5–24.75 Hz within the entire 12 h during the dark phase, as compared with darkness. In contrast, 12-h exposure of red light at 10 lx increased the power in the frequency range of 4.25–5.5 Hz within the first 3 h and of 4–5.25 Hz in the entire 12 h during the dark phase, compared with darkness (Figure 6e and 6f). These results indicated that 12-h exposure at 10 lx of both white and red light in the entire dark period influences the EEG power density.
Effects of 10 lx white or red light exposure for 12 h on sleep during the entire dark phase. (a) Time course changes in NREM sleep and REM sleep in mice exposed to 100 lx white light (WL) during the day and exposed to darkness, 10 lx WL and red light (RL) at night. Each cycle represents the hourly mean±SEM of NREM and REM sleep. Black, white and red circles indicate the profiles of darkness, white and red light treatments, respectively. The horizontal black, white and red bars on the x axes indicate darkness, white light and red light treatments, respectively. *P<0.05, **P<0.01 indicate significant differences between white light and darkness. #P<0.05, ##P<0.01 indicate significant differences between white light and red light. Data shown are assessed via two-way ANOVA followed by a Bonferroni test. (b) Total time spent in NREM sleep and REM sleep for 3 h after 10 lx white or red light treatment. Black, white and red bars show the profiles of darkness, white and red light treatments, respectively. **P<0.01 or ##P<0.01 indicates significant differences compared with darkness or to red light, respectively. Data were assessed via one-way ANOVA followed by a Bonferroni test. (c, d) Stage transition (c), episode number and mean duration (d) in a 3-h period after the treatment of 10 lx white or red light. Black, white and red bars show the profiles of darkness, white and red light treatments, respectively. *P<0.05, **P<0.01 indicate significant differences between two groups. Data were assessed via one-way ANOVA followed by a Bonferroni test. R, REM sleep; S, NREM sleep; W, Wake. (e, f), EEG power density of NREM sleep within the first 3 h (e) and the entire 12 h (f) during the dark phase. Values are means±SEM (n=5–8). Blue and red horizontal bars indicate the location of a statistically significant difference (P<0.05, two-tailed unpaired t-test) between white or red light and continuous darkness, respectively.
Our findings demonstrated that white or red light at an intensity of ⩾20 lx has a potent sleep-inducing effect on mice. The amount of light-inducing sleep depends on both the wavelength and the intensity of the light. We provided the first evidence of the effects of red and white light at various intensities on the amount and architecture of NREM sleep and REM sleep in mice and compared the different effects of red and white light on sleep–wake behavior. The results showed that both white and red light above 10 lx induced potent NREM sleep and REM sleep during the dark period, although it has often been reported that ipRGCs are not sensitive to red wavelength light14, 15, 16.
Studies have shown that ipRGCs are necessary in the direct photic regulation of sleep and that the ipRGCs-dependent photosomnolence effect is related to the activation of the ventrolateral preoptic area and superior colliculus12. Here, we showed that red light at an intensity only at or below 10 lx did not influence NREM sleep or REM sleep, thus suggesting that red light with an intensity above 10 lx is strong enough to activate the pathway from the ipRGCs to sleep-promoting nuclei. In contrast, white light had a potent effect on sleep, although the intensity was as low as 10 lx. These results are consistent with previous findings that the acute light-induction of sleep is mediated by ipRGCs, which are minimally sensitive to red light14, 15, 16.
Hypnotic candidate drugs are usually administered to experimental nocturnal animals, such as mice27 and rats28, during the animals’ active phases, and most of the time, tested drugs are administered during the dark period. A clear view of the surroundings is necessary for experimenters to perform the manipulations. Therefore, light exposure is inevitable in most nocturnal behavioral experiments. However, light exposure may have various effects on animals and lead to desynchrony between circadian rhythms and the external environment29, which might result in sub-health or emotional alterations30. Moreover, light has profound effects on sleep through both entrainment and photosomnolence effects31, 32. Thus, avoiding the influence of light is crucial while performing nocturnal experiments to ensure the reproducibility and reliability of the data. According to our results, red light at 10 lx appears to be the most appropriate light condition to adopt during nocturnal behavioral experiments.
Diazepam, one of the best-known sedative-hypnotics, is often tested as a positive control drug for screening assays in laboratories. It is of great importance to maintain the reproducibility of the effect of a positive control, which directly influences the reliability of the candidate drugs. However, the sleep latency and sleep duration induced by diazepam for identical dosages within the same species vary among laboratories33, 34, 35. In our opinion, the difference may be due to the various intensities of red light; the intensity of red light is recommended to be 10 lx or less.
Melatonin exerts critical regulatory effects on both central and peripheral systems, such as circadian rhythms, sleep, mood, metabolism, cardiovascular and immune systems, cell proliferation and pain. Nocturnal light exposure can acutely suppress melatonin secretion by the pineal gland and may disturb the circadian rhythms and other physiological functions in melatonin-proficient animals. However, the C57BL/6 mice used for all experimental procedures in the present study are referred to as ‘melatonin-deficient’, thus indicating that they produce very low levels of melatonin in the pineal gland36, 37. Therefore, melatonin may not mediate light-induced sleep in C57BL/6 mice.
Nighttime light exposure is increasingly common in modern society, and there are a variety of reasons for why so many individuals are exposed to light at night, including shift work and watching TV. Apart from that, many people sleep with a nightlight on to make it convenient to care for children or elderly household members. Thus, it is important to develop a type of nighttime light source that does not affect sleep.
Shift work may also increase the risk of developing mood disorders38, depression and impaired learning and memory39. For humans, light exposure is less harmful if most blue wavelengths are filtered out40, because the ipRGCs are maximally sensitive to blue light41. This finding is consistent with the viewpoint that the effects of nighttime light exposure on physiology and behavior are mediated by ipRGCs4. Our results suggest that red light at 10 lx cannot influence the amount or the architecture of sleep and alertness during a dark period because 10 lx red light might be too dim to activate ipRGCs. Therefore, red light at an intensity of 10 lx has the potential to be adopted as a healthy level of night illumination for use in places such as homes and hospitals.
Conclusions
In conclusion, red light induces potent NREM sleep and REM sleep at intensities above 10 lx in the dark phase. Our data indicate that 10 lx red light should be regarded as the appropriate illuminating condition for nocturnal sleep–wake-related behavioral experiments. Nocturnal light exposure is sometimes unavoidable during behavioral experiments taking place at night in laboratories and has become common in daily life. Our results highlight the need to limit both the wavelength and intensity of light in nocturnal sleep–wake-related behavioral experiments, and they further provide suggestions for healthy nighttime lighting for use in daily life.
References
Borbély AA . A two process model of sleep regulation. Hum Neurobiol 1982; 1: 195–204.
Daan S, Beersma DG, Borbély AA . Timing of human sleep: recovery process gated by a circadian pacemaker. Am J Physiol 1984; 246: R161–R183.
Muindi F, Zeitzer JM, Heller HC . Retino-hypothalamic regulation of light-induced murine sleep. Front Syst Neurosci 2014; 8: 135.
Bedrosian TA, Vaughn CA, Galan A, Daye G, Weil ZM et al. Nocturnal light exposure impairs affective responses in a wavelength-dependent manner. J Neurosci 2013; 33: 13081–13087.
Golombek DA, Rosenstein RE . Physiology of circadian entrainment. Physiol Rev 2010; 90: 1063–1102.
Nelson DE, Takahashi JS . Sensitivity and integration in a visual pathway for circadian entrainment in the hamster (Mesocricetus auratus. J Physiol 1991; 439: 115–145.
Cajochen C, Zeitzer JM, Czeisler CA, Dijk DJ . Dose-response relationship for light intensity and ocular and electroencephalographic correlates of human alertness. Behav Brain Res 2000; 115: 75–83.
Alföldi P, Franken P, Tobler I, Borbély AA . Short light-dark cycles influence sleep stages and EEG power spectra in the rat. Behav Brain Res 1991; 43: 125–131.
Borbély AA, Huston JP, Waser PG . Control of sleep states in the rat by short light-dark cycles. Brain Res 1975; 95: 89–101.
Borbély AA . Sleep and motor activity of the rat during ultra-short light-dark cycles. Brain Res 1976; 114: 305–317.
Altimus CM, Guler AD, Villa KL, McNeill DS, Legates TA et al. Rods-cones and melanopsin detect light and dark to modulate sleep independent of image formation. Proc Natl Acad Sci USA 2008; 105: 19998–20003.
Lupi D, Oster H, Thompson S, Foster RG . The acute light-induction of sleep is mediated by OPN4-based photoreception. Nat Neurosci 2008; 11: 1068–1073.
Tsai JW, Hannibal J, Hagiwara G, Colas D, Ruppert E et al. Melanopsin as a sleep modulator: circadian gating of the direct effects of light on sleep and altered sleep homeostasis in Opn4(−/−) mice. PLoS Biol 2009; 7: e1000125.
Lockley SW, Brainard GC, Czeisler CA . High sensitivity of the human circadian melatonin rhythm to resetting by short wavelength light. J Clin Endocrinol Metab 2003; 88: 4502–4505.
Newman LA, Walker MT, Brown RL, Cronin TW, Robinson PR . Melanopsin forms a functional short-wavelength photopigment. Biochemistry 2003; 42: 12734–12738.
Brainard GC, Sliney D, Hanifin JP, Glickman G, Byrne B et al. Sensitivity of the human circadian system to short-wavelength (420-nm) light. J Biol Rhythms 2008; 23: 379–386.
Rebec KM, Klanjšek-Gunde M, Bizjak G, Kobav MB . White LED compared with other light sources: age-dependent photobiological effects and parameters for evaluation. Int J Occup Saf Ergon 2015; 21: 391–398.
Huang ZL, Qu WM, Eguchi N, Chen JF, Schwarzschild MA et al. Adenosine A2A, but not A1, receptors mediate the arousal effect of caffeine. Nat Neurosci 2005; 8: 858–859.
Paxinos G, Franklin KBJ . The Mouse Brain in Stereotaxic Coordinates. Burlington: Gulf Professional Publishing; 2004.
Huang ZL, Qu WM, Li WD, Mochizuki T, Eguchi N et al. Arousal effect of orexin A depends on activation of the histaminergic system. Proc Natl Acad Sci USA 2001; 98: 9965–9970.
Qu WM, Huang ZL, Xu XH, Aritake K, Eguchi N et al. Lipocalin-type prostaglandin D synthase produces prostaglandin D2 involved in regulation of physiological sleep. Proc Natl Acad Sci USA 2006; 103: 17949–17954.
Qu WM, Xu XH, Yan MM, Wang YQ, Urade Y et al. Essential role of dopamine D2 receptor in the maintenance of wakefulness, but not in homeostatic regulation of sleep, in mice. J Neurosci 2010; 30: 4382–4389.
Yan MM, Xu XH, Huang ZL, Yao MH, Urade Y et al. Selection of optimal epoch duration in assessment of rodent sleep-wake profiles. Sleep Biol Rhythms 2011; 9: 46–55.
Chen L, Yin D, Wang TX, Guo W, Dong H et al. Basal forebrain cholinergic neurons primarily contribute to inhibition of electroencephalogram delta activity, rather than inducing behavioral wakefulness in mice. Neuropsychopharmacology 2016; 41: 2133–2146.
Huang ZL, Mochizuki T, Qu WM, Hong ZY, Watanabe T et al. Altered sleep-wake characteristics and lack of arousal response to H3 receptor antagonist in histamine H1 receptor knockout mice. Proc Natl Acad Sci USA 2006; 103: 4687–4692.
Oh JH, Yang SJ, Do YR . Healthy, natural, efficient and tunable lighting: four-package white LEDs for optimizing the circadian effect, color quality and vision performance. Light Sci Appl 2014; 3: e141, doi:10.1038/lsa.2014.22.
Arble DM, Bass J, Laposky AD, Vitaterna MH, Turek FW . Circadian timing of food intake contributes to weight gain. Obesity (Silver Spring) 2009; 17: 2100–2102.
Hotchkiss AK, Ostby JS, Vandenburgh JG, Gray LE Jr. . Androgens and environmental antiandrogens affect reproductive development and play behavior in the Sprague-Dawley rat. Environ Health Perspect 2002; 110 (Suppl 3): 435–439.
Bedrosian TA, Galan A, Vaughn CA, Weil ZM, Nelson RJ . Light at night alters daily patterns of cortisol and clock proteins in female siberian hamsters. J Neuroendocrinol 2013; 25: 590–596.
Healy D, Minors DS, Waterhouse JM . Shiftwork, helplessness and depression. J Affect Disord 1993; 29: 17–25.
LeGates TA, Fernandez DC, Hattar S . Light as a central modulator of circadian rhythms, sleep and affect. Nat Rev Neurosci 2014; 15: 443–454.
Morin LP . A path to sleep is through the eye. eNeuro 2015; 2 (2): e0069–14.2015.
Cho S, Yang H, Yoon M, Kim J, Kim D et al. Arousal inhibitory effect of phlorotannins on caffeine in pentobarbital-induced mice. Fish Aquat Sci 2014; 17: 13–18.
Cho S, Yoon M, Pae AN, Jin YH, Cho NC et al. Marine polyphenol phlorotannins promote non-rapid eye movement sleep in mice via the benzodiazepine site of the GABAA receptor. Psychopharmacology (Berl) 2014; 231: 2825–2837.
Qu WM, Yue XF, Sun Y, Fan K, Chen CR et al. Honokiol promotes non-rapid eye movement sleep via the benzodiazepine site of the GABAA receptor in mice. Brit J Pharmacol 2012; 167: 587–598.
Gómez-Corvera A, Cerrillo I, Molinero P, Naranjo MC, Lardone PJ et al. Evidence of immune system melatonin production by two pineal melatonin deficient mice, C57BL/6 and Swiss strains. J Pineal Res 2009; 47: 15–22.
Liu YY, Yin D, Chen L, Qu WM, Chen CR et al. Piromelatine exerts antinociceptive effect via melatonin, opioid, and 5HT1A receptors and hypnotic effect via melatonin receptors in a mouse model of neuropathic pain. Psychopharmacology (Berl) 2014; 231: 3973–3985.
Dumont M, Beaulieu C . Light exposure in the natural environment: relevance to mood and sleep disorders. Sleep Med 2007; 8: 557–565.
Fonken LK, Kitsmiller E, Smale L, Nelson RJ . Dim nighttime light impairs cognition and provokes depressive-like responses in a diurnal rodent. J Biol Rhythms 2012; 27: 319–327.
Ando K, Kripke DF . Light attenuation by the human eyelid. Biol Psychiatry 1996; 39: 22–25.
Bailes HJ, Lucas RJ . Human melanopsin forms a pigment maximally sensitive to blue light (λmax≈479 nm) supporting activation of Gq/11 and Gi/o signalling cascades. Proc Biol Sci 2013; 280: 20122987.
Acknowledgements
We thank Meng-Di Dong and Yan-Dan Lin (School of Information Science and Technology, Fudan University) for detecting the spectra of LED lamps. This work was supported in part by grants-in-aid for scientific research from the National Natural Science Foundation of China (31530035, 81420108015, 81301135, 31471064, 31421091, 81471344), the National Basic Research Program of China (2015CB856401, 2011CB711000), a key laboratory program of the Education Commission of Shanghai Municipality (ZDSYS14005), and the Shanghai Committee of Science and Technology (14JC1400900, 17ZR1402000, 13dz2260700).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Zhang, Z., Wang, HJ., Wang, DR. et al. Red light at intensities above 10 lx alters sleep–wake behavior in mice. Light Sci Appl 6, e16231 (2017). https://doi.org/10.1038/lsa.2016.231
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/lsa.2016.231
Keywords
This article is cited by
-
Insomnia-related rodent models in drug discovery
Acta Pharmacologica Sinica (2024)
-
GBA-AAV mitigates sleep disruptions and motor deficits in mice with REM sleep behavior disorder
npj Parkinson's Disease (2024)
-
Nucleus Accumbens Corticotropin-Releasing Hormone Neurons Projecting to the Bed Nucleus of the Stria Terminalis Promote Wakefulness and Positive Affective State
Neuroscience Bulletin (2024)
-
Parasubthalamic calretinin neurons modulate wakefulness associated with exploration in male mice
Nature Communications (2023)
-
Diurnal Characteristics of the Orexin System Genes and Its Effects on Pathology at Early Stage in 3xTg-AD Mice
NeuroMolecular Medicine (2023)