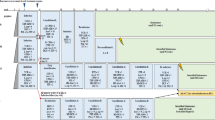
Abstract
Adults with acute lymphoblastic leukemia (ALL) do worse than children. From 7/2008 to 12/2014, Nordic and Baltic centers treated 1509 consecutive patients aged 1–45 years with Philadelphia chromosome-negative ALL according to the NOPHO ALL2008 without cranial irradiation. Overall, 1022 patients were of age 1–9 years (A), 266 were 10–17 years (B) and 221 were 18–45 years (C). Sixteen patients (three adults) died during induction. All others achieved remission after induction or 1–3 intensive blocks. Subsequently, 45 patients (12 adults) died, 122 patients relapsed (32 adults) with a median time to relapse of 1.6 years and 13 (no adult) developed a second malignancy. Median follow-up time was 4.6 years. Among the three age groups, older patients more often had higher risk ALL due to T-ALL (32%/25%/9%, P<0.001), KMT2A rearrangements (6%/5%/3%, P<0.001) and higher day 29 residual leukemia for B-lineage (P<0.001), but not T-ALL (P=0.53). Event-free survival rates (pEFS5y) were 89±1% (A), 80±3% (B) and 74±4% (C) with significant differences only for non-high risk groups. Except for thrombosis, pancreatitis and osteonecrosis, the risk of 19 specified toxicities was not enhanced by age above 10 years. In conclusion, a pediatric-based protocol is tolerable and effective for young adults, despite their increased frequency of higher risk features.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Pui CH, Yang JJ, Hunger SP, Pieters R, Schrappe M, Biondi A et al. Childhood acute lymphoblastic leukemia: progress through collaboration. J Clin Oncol 2015; 33: 2938–2948.
Toft N, Schmiegelow K, Klausen TW, Birgens H . Adult acute lymphoblastic leukaemia in Denmark. A national population-based retrospective study on acute lymphoblastic leukaemia in Denmark 1998–2008. Br J Haematol 2012; 157: 97–104.
Stock W, Johnson JL, Stone RM, Kolitz JE, Powell BL, Wetzler M et al. Dose intensification of daunorubicin and cytarabine during treatment of adult acute lymphoblastic leukemia: results of Cancer and Leukemia Group B Study 19802. Cancer 2013; 119: 90–98.
Schmiegelow K, Forestier E, Hellebostad M, Heyman M, Kristinsson J, Soderhall S et al. Long-term results of NOPHO ALL-92 and ALL-2000 studies of childhood acute lymphoblastic leukemia. Leukemia 2010; 24: 345–354.
Goldstone AH, Richards SM, Lazarus HM, Tallman MS, Buck G, Fielding AK et al. In adults with standard-risk acute lymphoblastic leukemia, the greatest benefit is achieved from a matched sibling allogeneic transplantation in first complete remission, and an autologous transplantation is less effective than conventional consolidation/maintenance chemotherapy in all patients: final results of the International ALL Trial (MRC UKALL XII/ECOG E2993). Blood 2008; 111: 1827–1833.
Boissel N, Auclerc MF, Lheritier V, Perel Y, Thomas X, Leblanc T et al. Should adolescents with acute lymphoblastic leukemia be treated as old children or young adults? Comparison of the French FRALLE-93 and LALA-94 trials. J Clin Oncol 2003; 21: 774–780.
de Bont JM, Holt B, Dekker AW, van der Does-van den Berg, Sonneveld P, Pieters R . Significant difference in outcome for adolescents with acute lymphoblastic leukemia treated on pediatric vs adult protocols in the Netherlands. Leukemia 2004; 18: 2032–2035.
Hallbook H, Gustafsson G, Smedmyr B, Soderhall S, Heyman M . Treatment outcome in young adults and children >10 years of age with acute lymphoblastic leukemia in Sweden: a comparison between a pediatric protocol and an adult protocol. Cancer 2006; 107: 1551–1561.
Nachman J, Sather HN, Buckley JD, Gaynon PS, Steinherz PG, Tubergen DG et al. Young adults 16-21 years of age at diagnosis entered on Childrens Cancer Group acute lymphoblastic leukemia and acute myeloblastic leukemia protocols: results of treatment. Cancer 1993; 71 (10 Suppl): 3377–3385.
Ramanujachar R, Richards S, Hann I, Goldstone A, Mitchell C, Vora A et al. Adolescents with acute lymphoblastic leukaemia: outcome on UK national paediatric (ALL97) and adult (UKALLXII/E2993) trials. Pediatr Blood Cancer 2007; 48: 254–261.
Stock W, La M, Sanford B, Bloomfield CD, Vardiman JW, Gaynon P et al. What determines the outcomes for adolescents and young adults with acute lymphoblastic leukemia treated on cooperative group protocols? A comparison of Children’s Cancer Group and Cancer and Leukemia Group B studies. Blood 2008; 112: 1646–1654.
Gokbuget N, Dombret H, Ribera JM, Fielding AK, Advani A, Bassan R et al. International reference analysis of outcomes in adults with B-precursor Ph-negative relapsed/refractory acute lymphoblastic leukemia. Haematologica 2016; 101: 1524–1533.
Keegan TH, Ries LA, Barr RD, Geiger AM, Dahlke DV, Pollock BH et al. Comparison of cancer survival trends in the United States of adolescents and young adults with those in children and older adults. Cancer 2016; 122: 1009–1016.
Roberts KG, Li Y, Payne-Turner D, Harvey RC, Yang YL, Pei D et al. Targetable kinase-activating lesions in Ph-like acute lymphoblastic leukemia. N Engl J Med 2014; 371: 1005–1015.
Schmiegelow K, Heyman M, Gustafsson G, Lausen B, Wesenberg F, Kristinsson J et al. The degree of myelosuppression during maintenance therapy of adolescents with B-lineage intermediate risk acute lymphoblastic leukemia predicts risk of relapse. Leukemia 2010; 24: 715–720.
Toft N, Birgens H, Abrahamsson J, Bernell P, Griskevicius L, Hallbook H et al. Risk group assignment differs for children and adults 1–45 yr with acute lymphoblastic leukemia treated by the NOPHO ALL-2008 protocol. Eur J Haematol 2013; 90: 404–412.
Frandsen TL, Heyman M, Abrahamsson J, Vettenranta K, Sberg A, Vaitkeviciene G et al. Complying with the European Clinical Trials directive while surviving the administrative pressure—an alternative approach to toxicity registration in a cancer trial. Eur J Cancer 2014; 50: 251–259.
van Dongen JJ, Langerak AW, Bruggemann M, Evans PA, Hummel M, Lavender FL et al. Design and standardization of PCR primers and protocols for detection of clonal immunoglobulin and T-cell receptor gene recombinations in suspect lymphoproliferations: report of the BIOMED-2 concerted action BMH4-CT98-3936. Leukemia 2003; 17: 2257–2317.
van der Velden VH, Hochhaus A, Cazzaniga G, Szczepanski T, Gabert J, van Dongen JJ . Detection of minimal residual disease in hematologic malignancies by real-time quantitative PCR: principles, approaches, and laboratory aspects. Leukemia 2003; 17: 1013–1034.
Bjorklund E, Mazur J, Soderhall S, Porwit-MacDonald A . Flow cytometric follow-up of minimal residual disease in bone marrow gives prognostic information in children with acute lymphoblastic leukemia. Leukemia 2003; 17: 138–148.
Fine JP, Gray JR . A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc 1999; 94: 496–509.
Toft N, Birgens H, Abrahamsson J, Griskevicius L, Hallbook H, Heyman M et al. Toxicity profile and treatment delays in NOPHO ALL2008-comparing adults and children with Philadelphia chromosome-negative acute lymphoblastic leukemia. Eur J Haematol 2016; 96: 160–169.
Boissel N, Sender LS . Best practices in adolescent and young adult patients with acute lymphoblastic leukemia: a focus on asparaginase. J Adolesc Young Adult Oncol 2015; 4: 118–128.
DeAngelo DJ, Stevenson KE, Dahlberg SE, Silverman LB, Couban S, Supko JG et al. Long-term outcome of a pediatric-inspired regimen used for adults aged 18-50 years with newly diagnosed acute lymphoblastic leukemia. Leukemia 2015; 29: 526–534.
Seftel MD, Neuberg D, Zhang MJ, Wang HL, Ballen KK, Bergeron J et al. Pediatric-inspired therapy compared to allografting for Philadelphia chromosome-negative adult ALL in first complete remission. Am J Hematol 2016; 91: 322–329.
Nygaard U, Schmiegelow K . Dose reduction of coadministered 6-mercaptopurine decreases myelotoxicity following high-dose methotrexate in childhood leukemia. Leukemia 2003; 17: 1344–1348.
Maury S, Chevret S, Thomas X, Heim D, Leguay T, Huguet F et al. Rituximab in B-lineage adult acute lymphoblastic leukemia. N Engl J Med 2016; 375: 1044–1053.
Rowe JM, Buck G, Burnett AK, Chopra R, Wiernik PH, Richards SM et al. Induction therapy for adults with acute lymphoblastic leukemia: results of more than 1500 patients from the international ALL trial: MRC UKALL XII/ECOG E2993. Blood 2005; 106: 3760–3767.
Thomas X, Boiron JM, Huguet F, Dombret H, Bradstock K, Vey N et al. Outcome of treatment in adults with acute lymphoblastic leukemia: analysis of the LALA-94 trial. J Clin Oncol 2004; 22: 4075–4086.
Stock W, Luger SM, Advani AS, Geyer S, Harvey RC, Mullighan CG et al. Favorable outcomes for older adolescents and young adults (AYA) with acute lymphoblastic leukemia (ALL): early results of U.S. Intergroup Trial C10403. Blood ASH Ann Meet Abstr 2014; 124: 796.
Lund B, Asberg A, Heyman M, Kanerva J, Harila-Saari A, Hasle H et al. Risk factors for treatment related mortality in childhood acute lymphoblastic leukaemia. Pediatr Blood Cancer 2011; 56: 551–559.
Patel B, Kirkwood AA, Dey A, Marks DI, McMillan AK, Menne TF et al. Pegylated-asparaginase during induction therapy for adult acute lymphoblastic leukaemia: toxicity data from the UKALL14 trial. Leukemia 2017; 31: 58–64.
Pieters R, de Groot-Kruseman H, Van dV V, Fiocco M, van den Berg H, de B E et al. Successful therapy reduction and intensification for childhood acute lymphoblastic leukemia based on minimal residual disease monitoring: study ALL10 from the Dutch Childhood Oncology Group. J Clin Oncol 2016; 34: 2591–2601.
Mattano LA Jr, Devidas M, Nachman JB, Sather HN, Hunger SP, Steinherz PG et al. Effect of alternate-week versus continuous dexamethasone scheduling on the risk of osteonecrosis in paediatric patients with acute lymphoblastic leukaemia: results from the CCG-1961 randomised cohort trial. Lancet Oncol 2012; 13: 906–915.
Schmiegelow K, Muller K, Mogensen SS, Mogensen PR, Wolthers BO, Stoltze UK et al. Non-infectious chemotherapy-associated acute toxicities during childhood acute lymphoblastic leukemia therapy. F1000Res 2017; 6: 444.
Schmiegelow K, Attarbaschi A, Barzilai S, Escherich G, Frandsen TL, Halsey C et al. Consensus definitions of 14 severe acute toxic effects for childhood lymphoblastic leukaemia treatment: a Delphi consensus. Lancet Oncol 2016; 17: e231–e239.
Mattano LA Jr., Sather HN, Trigg ME, Nachman JB . Osteonecrosis as a complication of treating acute lymphoblastic leukemia in children: a report from the Children’s Cancer Group. J Clin Oncol 2000; 18: 3262–3272.
Wolthers BO, Frandsen TL, Abrahamsson J, Albertsen BK, Helt LR, Heyman M et al. Asparaginase-associated pancreatitis: a study on pheno-and genotype in the NOPHO ALL2008 protocol. Leukemia 2017; 31: 325–332.
Armstrong GT, Chen Y, Yasui Y, Leisenring W, Gibson TM, Mertens AC et al. Reduction in late mortality among 5-year survivors of childhood cancer. N Engl J Med 2016; 374: 833–842.
Bhatia S, Landier W, Hageman L, Chen Y, Kim H, Sun CL et al. Systemic exposure to thiopurines and risk of relapse in children with acute lymphoblastic leukemia: a Children's Oncology Group Study. JAMA Oncol 2015; 1: 287–295.
Schmiegelow K, Nielsen SN, Frandsen TL, Nersting J . Mercaptopurine/methotrexate maintenance therapy of childhood acute lymphoblastic leukemia: clinical facts and fiction. J Pediatr Hematol Oncol 2014; 36: 503–517.
Roberts KG, Pei D, Campana D, Payne-Turner D, Li Y, Cheng C et al. Outcomes of children with BCR-ABL1-like acute lymphoblastic leukemia treated with risk-directed therapy based on the levels of minimal residual disease. J Clin Oncol 2014; 32: 3012–3020.
Nielsen SN, Grell K, Nersting J, Frandsen TL, Hjalgrim LL, Schmiegelow K . Measures of 6-mercaptopurine and methotrexate maintenance therapy intensity in childhood acute lymphoblastic leukemia. Cancer Chemother Pharmacol 2016; 78: 983–994.
Acknowledgements
We thank all patients involved and clinicians assisting in ALL treatment and data collection.
Author contributions
NT designed the study, collected, analyzed, and interpreted data, wrote and edited the manuscript; KS is principal investigator for NOPHO ALL2008, designed the study, interpreted data and critically reviewed the manuscript; HB designed the study, interpreted data, served as an investigator and critically reviewed the manuscript; TWC performed statistical analyses and interpreted data; JA designed the study, served as investigator, collected data and edited the manuscript; TF designed the study, served as investigator, supervised toxicity reporting and edited the manuscript; HH designed the study, served as investigator, collected data and edited the manuscript; LG designed the study, served as investigator, collected data and edited the manuscript; MH designed the study, served as investigator and data manager, collected data and edited the manuscript; OGJ designed the study, served as investigator, collected data and edited the manuscript; KPa designed the study, served as investigator, collected data and edited the manuscript; KPr designed the study, served as investigator, collected data and edited the manuscript; PQP designed the study, served as investigator, collected data and edited the manuscript; GV designed the study, served as investigator, collected data and edited the manuscript; KV designed the study, served as investigator, collected data and edited the manuscript; AÅ designed the study, served as investigator, collected data and edited the manuscript; HM served as flow cytometry investigator, collected data and edited the manuscript; HOM served as PCR investigator, collected data and edited the manuscript; UNN served as cytogenetic investigator, collected data and edited the manuscript. All authors approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Toft, N., Birgens, H., Abrahamsson, J. et al. Results of NOPHO ALL2008 treatment for patients aged 1–45 years with acute lymphoblastic leukemia. Leukemia 32, 606–615 (2018). https://doi.org/10.1038/leu.2017.265
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2017.265
This article is cited by
-
Diagnostic and treatment strategies for pediatric acute lymphoblastic leukemia in low- and middle-income countries
Leukemia (2024)
-
Acute lymphoblastic leukaemia
Nature Reviews Disease Primers (2024)
-
Allogeneic haematopoietic stem cell transplantation might overcome the poor prognosis of adolescents and adult patients with T-lineage acute lymphoblastic leukaemia and CDKN2 deletion
Bone Marrow Transplantation (2024)
-
Increase in peg-asparaginase clearance as a predictor for inactivation in patients with acute lymphoblastic leukemia
Leukemia (2024)
-
Integration of genetics and MRD to define low risk patients with B-cell precursor acute lymphoblastic leukaemia with intermediate MRD levels at the end of induction
Leukemia (2024)