Abstract
The treatment landscape for patients with multiple myeloma (MM) is constantly evolving. Over the past decade, the introduction of novel agents such as proteasome inhibitors and immunomodulatory drugs has led to notable changes in therapeutic strategy, and improvements in survival, yet MM remains incurable in the vast majority of cases. More recently, a targeted approach to MM treatment has emerged, using monoclonal antibodies (mAbs) to target antigens expressed on the surface of MM cells. MAbs tested to date kill MM cells via the host’s immune system and/or by promoting apoptosis, and appear to have generally improved tolerability compared with currently available treatments. Due to their distinct mode of action, mAbs are promising both for patients who have exhausted current regimens, and as part of first-line treatments in newly diagnosed patients. This review examines the recent developments in mAb-based therapy for MM, primarily focused on those agents in ongoing clinical testing.
Similar content being viewed by others
Introduction
Multiple myeloma (MM) is a malignancy of antibody-secreting plasma cells.1 Globally, over 80 000 new cases of MM are reported each year, representing ~1% of all new cancer cases and 10% of all hematologic malignancies.2, 3 The incidence of MM increases with age, indicative of the accumulation of epigenetic/genetic changes during the typical development of the disease from monoclonal gammopathy of undetermined significance, through smoldering (asymptomatic) myeloma, to symptomatic MM.4 Clinically, symptomatic MM is characterized by end-organ damage, generally involving hypercalcemia, renal failure, anemia and bone marrow lesions (CRAB features).5 Skeletal pain and fatigue are common symptoms of MM, and can severely impact the patient’s quality of life.6
The overall median survival is ~5–6 years from diagnosis of MM,7 yet disease outcomes are strongly influenced by the characteristics of the cancer (for example, high-risk cytogenetics) and/or the patient (for example, age). In younger patients, autologous stem cell transplantation has led to improved progression-free survival (PFS) and overall survival (OS).8, 9 Here, patients receive induction therapy, which is typically a combination regimen based on an alkylating agent and/or a proteasome inhibitor (PI; for example, bortezomib [BORT] and carfilzomib [CAR]) and/or an immunomodulatory drug (IMiD; for example, lenalidomide [LEN], thalidomide [THAL] and pomalidomide [POM]), to reduce disease burden before high-dose chemotherapy and stem cell transplantation. As mentioned, however, MM is most prevalent in elderly patients, the majority of whom are ineligible for autologous stem cell transplantation. Induction therapy with novel agents has also improved survival in this population, although management of elderly patients is often complicated by comorbidities.10 Regardless of eligibility for autologous stem cell transplantation, maintenance therapy using novel agents is typically administered with the intention of sustaining disease response.
The development of novel agents over the past decade has improved outcomes in patients with MM,7 although the vast majority of patients will eventually relapse. Outcomes are generally worse for patients who have failed currently available treatments, with a median OS of 9 months estimated for patients who are refractory to PIs and IMiDs.11 As such, there is an unmet need for new therapies to increase survival for patients with MM. The demand is clearly high in patients with relapsed and/or refractory MM (RRMM) who have exhausted current treatment options, yet there is also an opportunity to attain deeper and more sustained response in front-line, or early-line, therapy. Tolerability is also a limitation of current treatments,12, 13, 14 particularly in the increasing elderly population with MM who are generally more susceptible to adverse events (AEs). Indeed, careful selection and management of patients with RRMM has been recommended to optimize the benefits of current treatments.15 As such, reduced toxicity would be a key attribute for new agents to facilitate their use in a greater proportion of patients. The corollary of these unmet treatment needs is the extensive pipeline of anti-MM drugs, focused on delivering new agents with novel modes of action.
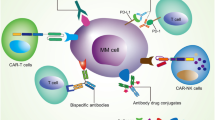
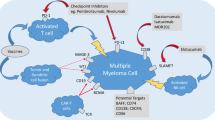
Of the spectrum of new agents in development for the treatment of MM, monoclonal antibodies (mAbs) have emerged as a potential strategy based on the range of antigens highly expressed on the surface of the malignant cell (Figure 1). In other cancers, mAb-based therapy is already established, with >10 antibodies having received approval from the FDA for solid or hematologic malignancies since 1997.16 Antibodies afford a targeted approach to treatment, with toxicity directed primarily against the malignant cell. Antibodies are also associated with a favorable tolerability profile, as most of the approved agents have different and less severe toxicities compared with standard chemotherapeutics.16 In this review, we evaluate the promise of targeted therapy for MM in light of the key clinical data, focusing on the exciting recent developments in mAb-based therapy for this disease.
CD38
CD38 is a multifunctional cell surface glycoprotein that serves as both a receptor for the transduction of activation/proliferation signals and an ectoenzyme that catalyzes the production of nucleotides involved in calcium signaling (Figure 2).17 As a receptor, CD38 engages the non-substrate ligand CD31, which is thought to elicit an intracellular signaling cascade via ZAP-70 and ERK1/2.17 As an ectoenzyme, CD38 catalyzes the conversion of nicotinamide adenine dinucleotide to cyclic adenosine diphosphate ribose, and is also involved in the hydrolysis of cyclic adenosine diphosphate ribose to adenosine diphosphate ribose.18 In MM, the operation of CD38 with fellow ectoenzymes PC-1 and CD73 has been postulated to enhance MM survival through the production of adenosine which can modulate the immune response.19 The ectoenzyme activity of CD38 also appears important in other malignancies, with postulated roles in proliferation and migration in chronic lymphocytic leukemia.20 CD38 expression is low in the normal bone marrow compartment, except on committed bone marrow progenitor cells. However, CD38 is highly expressed in >80% of cases of MM21 and serves as a marker for MM cells.
CD38 mode of action. ADCC, antibody-dependent cell-mediated cytotoxicity; ADCP, antibody-dependent cellular phagocytosis; ADPR, adenosine diphosphate ribose; cADPR, cyclic adenosine diphosphate ribose; CDC, complement-dependent cytotoxicity; mAb, monoclonal antibody; MAC, membrane attack complex; MM, multiple myeloma; NAADP, nicotinic acid adenine dinucleotide phosphate; NAD+, nicotinamide adenine dinucleotide; NADP, nicotinamide adenine dinucleotide phosphate; NK, natural killer.
There are presently three anti-CD38 mAbs in clinical development for the treatment of MM: daratumumab (DARA), SAR650984 and MOR202.
Daratumumab
DARA was generated from immunization of human transgenic HuMab-mice with recombinant CD38 protein.22 In preclinical models, DARA was shown to elicit cell death through four mechanisms: antibody-dependent cell-mediated cytotoxicity (ADCC), complement-dependent cytotoxicity (CDC), antibody-dependent cellular phagocytosis (ADCP) and apoptosis via crosslinking (crosslinkage with Fc receptors or anti-human IgG antibody).22, 23, 24, 25 ADCC was observed in all CD38+ MM cell lines, whereas CDC appeared dependent on CD38 expression level.22, 24 The anti-tumorigenic potential of DARA has also been explored in combination with other novel and established agents. Here, increased cell lysis was observed when DARA was added to LEN and/or BORT, or to the established triplet regimen of melphalan (MEL)–prednisone (PRED)–BORT.26, 27, 28
The clinical data reported for DARA have largely been from the phase I/II trials in patients with RRMM, either as monotherapy (NCT00574288; NCT01985126) or in combination with LEN and dexamethasone (DEX) (NCT01615029). In the expansion phase of the initial monotherapy trial, efficacy appeared dose related: the overall response rate (ORR; at least a partial response (PR)) was 10% for patients who received the 8 mg/kg dose and 35% for patients who received the higher 16 mg/kg dose (Table 1).29 Median PFS was also longer in the high-dose cohort (23 vs 14.9 weeks), although these data were immature at the time of presentation (May 2014).29 The apparent dose–response relationship is supported by pharmacokinetic (PK) data, which suggest that target-mediated clearance of this agent is reduced at higher doses,30 and the 16 mg/kg dose is being advanced in phase III trials. Results at the 16 mg/kg dose have also been recently presented from the second monotherapy trial (SIRIUS), testing daratumumab in 106 patients who had received at least three prior lines of therapy or were refractory to their most recent PI and IMiD combination.31 The ORR was 29% in this heavily pre-treated population (median of five prior therapies; 95% refractory to last PI and IMiD), with a median duration of response of 7.4 months. An ORR of ~20–30% was observed across subgroups (for example, according to age (⩾75 years) or renal function (creatinine clearance ⩾60 ml/min)), and irrespective of the agent(s) to which patients were previously refractory. The median PFS in this study was 3.7 months, and the estimated 1-year survival rate was 65%.31
In the combination study of DARA plus LEN and DEX in a moderately pre-treated population (median of two prior lines of therapy), the ORR was 91% (39/43) across the dose escalation and expansion phases.32 The majority of these patients (n=30) were assessed in the dose expansion phase at DARA 16 mg/kg, with ORR 87% in this section of the study.32 A high proportion of patients assessed in this combination study had received prior IMiD treatment (80%), although it should be noted that only 7% of the total population were refractory to LEN.32 Most recently, data have been released from an open label, phase Ib study of DARA in combination with a range of established backbone therapies (NCT01998971). Patients with newly diagnosed disease were treated with DARA plus BORT–DEX (n=6), BORT–MEL–PRED (n=8) or BORT–THAL–DEX (n=11), with all those evaluable achieving at least PR.33 The addition of DARA to POM–DEX was assessed in 24 patients with RRMM, with ORR 55% in the 11 patients available for efficacy assessment.33
The most common AEs associated with DARA treatment were infusion-related reactions, which were generally observed during the first infusion and predominantly Grade 1/2 in intensity.31, 34 In the monotherapy trials, prophylactic treatment was given, and infusion-related reactions were recorded in 43–50% of patients at the 16 mg/kg dose.29, 31 The most frequent Grade 3/4 AEs with single-agent daratumumab were thrombocytopenia and pneumonia in the dose expansion part of the original trial, and anemia and thrombocytopenia in the SIRIUS trial.29, 31 Of note, Grade ⩾3 thrombocytopenia and anemia occurred more frequently in patients who did not attain an objective response in the SIRIUS study, whereas there was no relationship between neutropenia and response.31 In the combination study with LEN–DEX, the majority of Grade ⩾3 AEs were hematologic, and the maximum tolerated dose (MTD) of DARA was not reached.35 DARA was also generally well tolerated in combination with alternate backbone regimens, with no major additional toxicity observed.33
Phase III studies of LEN–DEX±DARA (NCT02076009) and BORT–DEX±DARA (NCT02136134) in patients with RRMM, and BORT–MEL–PRED±DARA (NCT02195479) and LEN–DEX±DARA (NCT02252172) in newly diagnosed patients with MM, have been announced.
SAR650984
SAR650984 is a humanized mAb generated from immunization with murine 300-19 cells transfected to express human CD38.36 Similar to DARA, SAR650984 induces cell death via ADCC, ADCP and CDC in in vitro models.36 ADCC was observed in all the CD38+ lines tested, whereas the CDC activity was dependent on receptor density.36 SAR650984 also elicited a direct pro-apoptotic effect, capable of inducing crosslinking-independent apoptosis in addition to the crosslinking-dependent apoptosis observed with DARA.36, 37 Preclinical investigations also revealed that SAR650984 inhibited CD38 ectoenzyme activity, which has not been reported for DARA or MOR202.37 SAR650984 has demonstrated synergistic or additive antitumor effects in combination with LEN, BORT, CAR and MEL in mouse xenograft tumor models.38, 39, 40
Two ongoing phase I/II dose escalation studies are assessing SAR650984 in patients with RRMM: as a monotherapy (NCT01084252) or in combination with LEN and DEX (NCT01749969). In heavily pre-treated patients (median 6.5 prior lines of therapy including BORT, CAR, POM and LEN) who received single-agent SAR650984 0.3–20 mg/kg every other week, ORR was 27% overall, and 32% in patients who received SAR650984 at doses ⩾10 mg/kg41 Table 1). PK data indicated decreased clearance of SAR650984 with increasing dose, suggestive of a dose relationship.41 The MTD was not reached in this study, and additional dosing regimens are to be tested in the expansion phase of this study.
The combination therapy study, which also included heavily pre-treated patients (median seven prior treatment regimens), reported an ORR of 63% at SAR650984 doses of 10 mg/kg (Table 1).42 In addition, reductions in paraprotein of >90% were recorded in approximately one-third of patients at this dose.42 In contrast to the combination trial of DARA with LEN–DEX, the vast majority of patients in this study were relapsed or refractory to LEN, yet the ORR was 48% in this patient subpopulation.42 Notably, responses were also observed in patients refractory to BORT, CAR or POM.43 The overall median PFS was 6.2 months, yet in patients who had received only one to two lines of prior therapy (n=7) median PFS had not been reached at data cut-off.42 The PK data indicated no significant interaction between LEN and SAR650984.42, 43
As a monotherapy, the most common treatment-emergent AEs with SAR650984 were fatigue (53% of patients) and nausea (35%), and the most common drug-related Grade 3/4 event was pneumonia (8%).41 Infusion reactions (52%) were observed during the first cycle of treatment, and were predominantly Grade 1/2 in severity.41 Infusion reactions led to treatment discontinuation in two patients in the combination study, although these events decreased after Cycle 1 when prophylaxis was introduced.42 In the combination study, the most common Grade 3/4 events were cytopenias, specifically neutropenia and thrombocytopenia.42
MOR202
MOR202 is a fully human HuCAL IgG1 antibody, which has been shown to elicit cell death by ADCC and ADCP.44, 45 Preclinical data published to date have largely focused on MOR202 combination treatment, which has demonstrated that MOR202 antitumor activity is related to CD38 expression level, and is synergistic with POM and LEN and additive with BORT.44, 45, 46 Of note, both LEN and POM treatment were shown to increase CD38 expression, and thus enhance the cytotoxic effects of MOR202.44, 45, 46 In addition, treatment with both IMiDs induced activation of immune effector cells, further promoting ADCC and ADCP.44, 46 These independent activities of LEN and POM suggest that the combination of CD38-targeted agents with IMiDs should provide a potent strategy for MM treatment. In vivo studies using murine MM models have also demonstrated synergistic reductions in bone lysis when MOR202 was combined with LEN, BORT or MEL.47
Preliminary clinical data from a phase I/IIa study testing MOR202 with or without DEX in patients with RRMM have recently been reported.48 Among 23 patients assessed, only one achieved a response better than stable disease (Table 1). With the dosing schedule tested, the most common AEs were anemia, fatigue and nausea; the MTD of MOR202 was not reached in this study. Infusion-related reactions were observed in 31% of patients, yet only in those who received MOR202 alone, and these events occurred mostly during the first infusion. A transient antidrug antibody response to MOR202 was also observed in one patient. As part of the overall MOR202 development program, this mAb will also be evaluated in combination with LEN–DEX and POM–DEX in patients with RRMM.48
Anti-CD38 mAb summary
The anti-CD38 mAbs have shown potent activity in preclinical MM models both as single agents and in combination with other agents, with all three demonstrating ADCC and ADCP. ADCP may be particularly pertinent for treatment of MM, as high numbers of macrophages are present in the bone marrow. However, despite their similarities, there are also distinct differences between DARA, SAR650984 and MOR202. Each antibody targets a distinct epitope on CD38,22, 37 and there are differences in their modes of action. For example, DARA induces crosslinking-dependent apoptosis, whereas SAR650984 can promote apoptosis with or without crosslinking.49 Furthermore, SAR650984 is a much more potent inhibitor of CD38 ectoenzyme function than DARA or MOR202.37 This activity may provide another mechanism to suppress MM growth, based on the proposed role of CD38 ectoenzyme signaling in protecting MM cells in the bone marrow niche, although the clinical significance of such an activity is not clear. Combination therapy to date has focused on the addition of these mAbs to other novel agents, such as LEN which can enhance the activity of effector cells (for example, natural killer cells), and upregulate CD38 expression on MM cells. As receptor density impacts CDC for SAR650984 and DARA, and ADCC and ADCP for MOR202, LEN appears a natural partner for the anti-CD38 mAbs. Owing to their non-overlapping modes of action and distinct CD38-binding sites, the therapeutic combination of anti-CD38 mAbs is also conceivable.
The assessment of monoclonal immunoglobulin protein (M-protein) reduction by immunofixation electrophoresis is used to define the depth of response according to the International Myeloma Working Group criteria.50 Therapeutic mAbs may interfere with this assay and thereby influence clinical interpretation of response to therapy. Data were recently presented for a DARA immunofixation electrophoresis reflex assay (DIRA), in which the binding of an anti-idiotypic antibody to DARA altered its electrophoretic migration and thus distinguished this mAb from the disease-associated M-protein in patient samples.51 The use of such an assay may be necessary to confirm the reduction, and most specifically the absence (necessary for complete response (CR)50), of M-protein in future mAb trials.
CS1/SLAMF7
CS1 is a member of the signaling lymphocyte activating-molecule-related family and is highly expressed on the surface of normal plasma cells and MM cells.52 This molecule is also expressed on other lymphocytes, for example natural killer cells, although at lower levels. CS1 is normally involved in regulating the immune response,53 but appears to have a role in survival pathways in MM.54
Elotuzumab (ELO) is a humanized IgG1 mAb directed against human CS1.52 In preclinical models, ELO was shown to mediate cell death via ADCC and inhibit CS1-mediated MM cell adhesion to bone marrow stem cells, in a dose-dependent manner.55 In addition, ELO may act beyond ADCC by enhancing the cytotoxic activity of natural killer cells.56
Despite the promising preclinical data, a phase I clinical trial of ELO in patients with RRMM demonstrated that this agent was not effective as a monotherapy.57 Indeed, no objective responses were observed during the study and ~75% of patients had progressive disease.57 However, ELO at doses up to 20 mg/kg every other week was generally well tolerated,57 supporting its investigation as a part of a combination regimen.
In a phase I study of ELO plus BORT in patients with RRMM, the ORR was 48% (Table 1), including responses in two of three patients previously refractory to BORT.58 This level of response was also attained in 7 of 10 patients with high-risk cytogenetics.58 ELO–BORT–DEX was compared with BORT–DEX alone in a phase II trial, which demonstrated that, despite a similar ORR (65 vs 63%), the triplet regimen improved median PFS (9.7 months vs 6.9 months) over BORT–DEX alone in patients with RRMM.59 ELO has also been tested in a phase II trial with THAL and DEX in patients with RRMM, with an ORR of 40% reported.60
The majority of clinical data for ELO, however, has been attained in combination with LEN and DEX, with this triplet regimen tested in moderately pre-treated patients with RRMM in phase I, II and III trials. The phase I study demonstrated the tolerability and activity (ORR, 82%) of ELO–LEN–DEX61 with the 10 and 20 mg/kg ELO doses advanced to phase II. In a LEN-naive patient population, an overall ORR of 84% (61/73) was reported in the phase II study.62 However, efficacy appeared better in the 10 mg/kg cohort compared with the 20 mg/kg cohort, both in terms of ORR (92 vs 76%) and PFS (33 months vs 18.6 months).62 As expected, ORR and median PFS were reduced in patients with ⩾2 previous therapies compared with those who had received only one prior therapy (ORR, 78 vs 91%; PFS, 21.3 months vs 25.0 months),63 although these were the first data from a mAb study to demonstrate the impact of pre-treatment level on response. Comparison of response rates between different agents should thus take account of the number of lines of prior therapy. With the improved efficacy data at 10 mg/kg compared with 20 mg/kg, this lower dose was tested in 646 patients with RRMM in the recently reported phase III ELOQUENT-2 study. In a population who had received a median of two prior therapies (6% had received LEN), ELO–LEN–DEX significantly extended median PFS (co-primary end point) compared with LEN–DEX alone (19.4 months vs 14.9 months; hazard ratio, 0.57–0.85; P<0.0001).64 The benefit of ELO–LEN–DEX with respect to PFS was maintained in patients <65 and ⩾65 years, and in patients with the del(17p) and t(4;14) abnormalities.64 The 1-year PFS rate was higher in the ELO–LEN–DEX arm (68 vs 57%), and this difference was slightly greater at 2 years (41 vs 27%).64 ELO–LEN–DEX also improved ORR (co-primary end point) compared with LEN–DEX (79 vs 66%; P<0.0001)64 (Table 1); PFS was longer in patients who achieved at least PR with ELO–LEN–DEX than with LEN–DEX alone.64 The OS data were immature at the time of presentation.
As a single agent, the most common treatment-emergent AEs reported with ELO were chills and pyrexia, which were mostly Grade 1/2 in severity.57 Approximately 30% (10/34) of patients developed an infection during ELO therapy, although only 3/10 cases were considered related to ELO.57 In patients who received BORT and ELO in the phase II trial, the most frequent Grade ⩾3 events were thrombocytopenia and infections, although the rate of thrombocytopenia was slightly lower with ELO–BORT–DEX compared with BORT–DEX alone (9 vs 17%).59 Cytopenias were the most common Grade ⩾3 event when ELO was combined with LEN–DEX,62, 64 and were also observed with the ELO–THAL–DEX regimen,60 potentially due to the IMiDs used in these combinations. The most frequently observed non-hematologic Grade ⩾3 events with ELO plus THAL–DEX were asthenia, peripheral edema and fever.60 Infusion reactions were managed using prophylactic medication in the phase II and III combination regimens; these events occurred in ~10% of patients in the studies of ELO with LEN–DEX, BORT–DEX and THAL–DEX, and were mostly Grade 1/2 in intensity.59, 60, 63, 64
The ELO combination regimens appear more effective in patients who have received fewer lines of treatment, and a phase III trial of LEN–DEX±ELO is ongoing in patients with previously untreated MM (ELOQUENT-1; NCT01335399). Other ELO-based regimens are being tested, with encouraging initial safety data recently reported for ELO–LEN–BORT–DEX in newly diagnosed, high-risk patients,65 and a trial of ELO in combination with lirilumab (mAb that blocks interaction between killer-cell immunoglobulin receptors and their ligands66) or urelumab (anti-CD137 mAb that stimulates immune response67) currently recruiting (NCT02252263). Furthermore, a trial of single-agent ELO in patients with smoldering MM is planned (NCT01441973).
Interleukin-6
IL-6 is involved in the survival and proliferation of MM cells,68, 69 with a key role proposed during the early stages of disease development.70, 71 Siltuximab, a chimeric mAb against IL-6, has been shown to sensitize MM cells to DEX-mediated apoptosis72 and to enhance the activity of BORT and MEL, in preclinical models.73, 74 In combination with DEX, siltuximab was well tolerated but demonstrated only modest efficacy in patients with RRMM (ORR, 11%); as a single agent, siltuximab did not induce any objective responses.75 The benefit of adding siltuximab to BORT was also tested in BORT-naive patients with RRMM, but although ORR increased with the combination regimen (ORR, 55 vs 47%), there was no improvement in PFS or OS compared with BORT–placebo.76
As patients with RRMM may have residual MM cells that are less dependent on IL-6, siltuximab was tested in patients with newly diagnosed MM in a phase II study: siltuximab–BORT–MEL–PRED vs BORT–MEL–PRED. An EBMT ORR (PR+CR) of 88% was reported in patients who received siltuximab–BORT–MEL–PRED compared with 80% in the comparator arm, but the study did not meet its primary end point as the difference in CR rate between the study arms was <10%.77 The very good PR rate was significantly increased in the siltuximab arm, although this did not translate to differences in OS or PFS, raising questions over the association between this level of response and outcomes.77 The incidence of Grade ⩾3 events was slightly higher in the siltuximab arm (92 vs 81%), with an increased rate of hematologic events and infections.77
The possibility that IL-6 is involved during early MM development has also led to initial testing of siltuximab in patients with monoclonal gammopathy of undetermined significance or smoldering myeloma. Here, single-agent siltuximab reduced paraprotein in a subset of patients and did not yield any new safety signals.78 As such, siltuximab is being tested against placebo in a phase II trial in patients with high-risk smoldering myeloma (bone marrow plasma cells ⩾10% and either serum monoclonal protein ⩾3 g/dl, or abnormal free light chain ratio <0.126 or >8, and serum M-protein <3 g/dl but ⩾1 g/dl) (NCT01484275).
B-cell activating factor
B-cell activating factor (BAFF) is a member of the tumor necrosis factor superfamily that is produced in the bone marrow, and is overexpressed in MM cells compared with normal plasma cells at the transcript level.79 BAFF appears to promote survival of MM cells and protect them from DEX-induced apoptosis.80 Tabalumab (LY2127399) is a human mAb against BAFF that neutralizes the membrane-bound and soluble forms of this factor.81 In a phase I study in patients with RRMM (patients were not refractory to BORT), the combination of tabalumab with BORT–DEX achieved an ORR of 46%.82 Treatment discontinuation was reported owing to neuropathy, neuralgia, fatigue and thrombocytopenia.82 A phase II trial of tabalumab (100 or 300 mg) –BORT–DEX vs placebo–BORT–DEX has been completed (NCT01602224), although results have yet to be reported.
CD74
CD74 is a major histocompatibility complex class II chaperone involved in antigen presentation. It is also frequently expressed in MM cell lines and in the malignant plasma cells of patient samples,83 and is an attractive target based on its rapid internalization.84 Milatuzumab (hLL1) is a humanized anti-CD74 mAb that, when crosslinked, has shown anti-proliferative activity against MM cells both alone or in combination treatments in preclinical models.85 In a phase I dose escalation study in heavily pre-treated patients with RRMM (median five prior treatments), the best response achieved with 4 weeks’ milatuzumab monotherapy was stable disease.86 Modifications to infusion time and prophylactic treatment appeared to improve the tolerability of this agent, which was otherwise generally well tolerated.86 A phase I/II trial of milatuzumab conjugated with doxorubicin is ongoing in patients with RRMM (NCT01101594).
CD138
CD138/syndecan-1 is expressed during B-cell development, and is specifically located on plasma cells and MM cells in the bone marrow.87 Indatuximab ravtansine (BT062) comprises a mAb against CD138 conjugated to the cytotoxic maytansinoid DM4.88 As a monotherapy, indatuximab ravtansine treatment achieved at least PR in only 4% (1/27) of patients with RRMM, but was generally well tolerated.89 As a combination therapy with LEN–DEX, an ORR of 78% was attained and acceptable tolerability maintained in a moderately pre-treated RRMM population (median of three prior therapies).90 This trial is ongoing, although no future trials of indatuximab ravtansine have been announced. Elsewhere, a [213Bi] radiolabelled antibody against CD138 is in preclinical development.91
PD-1/PD-L1
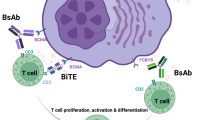
PD-1 is expressed on the surface of T and B cells, and inhibits T-cell activation and proliferation through its interaction with the PD-L1 ligand expressed on antigen-presenting cells.92, 93 PD-1/PD-L1 signaling is dysregulated in patients with MM, with PD-L1 expressed on MM cells, and PD-1 expressed on natural killer cells and upregulated on T cells.94 Thus, the PD-1/PD-L1 signaling axis provides a mechanism through which MM cells can interfere with the immune response and avoid death (Figure 3). The use of mAbs targeting PD-1/PD-L1 has been tested in the preclinical setting, with anti-myeloma activity demonstrated using an anti-PD-L1-based treatment protocol.95 The anti-PD-1 mAb most advanced in the clinic is nivolumab, although monotherapy with this agent has thus far achieved only disease stabilization (no objective responses reported) in patients with RRMM.96 However, nivolumab was shown to be tolerable, and its combination with the CTLA-4 mAb ipilimumab (activation of CTLA-4 dampens the host T-cell response97) or lirilumab is under assessment as part of the same phase I trial (NCT01592370). In addition, early-stage clinical trials of the anti-PD-1 antibodies pembrolizumab (MK-3475) and pidilizumab, typically in combination with IMiDs, are ongoing in patients with RRMM.
PD-1/PD-L1 mode of action. In normal subjects, active NK cells and T cells serve to destroy ‘unwanted’ cells. In patients with MM, PD-L1 is expressed on MM cells, and PD-1 by NK cells and T cells. PD-1/PD-L1 signaling in patients with MM inhibits the function of these immune cells, allowing MM to escape death. MM, multiple myeloma; NK, natural killer.
Other antigens
Immunophenotyping of MM cells has also revealed a number of other target antigens (Figure 1), and to date, mAbs to CD40, CD56, ICAM-1 and CXCR4 have been tested in clinical studies. Dacetuzumab and lucatumumab, mAbs to the tumor necrosis factor superfamily member CD40, had good tolerability but only weak efficacy as single agents98, 99 (Table 1). In addition, only modest ORR (39%) was attained when dacetuzumab was combined with LEN–DEX in patients with RRMM.100 Development of dacetuzumab has been discontinued, and no upcoming trials of lucatumumab have been reported in the MM setting.
Lorvotuzumab mertansine, a humanized mAb to CD56 conjugated to the cytotoxic maytansinoid derivative DM1, also displayed low activity as a monotherapy in patients with RRMM (ORR, 7%; Table 1).101 In combination with LEN–DEX, an ORR of 56% was achieved in patients with CD56+ RRMM, although this population had received only a median of two prior anti-myeloma therapies.102, 103 Dose-related neuropathy was observed in both the monotherapy and combination therapy studies,101, 102 and no new trials have been announced for lorvotuzumab mertansine.
Clinical data from a phase I trial of BI-505, a human mAb against ICAM-1, demonstrated that this agent can stabilize disease, although no objective responses were observed following 2 months’ treatment of patients with RRMM (Bioinvent press release). However, this agent was generally well tolerated and a phase II study of single-agent BI-505 in patients with smoldering myeloma is now recruiting (NCT01838369).
Similar to the above agents, the anti-CXCR4 mAb ulocuplumab is also under investigation in patients with RRMM, in combination with either LEN–DEX or BORT–DEX (NCT01359657). The ORR was 55% and 40% for the LEN–DEX and BORT–DEX groups, respectively, and an increase in circulating lymphocytes was observed after each ulocuplumab infusion at the two highest doses tested.104 Both combination regimens were generally well tolerated, with the MTD not reached during the dose escalation phase of this study.104
Conclusions
The introduction of PIs and IMiDs has improved outcomes for patients with MM, although these agents do not cure the disease and most patients will eventually relapse. As such, the need remains for newer agents with novel modes of action. Inspiration has been derived from knowledge of molecules expressed on the surface of myeloma cells, with a number of targets identified for mAb-based therapies. Of those agents which have reported clinical data, the anti-CD38 mAbs DARA and SAR650984 have shown robust single-agent activity, which has been enhanced through the addition of LEN–DEX, whereas the activity of other agents (for example, ELO) appears restricted to combination regimens.
The antibodies discussed here are generally well tolerated, and importantly most appear to have a safety profile that is distinct from current treatments. Combination therapy is thus a viable strategy, and in this regard LEN may have an inherent advantage due to its demonstrated positive effects on the immune responses through which a number of the mAbs operate. However, other combinations are under clinical testing, and the potential remains for combining the antibodies themselves providing that the modes of action are distinct. High response rates have been achieved in patients with RRMM with combination therapy, in some cases even in heavily pre-treated patients. However, these agents may also have a role in early-line therapy, with data from other novel agents suggesting increased ORR and deeper responses can be achieved in front-line therapy compared with treatment of RRMM.105, 106 As treatment paradigms shift, mAb therapy may also be utilized for patients with monoclonal gammopathy of undetermined significance or smoldering MM; toxicities associated with current regimens for symptomatic MM may deter their use in these patients, although a tolerable, efficacious mAb monotherapy would not face the same issues. Here, it is notable that single-agent clinical studies of both DARA and ELO in patients with smoldering MM have been announced. The development of mAbs thus represents an exciting new chapter in the battle against MM. Future data from larger studies will reveal how much of an impact these agents will make on long-term survival and the quality of life in patients with MM.
References
Kuehl WM, Bergsagel PL . Molecular pathogenesis of multiple myeloma and its premalignant precursor. J Clin Invest 2012; 122: 3456–3463.
National Cancer Institute. SEER database. Surveillance, Epidemiology, and End Results Program. 2014 [cited June 30]. Available from http://seer.cancer.gov/.
Becker N . Epidemiology of multiple myeloma. In: Moehler T, Goldschmidt H (eds). Multiple Myeloma: Recent Results in Cancer Research. Springer-Verlag: Berlin Heidelberg, Germany, 2011, 25–35.
Walker BA, Wardell CP, Melchor L, Brioli A, Johnson DC, Kaiser MF et al. Intraclonal heterogeneity is a critical early event in the development of myeloma and precedes the development of clinical symptoms. Leukemia 2014; 28: 384–390.
Kyle RA, Rajkumar SV . Criteria for diagnosis, staging, risk stratification and response assessment of multiple myeloma. Leukemia 2009; 23: 3–9.
Cömert M, Günes AE, Sahin F, Saydam G . Quality of life and supportive care in multiple myeloma. Turk J Haematol 2013; 30: 234–246.
Kumar SK, Rajkumar SV, Dispenzieri A, Lacy MQ, Hayman SR, Buadi FK et al. Improved survival in multiple myeloma and the impact of novel therapies. Blood 2008; 111: 2516–2520.
Rovira M, Rosiñol L, Fernández-Avilés F, Martinez C, Giné E, Esteve J et al. Is there a curative potential of autologous stem cell transplantation in multiple myeloma? Long-term results from a single-centre series. Bone Marrow Transplant 2009; 42: S147 (abstract P592).
Gahrton G, Svensson H, Cavo M, Apperly J, Bacigalupo A, Björkstrand B et al. Progress in allogenic bone marrow and peripheral blood stem cell transplantation for multiple myeloma: a comparison between transplants performed 1983-93 and 1994-8 at European Group for Blood and Marrow Transplantation centres. Br J Haematol 2001; 113: 209–216.
Wildes TM, Rosko A, Tuchman SA . Multiple myeloma in the older adult: better prospects, more challenges. J Clin Oncol 2014; 32: 2531–2540.
Kumar SK, Lee JH, Lahuerta JJ, Morgan G, Richardson PG, Crowley J et al. Risk of progression and survival in multiple myeloma relapsing after therapy with IMiDs and bortezomib: a multicenter international myeloma working group study. Leukemia 2012; 26: 149–157.
Zangari M, Tricot G, Polavaram L, Zhan F, Finlayson A, Knight R et al. Survival effect of venous thromboembolism in patients with multiple myeloma treated with lenalidomide and high-dose dexamethasone. J Clin Oncol 2010; 28: 132–135.
Mateos MV, San Miguel JF . Safety and efficacy of subcutaneous formulation of bortezomib versus the conventional intravenous formulation in multiple myeloma. Ther Adv Hematol 2012; 3: 117–124.
Glasmacher A, Hahn C, Hoffmann F, Naumann R, Goldschmidt H, von Lilienfeld-Toal M et al. A systematic review of phase-II trials of thalidomide monotherapy in patients with relapsed or refractory multiple myeloma. Br J Haematol 2006; 132: 584–593.
Dimopoulos MA, Richardson PG, Moreau P, Anderson KC . Current treatment landscape for relapsed and/or refractory multiple myeloma. Nat Rev Clin Oncol 2015; 12: 42–54.
Scott AM, Wolchok JD, Old LJ . Antibody therapy of cancer. Nat Rev Cancer 2012; 12: 278–287.
Malavasi F, Deaglio S, Funaro A, Ferrero E, Horenstein AL, Ortolan E et al. Evolution and function of the ADP ribosyl cyclase/CD38 gene family in physiology and pathology. Physiol Rev 2008; 88: 841–886.
Chillemi A, Zaccarello G, Quarona V, Ferracin M, Ghimenti C, Massaia M et al. Anti-CD38 antibody therapy: windows of opportunity yielded by the functional characteristics of the target molecule. Mol Med 2013; 19: 99–108.
Quarona V, Ferri V, Chillemi A, Bolzoni M, Mancini C, Zaccarello G et al. Unraveling the contribution of ectoenzymes to myeloma life and survival in the bone marrow niche. Ann N Y Acad Sci 2015; 1335: 10–22.
Vaisitti T, Audrito V, Serra S, Buonincontri R, Sociali G, Mannino E et al. The enzymatic activities of CD38 enhance CLL growth and trafficking: implications for therapeutic targeting. Leukemia 2015; 29: 356–368.
Lin P, Owens R, Tricot G, Wilson CS . Flow cytometric immunophenotypic analysis of 306 cases of multiple myeloma. Am J Clin Pathol 2004; 121: 482–488.
de Weers M, Tai YT, van der Veer MS, Bakker JM, Vink T, Jacobs DC et al. Daratumumab, a novel therapeutic human CD38 monoclonal antibody, induces killing of multiple myeloma and other hematological tumors. J Immunol 2011; 186: 1840–1848.
Overdijk MB, Verploegen S, Marjin B, van Egmond M, Groen WJ, Martens ACM et al. Phagocytosis in a mechanism of action for daratumumab. 54th American Society of Hematology (ASH) Annual Meeting. Atlanta, GA, USA, 8–11 December 2012 (abstract 4054).
Groen RW, van der Veer M, Hofhuis FM, van Kessel V, de Weers M, Parren PWHI et al. In vitro and in vivo efficacy of CD38 directed therapy with daratumumab in the treatment of multiple myeloma. 52nd American Society of Hematology (ASH) Annual Meeting. Orlando, FL, USA, 4–7 December 2010 (abstract 3058).
Jensen JHM, Boross P, Overdijk MB, Lammerts van Bueren JJ, Parren PWHI, Leusen JHW . Daratumumab, a human CD38 antibody induces apoptosis of myeloma tumor cells via Fc receptor-medicated crosslinking. 54th American Society of Hematology (ASH) Annual Meeting. Atlanta, GA, USA, 8–11 December 2012 (abstract 2974).
Mutis T, de Weers M, van der Veer MS, Kessel BV, Bakker JM, Wittebol S et al. The potential of the human CD38-specific antibody daratumumab to improve the antimyeloma effect of novel multidrug therapies including patients refractory to lenalidomide or bortezomib. American Society of Clinical Oncology (ASCO) Annual Meeting. Chicago, IL, USA, 3–7 June 2011 (abstract e18571).
van der Veer MS, de Weers M, van Kessel B, Bakker JM, Wittebol S, Parren PW et al. The therapeutic human CD38 antibody daratumumab improves the anti-myeloma effect of newly emerging multi-drug therapies. Blood Cancer J 2011; 1: e41.
Laubach JP, Richardson PG . CD38-targeted immunochemotherapy in refractory multiple myeloma: a new horizon. Clin Cancer Res 2015; 21: 2660–2662.
Lokhorst H, Laubach J, Nahi H, Plesner T, Gimsing P, Hansson M et al. Dose-dependent efficacy of daratumumab (DARA) as monotherapy in patients with relapsed or refractory multiple myeloma (RRMM). American Society of Clinical Oncology (ASCO) Annual Meeting. Chicago, IL, USA, 30 May–3 June 2014 (presentation no. 8513).
Laubach JP, Tai YT, Richardson PG, Anderson KC . Daratumumab granted breakthrough drug status. Expert Opin Investig Drugs 2014; 23: 445–452.
Lonial S, Weiss B, Usmani S, Singhal S, Chari A, Bahlis N et al. Phase 2 study of daratumumab (DARA) in patients with 3 lines of prior therapy or double refractory multiple myeloma: 54767414MMY2002 (Sirius). American Society of Clinical Oncology (ASCO) Annual Meeting. Chicago, IL, USA, 29 May–2 June 2015 (oral presentation LBA8512).
Plesner T, Arkenau HT, Lokhorst HM, Gimsing P, Krejcik J, Lemech C et al. Safety and efficacy of Daratumumab with Lenalidomide and Dexamethasone in relapsed or relapsed, refractory multiple myeloma. 56th American Society of Hematology (ASH) Annual Meeting and Exposition. San Francisco, CA, USA, 6–9 December 2014 (oral presentation 84).
Mateos MV, Moreau P, Comenzo R, Bladé J, Benboubker L, de la Rubia J et al. An ppen-label, multicenter, phase 1b study of daratumumab in combination with oomalidomide-dexamethasone and with backbone regimens in patients with multiple myeloma. 20th Congress of European Hematology Association (EHA). Vienna, Austria, 11–14 June 2015 (abstract P275).
Lokhorst H, Plesner T, Gimsing P, Nahi H, Lisby S, Richardson P . DARATUMUMAB, a CD38 monoclonal antibody study in advanced multiple myeloma – an open-label, dose escalation followed by open-label extension in a single-arm Phase I/II study. 18th Congress of the European Hematology Association. Stockholm, Sweden, 13–16 June 2013 (presentation no. S576).
Plesner T, Arkenau H-T, Lokhorst HM, Gimsing P, Krejcik J, Lemech CR et al. Safety and efficacy of daratumumab with lenalidomide and dexamethasone in relapsed or refractory multiple myeloma. American Society of Clinical Oncology (ASCO) Annual Meeting. Chicago, IL, USA, 30 May–3 June 2014 (poster).
Deckert J, Wetzel MC, Bartle LM, Skaletskaya A, Goldmacher VS, Vallée F et al. SAR650984, a novel humanized CD38-targeting antibody, demonstrates potent antitumor activity in models of multiple myeloma and other CD38+ hematologic malignancies. Clin Cancer Res 2014; 20: 4574–4583.
Genmab A/S. Webcast post-ASH Investor Seminar. Genmab A/S. 2013 [cited June 30]. Available from http://cache.merchantcantos.com/webcast/webcaster/4000/7464/16533/28919/Lobby/default.htm.
Hann B, Cai T, Wang D, Hom Y-K, Aftab BT, Martin T et al. SAR650984, an anti-CD38 antibody, shows anti-tumor activity in a preclinical model of multiple myeloma. American Association for Cancer Research (AACR) Annual Meeting. Washington, DC, USA, 6–10 April 2013 (abstract 5469).
Lejeune P, Blanc V, Courta J, Egile C, Vrignaud P, Deckert J et al. In vivo therapeutic synergy of SAR650984, a humanized anti-CD38 antibody, in combination with melphalan in a multiple myeloma xenograft. 100th Annual Meeting AACR. Denver, CO, USA, 18–22 April 2009 (abstract 2797).
Wetzel M-C, Nicolazzi C, Vallée F, Deckert J, Dumontet C, Plesa A et al. SAR650984: characterization of a potent phase I humanized anti-CD38 antibody for the treatment of multiple myeloma and other hematologic malignancies. Cancer Res 2013; 73: (abstract 4735).
Martin T, Strickland S, Glenn M, Hsu K, Charpentier E, Mikhael J . A phase I trial of SAR650984, a CD38 monoclonal antibody, in relapsed or refractory multiple myeloma. American Society of Clinical Oncology (ASCO) Annual Meeting. Chicago, IL, USA, 30 May–3 June 2014 (poster no. 8532).
Martin TG, Baz R, Benson DM, Lendvai N, Campana F, Charpentier E et al. A Phase 1b dose escalation trial of SAR650984 (anti-CD-38 mAb) in combination with Lenalidomide and Dexamethasone in relapsed/refractory multiple myeloma. 56th American Society of Hematology (ASH) Annual Meeting and Exposition. San Francisco, CA, USA, 6–9 December 2014 oral presentation 83.
Martin T, Baz R, Benson D, Lendvai N, Hsu K, Charpentier E et al. A phase Ib dose escalation trial of SAR650984 (anti-CD-38 mAb) in combination with lenalidomide and dexamethasone in relapsed/refractory multiple myeloma. American Society of Clinical Oncology (ASCO) Annual Meeting. Chicago, IL, USA, 30 May–3 June 2014 (abstract 8512).
Endell J, Samuelsson C, Boxhammer R, Strauβ S, Steidl S . Effect of MOR202, a human CD38 antibody, in combination with lenalidomide and bortezomib on bone lysis and tumor load in a physiological model of myeloma. American Society of Clinical Oncology (ASCO) Annual Meeting, Chicago, IL, USA, 3–6 June 2011 (poster no. 8078).
Boxhammer R, Wurzenberger C, Ness D, Steidl S, Endell J . The activity of MOR202, a fully human anti-CD38 antibody, is complemented by ADCP and is synergistically enhanced by lenalidomide in vitro and in vivo. 54th American Society of Hematology (ASH) Annual Meeting. Atlanta, GA, USA, 8–11 December 2012 (poster no. 4018).
Boxhammer R, Steidl S, Endell J . Effect of IMiD compounds on CD38 expression on multiple myeloma cells: MOR202, a human CD38 antibody in combination with pomalidomide. American Society of Clinical Oncology (ASCO) Annual Meeting. Chicago, IL, USA, 29 May–2 June 2015 (abstract 8588).
Endell J . From library to bedside: potential of the anti-CD38 antibody MOR202 in combination therapy of multiple myeloma. Bio M. 2013; [cited June 30]. Available from http://www.m4.de/uploads/media/m4_Seminar_12112013_Endell_web.pdf.
Raab MS, Goldschmidt H, Agis H, Blau I, Einsele H, Englehardt MM et al. A phase I/IIa study of the human anti-CD38 antibody MOR202 (MOR03087) in relapsed or refractory multiple myeloma (rrMM). American Society of Clinical Oncology (ASCO) Annual Meeting, Chicago, IL, USA, 29 May–2 June 2015 (abstract 8574).
Lammerts van Bueren J, Jakobs D, Kaldenhoven N, Roza M, Hiddingh S, Meesters J et al. Direct in vitro comparison of Daratumumab with surrogate analogs of CD38 antibodies MOR03087, SAR650984 and Ab79. 56th American Society of Hematology (ASH) Annual Meeting and Exposition. San Francisco, CA, USA, 6–9 December 2014 (abstract 3474).
Palumbo A, Rajkumar SV, San Miguel JF, Larocca A, Niesvizky R, Morgan G et al. International Myeloma Working Group consensus statement for the management, treatment, and supportive care of patients with myeloma not eligible for standard autologous stem-cell transplantation. J Clin Oncol 2014; 32: 587–600.
McCudden P, Axel A, Slaets D, Frans S, Bald J, Schecter JM et al. Assessing clinical response in multiple myeloma (MM) patients treated with monoclonal antibodies (mAbs): validation of a daratumumab IFE reflex assay to distinguish malignant M-protein from therapeutic antibody. American Society of Clinical Oncology (ASCO) Annual Meeting, Chicago, IL, USA, 29 May–2 June 2015 (abstract 8590).
Hsi ED, Steinle R, Balasa B, Szmania S, Draksharapu A, Shum BP et al. CS1, a potential new therapeutic antibody target for the treatment of multiple myeloma. Clin Cancer Res 2008; 14: 2775–2784.
Veillette A, Guo H . CS1, a SLAM family receptor involved in immune regulation, is a therapeutic target in multiple myeloma. Crit Rev Oncol Hematol 2013; 88: 168–177.
Tai Y-T, Soydan E, Song W, Fulciniti M, Kim K, Hong F et al. Retraction. CS1 promotes multiple myeloma cell adhesion, clonogenic growth, and tumorigenicity via c-maf-mediated interactions with bone marrow stromal cells. Blood 2010; 115: 2983.
Tai YT, Dillon M, Song W, Leiba M, Li XF, Burger P et al. Anti-CS1 humanized monoclonal antibody HuLuc63 inhibits myeloma cell adhesion and induces antibody-dependent cellular cytotoxicity in the bone marrow milieu. Blood 2008; 112: 1329–1337.
Collins SM, Bakan CE, Swartzel GD, Hofmeister CC, Efebera YA, Kwon H et al. Elotuzumab directly enhances NK cell cytotoxicity against myeloma via CS1 ligation: evidence for augmented NK cell function complementing ADCC. Cancer Immunol Immunother 2013; 62: 1841–1849.
Zonder JA, Mohrbacher AF, Singhal S, van Rhee F, Bensinger WI, Ding H et al. A phase 1, multicenter, open-label, dose escalation study of elotuzumab in patients with advanced multiple myeloma. Blood 2012; 120: 552–559.
Jakubowiak AJ, Benson DM, Bensinger W, Siegel DS, Zimmerman TM, Mohrbacher A et al. Phase I trial of anti-CS1 monoclonal antibody elotuzumab in combination with bortezomib in the treatment of relapsed/refractory multiple myeloma. J Clin Oncol 2012; 30: 1960–1965.
Jakubowiak AJ, Offidani M, Pegourie B, de la Rubia J, Garderet L, Laribi K et al. A randomized phase II study of bortezomib (Btz)/dexamethasone (dex) with or without elotuzumab (Elo) in patients (pts) with relapsed/refractory multiple myeloma (RRMM). American Society of Clinical Oncology (ASCO) Annual Meeting. Chicago, IL, USA, 29 May–2 June 2015 (abstract 8573).
Mateos MV, Granell M, Rocafiguera AO, Lopez JM, Bladé J, Hernandez MT et al A phase II single-arm safety study of elotuzumab in combination with thalidomide and low dose dexamethasone in patients with relapsed and/or refractory multiple myeloma. 19th Congress of European Haematology Association, Milan, Italy, 12–15 June 2014 (abstract P959).
Lonial S, Vij R, Harousseau JL, Facon T, Moreau P, Mazumder A et al. Elotuzumab in combination with lenalidomide and low-dose dexamethasone in relapsed or refractory multiple myeloma. J Clin Oncol 2012; 30: 1953–1959.
Lonial S, Jagannath S, Moreau P, Jakubowiak AJ, Raab MS, Facon T et al. Phase (Ph) I/II study of elotuzumab (Elo) plus lenalidomide/dexamethasone (Len/dex) in relapsed/refractory multiple myeloma (RR MM): updated Ph II results and Ph I/II long-term safety. American Society of Clinical Oncology (ASCO) Annual Meeting. Chicago, IL, USA, 31 May–4 June 2013 (presentation).
Richardson PG, Jagannath S, Moreau P, Jakubowiak A, Raab MS, Facon T et al. A Phase 2 study of elotuzumab in combination with lenalidomide and low-dose dexamethasone in patients with relapsed/refractory multiple myeloma: updated results. 54th American Society of Hematology (ASH) Annual Meeting. Atlanta, GA, USA, 8–11 December 2012 (presentation no. 202).
Lonial S, Dimopoulos M, Palumbo A, White D, Grosicki S, Spicka I et al. ELOQUENT-2: a phase 3, randomized, ppen-label study of lenalidomide/dexamethasone with/without elotuzumab in patients with relapsed/refractory multiple myeloma. American Society of Clinical Oncology (ASCO) Annual Meeting. Chicago, IL, USA, 29 May–2 June 2015 (oral presentation 8508).
Usmani SZ, Sexton R, Ailawadhi S, Shah JJ, Callander NS, Zimmerman T et al. SWOG 1211: initial report on Phase I trial of RVD-Elotuzumab for newly diagnosed high risk multiple myeloma (HRMM). 56th American Society of Hematology (ASH) Annual Meeting and Exposition. San Francisco, CA, USA, 6–9 December 2014 abstract 4762.
Benson DM Jr, Bakan CE, Zhang S, Collins SM, Liang J, Srivastava S et al. IPH2101, a novel anti-inhibitory KIR antibody, and lenalidomide combine to enhance the natural killer cell versus multiple myeloma effect. Blood 2011; 118: 6387–6391.
Kohrt HE, Godwin JE, Lossos IS, Williams ME, Timmerman J, Link BK et al. A phase 1b, open-label, multicenter study of urelumab (BMS-663513) in combination with rituximab in subjects with relapsed/refractory B-cell malignancies. American Society of Clinical Oncology (ASCO) Annual Meeeting. Chicago, IL, USA, 31 May–4 June 2013 (abstract TPS3108).
Kishimoto T . The biology of interleukin-6. Blood 1989; 74: 1–10.
Chauhan D, Uchiyama H, Akbarali Y, Urashima M, Yamamoto K, Libermann TA et al. Multiple myeloma cell adhesion-induced interleukin-6 expression in bone marrow stromal cells involves activation of NF-kappa B. Blood 1996; 87: 1104–1112.
Hilbert DM, Kopf M, Mock BA, Köhler G, Rudikoff S . Interleukin 6 is essential for in vivo development of B lineage neoplasms. J Exp Med 1995; 182: 243–248.
Hata H, Xiao H, Petrucci MT, Woodliff J, Chang R, Epstein J . Interleukin-6 gene expression in multiple myeloma: a characteristic of immature tumor cells. Blood 1993; 81: 3357–3364.
Voorhees PM, Chen Q, Small GW, Kuhn DJ, Hunsucker SA, Nemeth JA et al. Targeted inhibition of interleukin-6 with CNTO 328 sensitizes pre-clinical models of multiple myeloma to dexamethasone-mediated cell death. Br J Haematol 2009; 145: 481–490.
Voorhees PM, Chen Q, Kuhn DJ, Small GW, Hunsucker SA, Strader JS et al. Inhibition of interleukin-6 signaling with CNTO 328 enhances the activity of bortezomib in preclinical models of multiple myeloma. Clin Cancer Res 2007; 13: 6469–6478.
Hunsucker SA, Magarotto V, Kuhn DJ, Kornblau SM, Wang M, Weber DM et al. Blockade of interleukin-6 signalling with siltuximab enhances melphalan cytotoxicity in preclinical models of multiple myeloma. Br J Haematol 2011; 152: 579–592.
Voorhees PM, Manges RF, Sonneveld P, Jagannath S, Somlo G, Krishnan A et al. A phase 2 multicentre study of siltuximab, an anti-interleukin-6 monoclonal antibody, in patients with relapsed or refractory multiple myeloma. Br J Haematol 2013; 161: 357–366.
Orlowski RZ, Gercheva L, Williams C, Sutherland H, Robak T, Masszi T et al. A phase 2, randomized, double-blind, placebo-controlled study of siltuximab (anti-IL-6 mAb) and bortezomib versus bortezomib alone in patients with relapsed or refractory multiple myeloma. Am J Hematol 2015; 90: 42–49.
San-Miguel J, Bladé J, Shpilberg O, Grosicki S, Maloisel F, Min CK et al. Phase 2 randomized study of bortezomib-melphalan-prednisone with or without siltuximab (anti-IL-6) in multiple myeloma. Blood 2014; 123: 4136–4142.
Thomas SK, Suvorov A, Noens L, Rukavitsin O, Fay J, Wu KL et al. Evaluation of the QTc prolongation potential of a monoclonal antibody, siltuximab, in patients with monoclonal gammopathy of undetermined significance, smoldering multiple myeloma, or low-volume multiple myeloma. Cancer Chemother Pharmacol 2014; 73: 35–42.
Neri P, Kumar S, Fulciniti MT, Vallet S, Chhetri S, Mukherjee S et al. Neutralizing B-cell activating factor antibody improves survival and inhibits osteoclastogenesis in a severe combined immunodeficient human multiple myeloma model. Clin Cancer Res 2007; 13: 5903–5909.
Moreaux J, Legouffe E, Jourdan E, Quittet P, Rème T, Lugagne C et al. BAFF and APRIL protect myeloma cells from apoptosis induced by interleukin 6 deprivation and dexamethasone. Blood 2004; 103: 3148–3157.
Raje NS, Hohl RJ, Faber EA, Richardson PGG, Forero-Torres A, Schiller GJ et al. Phase I study of LY2127399, a human anti-BAFF antibody, and bortezomib in patients with previously treated multiple myeloma. American Society of Clinical Oncology (ASCO) Annual Meeeting. Chicago, IL, USA, 3–7 June 2011 (abstract 8012).
Raje NS, Faber EA, Richardson PG, Schiller GJ, Hohl RJ, Cohen AD et al. Phase 1 study of tabalumab, a human anti-BAFF antibody and bortezomib in patients with previously-treated multiple myeloma. 54th American Society of Hematology (ASH) Annual Meeting, Atlanta, GA, USA, 8–11 December 2012 oral presentation 447.
Burton JD, Ely S, Reddy PK, Stein R, Gold DV, Cardillo TM et al. CD74 is expressed by multiple myeloma and is a promising target for therapy. Clin Cancer Res 2004; 10: 6606–6611.
Stein R, Mattes MJ, Cardillo TM, Hansen HJ, Chang CH, Burton J et al. CD74: a new candidate target for the immunotherapy of B-cell neoplasms. Clin Cancer Res 2007; 13: 5556s–5563s.
Stein R, Smith MR, Chen S, Zalath M, Goldenberg DM . Combining milatuzumab with bortezomib, doxorubicin, or dexamethasone improves responses in multiple myeloma cell lines. Clin Cancer Res 2009; 15: 2808–2817.
Kaufman JL, Niesvizky R, Stadtmauer EA, Chanan-Khan A, Siegel D, Horne H et al. Phase I, multicentre, dose-escalation trial of monotherapy with milatuzumab (humanized anti-CD74 monoclonal antibody) in relapsed or refractory multiple myeloma. Br J Haematol 2013; 163: 478–486.
Calame KL . Plasma cells: finding new light at the end of B cell development. Nat Immunol 2001; 2: 1103–1108.
Ikeda H, Hideshima T, Fulciniti M, Lutz RJ, Yasui H, Okawa Y et al. The monoclonal antibody nBT062 conjugated to cytotoxic Maytansinoids has selective cytotoxicity against CD138-positive multiple myeloma cells in vitro and in vivo. Clin Cancer Res 2009; 15: 4028–4037.
Heffner LT, Jagannath S, Zimmerman TM, Lee KP, Rosenblatt J, Lonial S et al. BT062, an antibody-drug conjugate directed against CD138, given weekly for 3 weeks in each 4 week cycle: safety and further evidence of clinical activity. 54th American Society of Hematology (ASH) Annual Meeting. Atlanta, GA, USA, 8–11 December 2012 (poster presentation 4042).
Kelly KR, Chanan-Khan A, Heffner LT, Somio G, Siegel DS, Zimmerman T et al. Indatuximab ravtansine (BT062) in combination with Lenalidomide and low-dose Dexamethasone in patients with relapsed and/or refractory multiple myeloma: clinical activity in patients already exposed to Lenalidomide and Bortezomib. 56th American Society of Hematology (ASH) Annual Meeting and Exposition. San Francisco, CA, USA, 6–9 December 2014 poster presentation 4736.
Gouard S, Pallardy A, Gaschet J, Faivre-Chauvet A, Bruchertseifer F, Morgenstern A et al. Comparative analysis of multiple myeloma treatment by CD138 antigen targeting with bismuth-213 and Melphalan chemotherapy. Nucl Med Biol 2014; 41: e30–e35.
Freeman GJ, Long AJ, Iwai Y, Bourque K, Chernova T, Nishimura H et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med 2000; 192: 1027–1034.
Carter L, Fouser LA, Jussif J, Fitz L, Deng B, Wood CR et al. PD-1:PD-L inhibitory pathway affects both CD4(+) and CD8(+) T cells and is overcome by IL-2. Eur J Immunol 2002; 32: 634–643.
Atanackovic D, Luetkens T, Kröger N . Coinhibitory molecule PD-1 as a potential target for the immunotherapy of multiple myeloma. Leukemia 2014; 28: 993–1000.
Kearl TJ, Jing W, Gershan JA, Johnson BD . Programmed death receptor-1/programmed death receptor ligand-1 blockade after transient lymphodepletion to treat myeloma. J Immunol 2013; 190: 5620–5628.
Lesokhin AM, Ansell SM, Armand P, Scott EC, Halwani A, Gutierrez M et al. Preliminary results of a Phase I study of nivolumab (BMS-936558) in patients with relapsed or refractory lymphoid malignancies. 56th American Society of Hematology (ASH) Annual Meeting and Exposition. San Francisco, CA, USA, 6–9 December 2014 abstract 291.
Walunas TL, Lenschow DJ, Bakker CY, Linsley PS, Freeman GJ, Green JM et al. CTLA-4 can function as a negative regulator of T cell activation. Immunity 1994; 1: 405–413.
Hussein M, Berenson JR, Niesvizky R, Munshi N, Matous J, Sobecks R et al. A phase I multidose study of dacetuzumab (SGN-40; humanized anti-CD40 monoclonal antibody) in patients with multiple myeloma. Haematologica 2010; 95: 845–848.
Bensinger W, Maziarz RT, Jagannath S, Spencer A, Durrant S, Becker PS et al. A phase 1 study of lucatumumab, a fully human anti-CD40 antagonist monoclonal antibody administered intravenously to patients with relapsed or refractory multiple myeloma. Br J Haematol 2012; 159: 58–66.
Agura E, Niesvizky R, Matous J, Munshi N, Hussein M, Parameswaran RV et al. Dacetuzumab (SGN-40), lenalidomide, and weekly dexamethasone in relapsed or refractory multiple myeloma: multiple responses observed in a Phase 1b study. 51st American Society of Hematology (ASH) Annual Meeting. Atlanta, GA, USA, 5–9 December 2009 abstract 2870.
Chanan-Khan A, Wolf JL, Garcia J, Gharibo M, Jagannath S, Manfredi D et al. Efficacy analysis from Phase I study of lorvotuzumab mertansine (IMGN901), used as monotherapy, in patients with heavily pre-treated CD56-positive multiple myeloma - A preliminary efficacy analysis. 52nd American Society of Hematology (ASH) Annual Meeting. Orlando, FL, USA, 4–7 December 2010 abstract 1962.
Berdeja JG, Hernandez-Ilizaliturri F, Chanan-Khan A, Patel M, Kelly KR, Running KL et al. Phase I study of lorvotuzumab mertansine (LM, IMGN901) in combination with lenalidomide (Len) and dexamethasone (Dex) in patients with CD56-positive relapsed or relapsed/refractory multiple myeloma (MM). 54th American Society of Hematology (ASH) Annual Meeting. Atlanta, GA, USA, 8–11 December 2012 oral presentation 728.
Berdeja JG . Lorvotuzumab mertansine: antibody-drug-conjugate for CD56+ multiple myeloma. Front Biosci (Landmark Ed) 2014; 19: 163–170.
Ghobrial IM, Perez M, Baz R, Richardson PG, Anderson KC, Sabbatini P et al. Phase 1b study of the novel anti-CXCR4 antibody ulocuplumab (BMS-936564) in combination with Lenalidomide plus low-dose Dexamethasone, or with Bortezomib plus Dexamethasone in subjects with relapsed or refractory multiple myeloma. 56th American Society of Hematology (ASH) Annual Meeting and Exposition. San Francisco, CA, USA, 6–9 December 2014 abstract 3483.
Niesvizky R, Martin TG III, Bensinger WI, Alsina M, Siegel DS, Kunkel LA et al. Phase Ib dose-escalation study (PX-171-006) of carfilzomib, lenalidomide, and low-dose dexamethasone in relapsed or progressive multiple myeloma. Clin Cancer Res 2013; 19: 2248–2256.
Jakubowiak AJ, Dytfeld D, Griffith KA, Lebovic D, Vesole DH, Jagannath S et al. A phase 1/2 study of carfilzomib in combination with lenalidomide and low-dose dexamethasone as a frontline treatment for multiple myeloma. Blood 2012; 120: 1801–1809.
Donato F, Gay F, Bringhen S, Troia R, Palumbo A . Monoclonal antibodies currently in Phase II and III trials for multiple myeloma. Expert Opin Biol Ther 2014; 14: 1127–1144.
Atanackovic D, Panse J, Hildebrandt Y, Jadczak A, Kobold S, Cao Y et al. Surface molecule CD229 as a novel target for the diagnosis and treatment of multiple myeloma. Haematologica 2011; 96: 1512–1520.
Mateo G, Montalbán MA, Vidriales MB, Lahuerta JJ, Mateos MV, Gutiérrez N et al. Prognostic value of immunophenotyping in multiple myeloma: a study by the PETHEMA/GEM cooperative study groups on patients uniformly treated with high-dose therapy. J Clin Oncol 2008; 26: 2737–2744.
Yang J, Yi Q . Therapeutic monoclonal antibodies for multiple myeloma: an update and future perspectives. Am J Blood Res 2011; 1: 22–33.
Durie BG, Harousseau JL, Miguel JS, Bladé J, Barlogie B, Anderson K et al. International uniform response criteria for multiple myeloma. Leukemia 2006; 20: 1467–1473; erratum in Leukemia 2007; 21: 1134.
Bladé J, Samson D, Reece D, Apperley J, Björkstrand B, Gahrton G et al. Criteria for evaluating disease response and progression in patients with multiple myeloma treated by high-dose therapy and haemopoietic stem cell transplantation. Myeloma Subcommittee of the EBMT. European Group for Blood and Marrow Transplant. Br J Haematol 1998; 102: 1115–1123.
Acknowledgements
The authors received editorial assistance from Neil Harrison of Adelphi Communications Ltd, funded by Sanofi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
SL has acted as a consultant for Millennium, Celgene, Novartis, Bristol-Myers Squibb, Onyx and Sanofi. AP has acted as a consultant and received honoraria from Amgen, Bristol-Myers Squibb, Genmab A/S, Celgene, Janssen-Cilag, Millennium and Onyx. He has also received honoraria from Novartis and Sanofi. JSM has served on advisory committees for Millennium, Celgene, Novartis, Onyx, Janssen, Bristol-Myers Squibb, Merck Sharpe & Dohme and Sanofi. The remaining author declares no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Lonial, S., Durie, B., Palumbo, A. et al. Monoclonal antibodies in the treatment of multiple myeloma: current status and future perspectives. Leukemia 30, 526–535 (2016). https://doi.org/10.1038/leu.2015.223
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2015.223
This article is cited by
-
The role of minimal residual disease and serum free light chain ratio in the management of multiple myeloma
Discover Oncology (2024)
-
Dynamic CD138 surface expression regulates switch between myeloma growth and dissemination
Leukemia (2020)
-
Phase I trial of isatuximab monotherapy in the treatment of refractory multiple myeloma
Blood Cancer Journal (2019)
-
A preliminary study of the anti-κ myeloma antigen monoclonal antibody KappaMab (MDX-1097) in pretreated patients with κ-restricted multiple myeloma
Blood Cancer Journal (2019)
-
Navigating the treatment landscape in multiple myeloma: which combinations to use and when?
Annals of Hematology (2019)