Abstract
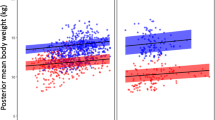
The revised European Directive 2010/63/EU on the protection of animals used for scientific purposes encourages the scientific community to consider the feasibility of transitioning from the use of wild-caught breeders toward the exclusive use of captive-born breeders in non-human primate breeding centers. Little is known about how such a transition will affect animal health, productivity and accompanying husbandry practices. These concerns are important both for the efficient operation of a breeding facility and for the optimal welfare of the animals in its care. The authors analyzed records of wild-caught and captive-born female cynomolgus macaques (Macaca fascicularis) used for breeding at a facility between 2006 and 2011. Productivity was measured by proportional birth rate and interbirth interval; perinatal mortality was quantified by cause and type of death; and body condition was estimated using recorded visual assessments. Captive-born breeders generally showed lower productivity, higher perinatal mortality and poorer body condition than wild-caught breeders in the same husbandry conditions. These findings indicate that facilities transitioning to the use of captive-born macaques in breeding might need to explore new and revised husbandry strategies that address potential developmental and behavioral differences of captive-born breeders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Scientific Committee on Health and Environmental Risks. SCHER Scientific Opinion on the Need for Non-human Primates in Biomedical Research, Production and Testing of Products and Devices (13 January 2009).
Hartung, T. Comparative analysis of the revised Directive 2010/63/EU for the protection of laboratory animals with its predecessor 86/609/EEC—a t4 Report. ALTEX 27, 285–303 (2010).
Ha, J.C., Robinette, R.L. & Sackett, G.P. Social housing and pregnancy outcome in captive pigtailed macaques. Am. J. Primatol. 47, 153–163 (1999).
Houghton, P. Virus-free African cynomolgus macaques. Lab Anim. (NY) 17, 36–40 (1988).
Gagliardi, C., Liukkonen, J.R., Phillippi-Falkenstein, K.M., Harrison, R.M. & Kubisch, H.M. Age as a determinant of reproductive success among captive female rhesus macaques (Macaca mulatta). Reproduction 133, 819–826 (2007).
Koyama, N., Takahata, Y., Huffman, M.A., Norikoshi, K. & Suzuki, H. Reproductive parameters of female Japanese macaques: Thirty years data from the Arashiyama troops, Japan. Primates 33, 33–47 (1992).
Williams, R.F. The interbirth interval in primates: effects of pregnancy and nursing in Primates: Proceedings in Life Sciences (Springer, New York, 1986).
Silk, J.B. Sources of variation in interbirth intervals among captive bonnet macaques (Macaca radiata). Am. J. Phys. Anthropol. 82, 213–230 (1990).
Tsuchida, J., Yoshida, T., Sankai, T. & Yasutomi, Y. Maternal behavior of laboratory-born, individually reared long-tailed macaques (Macaca fascicularis). J. Am. Assoc. Lab. Anim. Sci. 47, 29–34 (2008).
Nakamichi, M., Minami, T. & Cho, F. Comparison between wild-born mother-female infant interactions and laboratory-born mother-female infant interactions during the first 14 weeks after birth in individually caged cynomolgus macaques. Primates 37, 155–166 (1996).
Camus, S.M. et al. Birth origin differentially affects depressive-like behaviours: are captive-born cynomolgus monkeys more vulnerable to depression than their wild-born counterparts? PLoS ONE 8, e67711 (2013).
Shively, C.A. & Wallace, J.M. Social status, social stress and fat distribution in primates. Psychosomatic Med. 61, 107 (1999).
Bauer, S.A., Arndt, T.P., Leslie, K.E., Pearl, D.L. & Turner, P.V. Obesity in rhesus and cynomolgus macaques: a comparative review of the condition and its implications for research. Comp. Med. 61, 514–526 (2011).
Solanki, G.S. & Zothansiama . Infanticide in captive stump-tailed macaques (Macaca arctoides) is in accordance with the sexual selection hypothesis. Curr. Sci. 104, 1081–1083 (2013).
Soltis, J., Thomsen, R., Matsubayashi, K. & Takenaka, O. Infanticide by resident males and female counter-strategies in wild Japanese macaques (Macaca fuscata). Behav. Ecol. Sociobiol. 48, 195–202 (2000).
Maestripieri, D. Infant abuse associated with psychosocial stress in a group-living pigtail macaque (Macaca nemestrina) mother. Am. J. Primatol. 32, 41–49 (1994).
Maestripieri, D. & Carroll, K.A. Causes and consequences of infant abuse and neglect in monkeys. Aggression Violent Behav. 5, 245–254 (2000).
Maestripieri, D., Lindell, S.G., Ayala, A., Gold, P.W. & Higley, J.D. Neurobiological characteristics of rhesus macaque abusive mothers and their relation to social and maternal behavior. Neurosci. Biobehav. Rev. 29, 51–57 (2005).
De Waal, F.B. & Bonnie, K.E. In tune with others: the social side of primate culture. in The Question of Animal Culture (eds. Laland, K.N. & Galef, B.G.) 19–39 (Harvard University Press, Cambridge, MA, 2009).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Levallois, L., de Marigny, S. Reproductive success of wild-caught and captive-bred cynomolgus macaques at a breeding facility. Lab Anim 44, 387–393 (2015). https://doi.org/10.1038/laban.733
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/laban.733
This article is cited by
-
A meta-analysis of birth-origin effects on reproduction in diverse captive environments
Nature Communications (2018)