Abstract
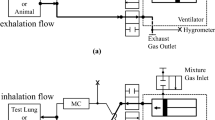
Establishing a program to monitor waste anesthetic gas (WAG) in order to limit personnel exposure requires measuring the levels of WAG emitted and determining the effectiveness of scavenging methods to reduce such levels. In this study, the authors used infrared spectroscopy to measure levels of WAG emitted while anesthetizing mice with isoflurane for 15 min. They evaluated four different WAG scavenging conditions during induction and maintenance anesthesia: two conditions that used passive techniques and two that used active techniques. Isoflurane concentrations were measured at three different locations: in the operator's vicinity, at the mouse-facemask interface and in the room environment. Passive scavenging of WAG improved when chambers were purged with oxygen after induction and when a diaphragm-sealed facemask delivered a reduced anesthetic flow rate during maintenance anesthesia. Active scavenging of WAG improved when a relief intake opening was provided in the induction chamber's vacuum line, vacuum draw after induction was regulated and the anesthetic flow rate and vacuum scavenging draw were balanced during maintenance anesthesia using a facemask that separated the breathing space from the scavenging zone. Additionally, time-weighted average isoflurane WAG levels detected by personal dosimeters correlated with real-time measurements made using infrared spectroscopy. These observations contribute to the development of a substantiated program for monitoring WAG air quality.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
20 March 2014
The machined facemask used for maintenance anesthesia in Condition 4 and shown in Figure 5 (left) was incorrectly identified in the Methods (page 375, first column, sixth sentence). The facemask is a product of VetEquip, Inc. (Pleasanton, CA).
References
Smith, J.C. in Anesthesia and Analgesia in Laboratory Animals 2nd edn. (eds. Fish, R.E., Danneman, P.J. & Brown, M.) 183–194 (Academic, San Diego, CA, 2008).
American Society of Anesthesiologists. Waste anesthetic gases: information for management in anesthetizing areas and post-anesthesia care unit (PACU). (American Society of Anesthesiologists, Park Ridge, IL, 1999).
Occupational Safety and Health Administration. Anesthetic gases: guidelines for workplace exposures. (US Department of Labor, Washington, DC, 1999; revised 18 May 2000). http://www.osha.gov/dts/osta/anestheticgases/index.html.
Institute for Laboratory Animal Research. Guide for the Care and Use of Laboratory Animals 8th edn. (National Academies Press, Washington, DC, 2011).
Institute of Laboratory Animal Resources. Occupational Health and Safety in the Care and Use of Research Animals (National Academies Press, Washington, DC, 1997).
Smith, J.C. & Bolon, B. Atmospheric waste isoflurane concentrations using conventional equipment and rat anesthesia protocols. Contemp. Top. Lab. Anim. Sci. 41, 10–17 (2002).
Smith, J.C. & Bolon, B. Comparison of three commercially available activated charcoal canisters for passive scavenging of waste isoflurane during conventional rodent anesthesia. Contemp. Top. Lab. Anim. Sci. 42, 10–15 (2003).
Smith, J.C. & Bolon, B. Isoflurane leakage from non-rebreathing rodent anaesthesia circuits: comparison of emissions from conventional and modified ports. Lab. Anim. 40, 200–209 (2006).
Dorsch, J.A. & Dorsch, S.E. (eds.) Understanding Anesthesia Equipment 4th edn. 355–398 (Williams & Wilkins, Baltimore, MD, 1999).
Wolforth, J. & Dyson, M.C. Flushing induction chambers used for rodent anesthesia to reduce waste anesthetic gas. Lab Anim. (NY) 40, 76–83 (2011).
Glen, J.B., Cliff, G.S. & Jamieson, A. Evaluation of a scavenging system for use with inhalation anesthesia techniques in rats. Lab. Anim. 14, 207–211 (1980).
Hunter, S.C., Glen, J.B. & Butcher, C.J. A modified anaesthetic vapor extraction system. Lab. Anim. 18, 42–44 (1984).
Peters, B. Use of the anesthetic gas isoflurane within a BSC. Technical Bulletin GTB0158 (Nuaire, Plymouth, MN, 18 August 2006).
Taylor, D.K. & Mook, D.M. Isoflurane waste anesthetic gas concentrations associated with the open-drop method. J. Am. Assoc. Lab. Anim. Sci. 48, 61–64 (2009).
American College of Veterinary Anesthesiologists. Commentary and recommendations on control of waste anesthetic gases in the workplace. J. Am. Vet. Med. Assoc. 209, 75–77 (1996).
National Institute for Occupational Safety and Health. Criteria for a recommended standard: occupational exposure to waste anesthetic gases and vapors. DHEW Publication No. 77-140 (US Department of Health, Education, and Welfare, Public Health Service, Center for Disease Control, National Institute for Occupational Safety and Health, Washington, DC, 1977).
Abbott Laboratories Material Safety Data Sheet, Isoflurane, USP, 5260/B506 (Abbott Park, IL, 2007). http://www.abbottanimalhealth.com/static/cms_workspace/Updates/IsoFlo_MSDS_030310.pdf.
Public Health Service. Policy on Humane Care and Use of Laboratory Animals (US Department of Health and Human Services, Washington, DC, 1986; amended 2002).
Public Health Service. US Government Principles for the Utilization and Care of Vertebrate Animals Used in Testing, Research, and Training (US Department of Health and Human Services, Washington, DC, 2002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Todd, T., Morse, J., Casagni, T. et al. Monitoring and mitigating isoflurane emissions during inhalational anesthesia of mice. Lab Anim 42, 371–379 (2013). https://doi.org/10.1038/laban.282
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/laban.282