Abstract
Objective:
The use of inhaled nitric oxide (iNO) in preterm infants remains controversial. In October 2010, a National Institutes of Health consensus development conference cautioned against use of iNO in preterm infants. This study aims (1) to determine the prevalence and variability in use of iNO in the Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network (NICHD NRN) before and after the consensus conference and (2) separately, to examine associations between iNO use and severe bronchopulmonary dysplasia (BPD) or death.
Study design:
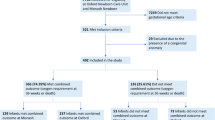
The NICHD NRN Generic Database collects data including iNO use on very preterm infants. A total of 13 centers contributed data across the time period 2008 to 2011. Infants exposed or not to iNO were compared using logistic regression, which included factors related to risk as well as their likelihood of being exposed to iNO.
Result:
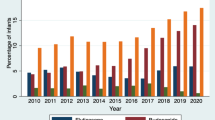
A total of 4885 infants were assessed between 2008 and 2011; 128 (2.6%) received iNO before day 7, 140 (2.9%) between day 7 and 28, and 47 (1.0%) at >28 days. Center-specific iNO use during 2008 to 2010 ranged from 21.9 to 0.4%; 12 of 13 sites reduced usage and overall NRN iNO usage decreased from 4.6 to 1.6% (P<0.001) in 2011. The use of iNO started between day 7 and day 14 was more prevalent among younger infants with more severe courses in week 1 and associated with increased risk of severe BPD or death (odds ratio 2.24; 95% confidence interval 1.23 to 4.07).
Conclusion:
The variability and total use of iNO decreased in 2011 compared with 2008 to 2010. iNO administration started at ⩾day 7 was associated with more severe outcomes compared with infants without iNO exposure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Neonatal Inhaled Nitric Oxide Study Group. Inhaled nitric oxide for term and near term infants for hypoxic respiratory failure. N Eng J Med 1997; 336: 597–604.
Finer NN, Barrington KJ . Nitric oxide for respiratory failure in infants born at or near term. Cochrane Database Syst Rev 2006; ((4)): CD000399.
Truog WE, Castor CA, Sheffield MJ . Neonatal nitric oxide use: predictors of response and financial implications. J Perinatol 2003; 23 (2): 128–132.
Schreiber MD, Gin-Mestan K, Marks JD, Huo D, Lee G, Srisuparp P . Inhaled nitric oxide in premature infants with the respiratory distress syndrome. N Engl J Med 2003; 349 (22): 2099–2107.
Ballard RA, Truog WE, Cnaan A, Martin RJ, Ballard PL, Merrill JD et al. Inhaled nitric oxide in preterm infants undergoing mechanical ventilation. N Engl J Med 2006; 355 (4): 343–353.
Van Meurs KP, Wright LL, Ehrenkranz RA, Lemons JA, Ball MB, Poole WK et al. Inhaled nitric oxide for premature infants with severe Respiratory Failure. N Engl J Med 2005; 353 (1): 13–22.
Kinsella JP, Cutter GR, Walsh WF, Gerstmann DR, Bose CL, Hart C et al. Early inhaled nitric oxide therapy in premature newborns with respiratory failure. N Engl J Med 2006; 355: 354–364.
Mercier JC, Hummler H, Durrmeyer X, Sanchez-Luna M, Carnielli V, Field D et al. EUNO Study Group. Inhaled nitric oxide for prevention of bronchopulmonary dysplasia in premature babies (EUNO): a RCT. Lancet 2010; 376: 346–354.
Mestan KK, Marks JD, Hecox K, Huo D, Schreiber MD . Neurodevelopmental outcomes of premature infants treated with inhaled nitric oxide. N Engl J Med 2005; 353 (1): 23–32.
Patrianakos-Hoobler AI, Marks JD, Msall ME, Huo D, Schreiber MD . Safety and efficacy of inhaled nitric oxide treatment for premature infants with respiratory distress syndrome: follow-up evaluation at early school age. Acta Paediatr 2011; 100 (4): 524–528.
Hibbs AM, Walsh MC, Martin RJ, Truog WE, Lorch SA, Alessandrini E et al. One-year respiratory outcomes of preterm infants enrolled in the Nitric Oxide (to Prevent) Chronic Lung Disease Trial. J Pediatr 2008; 153 (4): 525.
Zupancic JA, Hibbs AM, Palermo L, Truog WE, Cnaan A, Black DM et al. Economic evaluation of inhaled nitric oxide in preterm infants undergoing mechanical ventilation. Pediatrics 2009; 124 (5): 1325–1332.
Barrington KJ, Finer NN . Inhaled nitric oxide for respiratory failure in preterm infants. Cochrane Database Syst Rev 2007; ((12)): CD000509.
Donohoe PK, Gilmore MM, Cristofalo E, Wilson RF, Weiner JZ, Lau BD et al. Inhaled nitric oxide in preterm infants: a systematic review. Pediatrics 2011; 355: 413–421.
Askie LM, Ballard RA, Cutter G, Dani C, Elbourne D, Field D et al. Inhaled nitric oxide in preterm infants: a systematic review and individual patient data meta-analysis. BMC Pediatr 2010; 23: 10–15.
Clark RH, Ursprung RL, Walker MW, Ellsbury DL, Spitzer AR . The changing pattern of inhaled nitric oxide use in the neonatal intensive care unit. J Perinatol 2011; 30 (12): 800–804.
Padula MA, Grover TR, Brozanski B, Zaniletti I, Nelin LD, Asselin JM et al. Therapeutic interventions and short-term outcomes for infants with severe bronchopulmonary dysplasia born at <32 weeks’ gestation. J Perinatol 2013; 33: 877–881.
Cole FS, Alleyne C, Barks JD, Boyle RJ, Carroll JL, Dokken D et al. NIH Consensus Developmental Conference Statement: Inhaled nitric oxide therapy for premature infants. Pediatrics 2011; 127: 363–369.
Braitman L, Rosenbaum P . Rare outcomes, common treatment: analytic strategies using propensity scores. Annals Internal Med 2002; 137 (8): 693–695.
Walsh MC, Yao Q, Gettner P, Hale E, Collins M, Hensman A et al. Impact of a physiologic definition on bronchopulmonary dysplasia rates. Pediatrics 2004; 114: 1305–1311.
Ehrenkranz RA, Walsh MC, Vohr BR, Jobe AH, Wright LL, Fanaroff AA et al. National Institutes of Health consensus definition of bronchopulmonary dysplasia. Pediatrics 2005; 116: 1353–1360.
Rosenbaum PR, Rubin DB . The central role of the propensity score in observational studies for causal effects. Biometrika 1983; 70: 41.
D’Agostino RB Jr . Propensity score methods for bias reduction in the comparison of a treatment to a non-randomized control group. Statist Med 1998; 17: 2265–2281.
Freeman VA . Very Low Birth Weight Babies Delivered at Facilities for high-Risk Neonates: A Review of Title V National Performance Measure 17. University of North Carolina at Chapel Hill, Cecil G. Sheps Center for Health Services Research: Chapel Hill, NC, 2010.
Stenger M, Slaughter J, Kelleher K, Shepherd E, Klebanoff M, Reagan P et al. Hospital variation in nitric oxide use for premature infants. Pediatrics 2012; 129: 945–951.
Peliowski A and the Canadian Paediatric Society, Fetus and Newborn Committee. Inhaled nitric oxide use in newborns. Pediatr Child Health 2012; 17: 95–97.
Kumar P and the Committee the Fetus and Newborn. Use of inhaled nitric oxide in preterm infants. Pediatrics 2014; 133: 167–170.
Concato J . Study design and ‘evidence’ in patient-oriented research. Am J Respir Crit Care Med 2013; 187: 1167–1172.
Albert RK . ‘Lies, damned lies…’ and observational studies in comparative effectiveness research. Am J Respir Crit Care Med 2013; 187: 1173–1177.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Truog, W., Nelin, L., Das, A. et al. Inhaled nitric oxide usage in preterm infants in the NICHD neonatal research network: inter-site variation and propensity evaluation. J Perinatol 34, 842–846 (2014). https://doi.org/10.1038/jp.2014.105
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2014.105
This article is cited by
-
Inter-center variation in death or tracheostomy placement in infants with severe bronchopulmonary dysplasia
Journal of Perinatology (2017)
-
Inhaled nitric oxide use in preterm infants in California neonatal intensive care units
Journal of Perinatology (2016)