Abstract
Objective:
To assess chlorhexidine absorption and skin tolerability in premature infants, following skin antisepsis with 2% aqueous chlorhexidine gluconate (CHG) prior to peripherally inserted central catheter (PICC) placement.
Study Design:
Neonates less than 32 weeks gestation had skin cleansed with CHG prior to PICC placement. CHG concentrations were measured on serial blood samples. Skin integrity was evaluated for 2 weeks after CHG exposure.
Result:
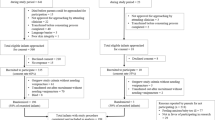
Twenty infants were enrolled; median gestational age was 28 2/7 weeks (range 24 3/7 to 31 4/7). Ten infants had detectable serum chlorhexidine concentrations (range 1.6 to 206 ng ml−1). Seven of these infants had their highest serum concentration 2 to 3 days following exposure. No CHG-related skin irritation occurred in any infant.
Conclusion:
CHG was detected in the blood of preterm infants receiving CHG skin antisepsis for PICC insertion. Highest serum concentrations occurred 2 to 3 days after exposure. Further investigation is needed to determine the clinical relevance of CHG absorption in preterm infants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Milstone AM, Passaretti CL, Perl TM . Chlorhexidine: expanding the armamentarium for infection control and prevention. Clin Infect Dis 2008; 46 (2): 274–281.
O’Grady NP, Alexander M, Dellinger EP, Gerberding JL, Heard SO, Maki DG et al. Guidelines for the prevention of intravascular catheter-related infections. The Hospital Infection Control Practices Advisory Committee, Center for Disese Control and Prevention. Pediatrics 2002; 110 (5): e51.
Tamma PD, Aucott SW, Milstone AM . Chlorhexidine use in the neonatal intensive care unit: results from a national survey. Infect Control Hosp Epidemiol 2010; 31 (8): 846–849.
U.S. Food and Drug Administration MedWatch (Internet)., Silver Spring, MD, USAhttp://www.fda.gov/Safety/MedWatch/SafetyInformation/Safety-RelatedDrugLabelingChanges/ucm307387.htm . (U.S. Food and Drug Administration; 2012 (updated 12 June 2012; cited 27 November 2012)).
Aggett PJ, Cooper LV, Ellis SH, McAinsh J . Percutaneous absorption of chlorhexidine in neonatal cord care. Arch Dis Child 1981; 56 (11): 878–880.
Cowen J, Ellis SH, McAinsh J . Absorption of chlorhexidine from the intact skin of newborn infants. Arch Dis Child 1979; 54 (5): 379–383.
Garland JS, Alex CP, Uhing MR, Peterside IE, Rentz A, Harris MC . Pilot trial to compare tolerance of chlorhexidine gluconate to povidone-iodine antisepsis for central venous catheter placement in neonates. J Perinatol 2009; 29 (12): 808–813.
Johnsson J, Seeberg S, Kjellmer I . Blood concentrations of chlorhexidine in neonates undergoing routine cord care with 4% chlorhexidine gluconate solution. Acta Paediatr Scand 1987; 76 (4): 675–676.
Mullany LC, Khatry SK, Sherchand JB, Leclerq SC, Darmstadt GL, Katz J et al. A randomized controlled trial of the impact of chlorhexidine skin cleansing on bacterial colonization of hospital-born infants in Nepal. Pediatr Infect Dis J 2008; 27 (6): 505–511.
O’Brien CABJ, Speck WT, Carr H . Effect of bathing with a 4 per cent chlorhexidine gluconate solution on neonatal bacterial colonization. J Hosp Infect 1984; 5 (suppl 1): 141.
O’Neill JHM, Challop R, Driscoll J, Speck W, Sprunt K . Percutaneous absorption potential of chlorhexidine in neonates. Curr Ther Res 1982; 31 (3): 485–489.
Mullany LC, Darmstadt GL, Tielsch JM . Safety and impact of chlorhexidine antisepsis interventions for improving neonatal health in developing countries. Pediatr Infect Dis J 2006; 25 (8): 665–675.
Chapman AK, Aucott SW, Milstone AM . Safety of chlorhexidine gluconate used for skin antisepsis in the preterm infant. J Perinatol 2012; 32 (1): 4–9.
Milstone ABP, Aucott S, Bearer CF, Tang N . Chlorhexidine reduces L1-mediated neurite outgrowth. Annual Meeting of the Pediatric Academic Society. Denver, 2011.
Davies GE, Francis J, Martin AR, Rose FL, Swain G . 1:6-Di-4'-chlorophenyldiguanidohexane (hibitane); laboratory investigation of a new antibacterial agent of high potency. Br J Pharmacol Chemother 1954; 9 (2): 192–196.
Sebben JE . Surgical antiseptics. J Am Acad Dermatol 1983; 9 (5): 759–765.
Greaves SJ, Ferry DG, McQueen EG, Malcolm DS, Buckfield PM . Serial hexachlorophene blood levels in the premature infant. N Z Med J 1975; 81 (537): 334–336.
Kalia YN, Nonato LB, Lund CH, Guy RH . Development of skin barrier function in premature infants. J Invest Dermatol 1998; 111 (2): 320–326.
Barker N, Hadgraft J, Rutter N . Skin permeability in the newborn. J Invest Dermatol 1987; 88 (4): 409–411.
Reynolds PR, Banerjee S, Meek JH . Alcohol burns in extremely low birthweight infants: still occurring. Arch Dis Child Fetal Neonatal Ed 2005; 90 (1): F10.
Watkins AM, Keogh EJ . Alcohol burns in the neonate. J Paediatr Child Health 1992; 28 (4): 306–308.
Bringue Espuny X, Soria X, Sole E, Garcia J, Marco JJ, Ortega J et al. Chlorhexidine-methanol burns in two extreme preterm newborns. Pediatr Dermatol 2010; 27 (6): 676–678.
Mannan K, Chow P, Lissauer T, Godambe S . Mistaken identity of skin cleansing solution leading to extensive chemical burns in an extremely preterm infant. Acta Paediatr 2007; 96 (10): 1536–1537.
Andersen C, Hart J, Vemgal P, Harrison C . Prospective evaluation of a multi-factorial prevention strategy on the impact of nosocomial infection in very-low-birthweight infants. J Hosp Infect 2005; 61 (2): 162–167.
Lashkari HP, Chow P, Godambe S . Aqueous 2% chlorhexidine-induced chemical burns in an extremely premature infant. Arch Dis Child Fetal Neonatal Ed 2012; 97 (1): F64.
Arifeen SE, Mullany LC, Shah R, Mannan I, Rahman SM, Talukder MR et al. The effect of cord cleansing with chlorhexidine on neonatal mortality in rural Bangladesh: a community-based, cluster-randomised trial. Lancet 2012; 379 (9820): 1022–1028.
Acknowledgements
This manuscript was supported by Grant Number UL1 RR 025005 from the National Center for Research Resources (NCRR), a component of the National Institutes of Health (NIH) and NIH Roadmap for Medical Research, and its contents are solely the responsibility of the authors and do not necessarily represent the official view of NCRR or NIH. We thank Dr Craig Hendrix, the Johns Hopkins Hospital and Johns Hopkins Bayview Medical Center NICU nursing staffs, the Johns Hopkins Bayview Medical Center Neonatal Nurse Practitioners, and the Johns Hopkins ICTR Navigators.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
AM received grant support from Sage Products. The remaining authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Chapman, A., Aucott, S., Gilmore, M. et al. Absorption and tolerability of aqueous chlorhexidine gluconate used for skin antisepsis prior to catheter insertion in preterm neonates. J Perinatol 33, 768–771 (2013). https://doi.org/10.1038/jp.2013.61
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jp.2013.61
Keywords
This article is cited by
-
Microbiome dysbiosis: a modifiable state and target to prevent Staphylococcus aureus infections and other diseases in neonates
Journal of Perinatology (2024)
-
Myelin toxicity of chlorhexidine in zebrafish larvae
Pediatric Research (2023)
-
Global variation in skin injures and skincare practices in extremely preterm infants
World Journal of Pediatrics (2023)
-
The downside of antimicrobial agents for wound healing
European Journal of Clinical Microbiology & Infectious Diseases (2019)
-
Prävention von Gefäßkatheter-assoziierten Infektionen bei Früh- und Neugeborenen
Bundesgesundheitsblatt - Gesundheitsforschung - Gesundheitsschutz (2018)