Abstract
Childhood obesity in the United States has doubled over the last three decades and currently affects 17% of children and adolescents. While much research has focused on individual behaviors impacting obesity, little research has emphasized the complex interactions of numerous chemical and non-chemical stressors found in a child’s environment and how these interactions affect a child’s health and well-being. The objectives of this systematic scoping review were to (1) identify potential chemical stressors in the context of non-chemical stressors that impact childhood obesity; and, (2) summarize our observations for chemical and non-chemical stressors in regards to child-specific environments within a community setting. A review was conducted to identify chemical and non-chemical stressors related to childhood obesity for the childhood life stages ranging from prenatal to adolescence. Stressors were identified and grouped into domains: individual behaviors, family/household behaviors, community stressors, and chemical exposures. Stressors were related to the child and the child’s everyday environments and used to characterize child health and well-being. This review suggests that the interactions of chemical and non-chemical stressors are important for understanding a child’s overall health and well-being. By considering these relationships, the exposure science research community can better design and implement strategies to reduce childhood obesity.
Similar content being viewed by others
Introduction
Obesity is a worldwide problem affecting both affluent and less affluent countries. According to the World Health Organization (WHO), obesity has nearly doubled since 1980 and, as of 2008, there were 40 million (6%) preschool-aged children worldwide classified as overweight.1 De Onis et al.2 estimated that by 2020 the percentage is expected to increase to 9.1%, resulting in approximately 60 million preschool-aged children who are overweight or obese. In the United States, the Centers for Disease Control and Prevention (CDC) estimates that childhood obesity affects 17% of all children and adolescents, a rate which has tripled from one generation ago.3, 4, 5
The United Nations (UN) recognizes that reducing obesity requires urgent global action and has responded with policies and procedures that require the involvement of stakeholders from many sectors (such as health, education, nutrition, policy, agriculture, industry and trade, finance) to combat this epidemic.1 The UN realizes that childhood obesity will only be resolved with a combination of interventions catering to the specific needs of the geographic area and population receiving the intervention. These interventions have been implemented as part of programs already in place to improve health and well-being.1
Obesity and being overweight result when the body accumulates fat over time above what are considered normal adiposity levels. It is increasingly recognized that obesity is a complex issue that results from the interactions of many factors, including but not limited to genetic, metabolic, behavioral, and environmental. Much research has been undertaken to study the link between any one individual factor and obesity.6, 7, 8, 9, 10, 11, 12, 13, 14 For example, metabolic risk factors such as diabetes have been shown to be linked to obesity, and there is growing research exploring the associations between chemical exposures and diabetes.15, 16 However, the inter-relationships between stressors are not well understood. Generally, the obesity epidemic is thought to be a result of chronic energy imbalance between calories consumed and calories expended.17, 18, 19, 20, 21, 22, 23 In addition to physical activity and caloric intake, other non-chemical stressors that may influence weight include personal habits (e.g., smoking); psychosocial stress (e.g., parental divorce, domestic violence, exercising poor judgement, depression); access to health care; and/or aspects of the built and natural environments.24, 25, 26, 27 There is also increasing evidence that obesogens (i.e., chemical stressors) may influence obesity by affecting metabolic activities in the body. Finally, genetic predisposition plays a large role, accounting for >40% of the population variation in body mass index (BMI), suggesting that genes factor into how the body captures, stores, and uses energy from food consumption.28 Some health effects resulting from exposures to chemical and non-chemical stressors during the prenatal period are considered to have lifelong impacts, suggesting that reduced exposures to obesogens and non-chemical stressors may need to begin early in life to serve as a preventive measure.29
The goal of this systematic scoping review is to examine recent evidence from the peer-reviewed literature about the role of chemicals as obesogens in human populations and the interrelationships between chemical and non-chemical stressors in impacting childhood obesity. The specific objectives are to (1) identify potential chemical stressors in the context of non-chemical stressors that impact childhood obesity and (2) summarize our observations for the chemical and non-chemical stressors in regards to child-specific environments (i.e., where a child lives, learns, and plays) in a community setting. A better understanding of the cumulative exposures that children experience may inform improved approaches to manage and reduce the incidence of childhood obesity.
Methods
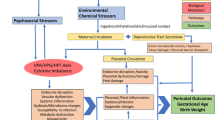
This literature review focused on a holistic analysis of childhood obesity by considering myriad influencing factors, including individual behaviors; social determinants; the natural environment; the built environment; and potential exposures to environmental chemicals. We examined multiple stressors for childhood lifestages guided in part by EPA’s age grouping guidance.30
Stressors are defined as any physical, chemical, or biological entity that can induce an adverse response. Chemical stressors are exogenous environmental compounds which change or damage living organisms or ecosystems.31, 32, 33 Non-chemical stressors are factors found in the built, natural, and social environments such as noise, temperature, humidity, stress from the built environment, social factors, and lack of health care.24, 26, 33
Using PubMed and Google Scholar, we identified relevant literature articles using several search strings (Figure 1—Step 1). More information on the search strings (including specific stressor terms) can be found in the Supplementary Information. Oftentimes published articles led to additional relevant articles or books. All references were first screened by title to determine relevance. For each stressor identified, if the childhood data were not available, we included in vitro/in vivo studies to support the limited epidemiological findings.
Selected articles were published between January 2004 and July 2014, written in English, and addressed the topic of childhood obesity and chemical and or non-chemical stressors (Figure 1—Step 2). Abstracts were then reviewed (Figure 1—Step 3), and duplicate articles, concept papers, any study that excluded data collection and analysis, any study focused on obesity in adults, and any study focused predominantly on diabetes and asthma were removed from further consideration, reducing the number of manuscripts for full review to 723 (Figure 1—Step 4). Full text copies of manuscripts were obtained and evaluated based on the following criteria:
-
1)
Focus on children ranging in age from prenatal to 18 years
-
2)
Type of study (e.g., longitudinal, retrospective, prospective, cross-sectional)
-
3)
Available data (with supporting information (e.g., methods) to assess relevance and data quality).
In the end, 234 articles were included in this review (Figure 1—Step 5). When available, review papers were selected for well-studied stressors (e.g., activity, diet, socioeconomic status, prenatal exposures, environmental tobacco smoke). Citations were managed with EndNote X7.
This manuscript is organized into the following sections: individual and family/household behaviors, community stressors, and chemical exposures to align with the Sustainable and Healthy Communities research program (https://www.epa.gov/research).
Results
Individual Behaviors
A summary of papers included in this review can be found in Supplementary Tables S1–S4. Individual behaviors include: diet, activity level, and sleep. Because the relationships between diet, activity level, and sleep, and childhood obesity are well studied, we only provide brief summaries of these important factors here. More information can be found in the Supplementary Information.
Diet
Many research efforts have studied the relationship between dietary factors and childhood obesity including diet during developmental periods (including maternal diet during prenatal development), parental influence, food advertisements, and neighborhood food (e.g., fast-food restaurants, convenience stores, and small food stores), sweetened beverages, high-fructose corn syrup, processed foods, junk food, and meal frequency. Inconsistencies indicate that increased energy intake alone may not be the leading cause of childhood obesity. More details on associations between diet and childhood obesity can be found in the Supplementary Information.
Activity Level
Research has examined activity levels and obesity in children including aspects of a child’s built environment, socioeconomic status, gender, race, social support, technology, and home environment. Evidence in the literature suggests that activity level alone may not explain childhood obesity. More details on associations between activity level and childhood obesity can be found in the Supplementary Information.
Sleep
Sleep plays an important role in all aspects of a child’s life including overall health. However, inconsistencies indicate that sleep alone may not explain childhood obesity. More details on the relationship between sleep and childhood overweight/obesity can be found in the Supplementary Information.
Family and Social Stressors
Family and social stressors include socioeconomic status (SES), psychosocial stress, maltreatment (Supplementary information), and prenatal and postnatal exposures that children cannot avoid on their own (e.g., maternal smoking, secondhand smoke exposure (SHS), illicit substance use (Supplementary Information)).
Socioeconomic Status (SES)
SES is a combined measure of economic and social position as it relates to others based on race/ethnicity, income, education, and occupation. Studies report conflicting results when examining levels of SES and obesity. Lower SES neighborhoods are at a higher risk for childhood obesity since they often have limited access to healthy food options and facilities for activity as compared to middle and upper SES neighborhoods.34, 35 Aitsi-Selmi et al.36 found that higher SES Brazilian girls had lower adiposity as adults, but the opposite was found for boys. Another study of Brazilian children who were followed until age 15 years found that higher SES children were more likely to be overweight and obese than lower SES children.37
Studies of children in the United States, Canada, and Europe have shown that children from higher SES families are more physically active, eat healthier food options, and participate in more organized recreational activities as compared to lower SES families.38, 39 Studies examining SES have shown an association with low SES, increased childhood obesity and greater adiposity.34, 40, 41, 42, 43, 44 Data collected in a German health interview of 13,450 obese/overweight children found that obesity was strongly related to both low SES and parental obesity.45 Similarly, low SES backgrounds in conjunction with high amounts of television viewing put children at a higher risk for being overweight.46, 47
Other aspects of SES have also been examined. Morrissey et al.48 examined the association between maternal work and children’s BMI. They found that maternal employment has a cumulative influence associated with increases in the child’s BMI, especially for those children in the sixth grade, as compared to younger children.48 Chen and Escarce49 found that children were more likely to be obese by the fifth grade if they lived in a single parent household, did not have any siblings, or both.
Studies examining race/ethnicity related to weight and SES have found varied results. For example, African-American children have a prevalence of being overweight when compared to Caucasian children.50, 51 Whitaker and Orzol52 found that race/ethnicity and other SES indicators did not explain the higher rates of obesity in Hispanic children when compared to Caucasian or African American children. Other studies reported in the literature have shown similar conflicting results.53, 54, 55
Psychosocial Stress
Psychosocial stress has been identified as a possible factor contributing to childhood obesity. Psychosocial stress may result from a child’s school, family, or other interpersonal environments they encounter daily. It is hypothesized that these psychosocial stressors lead to metabolic changes that increase cortisol and catecholamine levels, leading to behavioral changes like emotional eating, inactivity, and disruption of sleep. The relationship between obesity and various psychosocial stressors is not consistent.
Parental stress could potentially preprogram the fetus resulting in childhood obesity as a potential outcome.56, 57 A Danish National Birth cohort of 37,764 women and children examined associations between anxiety, depression, and stress of the mother during pregnancy and the child’s rate of being overweight at age 7 years. The results indicated maternal stress during pregnancy did not appear to influence a child’s weight at age 7 years.56 Another study by Van Dijk et al.57 found that increased cortisol production and work strain while the mother was pregnant may not have pre-programmable obesity and adiposity effects on children at age 5 years.
Conversely, a Danish study found that children whose mothers were stressed during 6 to 0 months before birth due to bereavement of someone close had increased BMI values as compared to children whose mothers did not report a death during their pregnancy. The increased BMI values, however, were not significant until the age of 10 years and beyond, suggesting that early-life exposures to stress may have effects later on in childhood.58
Other parental stressors that influence BMI levels in children include violence, serious life events, food or housing insecurity, maternal depression or substance abuse, and paternal incarceration.59, 60, 61, 62, 63, 64, 65, 66, 67 For many studies, SES was a controlled variable in order to determine associations with other psychosocial stressors, but an article by Garasky et al.68 found that financial strain was positively associated with children being overweight or obese.
Besides familial stress, childhood depression has been associated with obesity. Blaine et al.69 found that children who were depressed were two and a half times more likely to be obese than children who were not depressed. Similar associations were found in other studies examining children with depression, anxiety, stress, unhealthy peer relationships alone or in combination.65, 70, 71, 72, 73
Prenatal Exposure
Research evaluating childhood obesity also examines how the child responds to shared environmental stressors between mother and child. Parental obesity is associated with infant and early childhood weight gain.10, 74, 75 Studies have shown an association with a mother’s weight gain during pregnancy and a child’s increased risk of being obese later in life.74, 75, 76 To determine whether a mother’s weight gain during pregnancy was the cause and not the infant’s surrounding environment, two studies examined the mother and father’s BMI values and their relationship to the child’s BMI value and found that the association between maternal BMI and child BMI was statistically similar to the paternal BMI and child BMI, suggesting that increased adiposity could be more related to the postnatal environment and not a maternal effect.10, 77
Secondhand Smoke Exposure (SHS)
The role of prenatal cigarette smoke exposure and childhood obesity has long been a focus of many adverse health outcomes including obesity. These studies found an association between maternal prenatal smoking and childhood obesity even after accounting for various confounding factors.12, 16, 76, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91 Although there is a compelling link between prenatal smoking and childhood obesity, it is not always clear-cut for both genders and all maternal smoking levels.86, 87 One study followed children from 2 to 10 years of age and found that males whose mothers smoked during pregnancy had a lower BMI at age 2 years than those born to non-smoking mothers or had partners who smoked. The study further indicated that female offspring had a slightly higher BMI and this was not attenuated by adjusting for confounders.92
Other studies not only looked at the mother’s smoking status during pregnancy, but also the association with paternal smoking both during and after pregnancy. Weak paternal associations were found by Leary et al.88 suggesting confounding by social factors rather than a direct effect from maternal smoking. Another study examined the role of parental smoking and childhood obesity and found an association from paternal smoking when the child was 6 years old. They further examined the paternal association as a confounder, but it only partially explained the effect of maternal smoking and childhood obesity, suggesting that paternal smoking might be a factor for obesogenic outcomes.93
Community Stressors
Community stressors include the built and natural environments and neighborhood food outlets in a child’s environment.
Built Environment
The built environment (BE) represents all infrastructure found in a community, including but not limited to roads, sidewalks, parks, trails, recreational facilities, neighborhood surroundings, playgrounds, buildings, and homes. These neighborhood characteristics can promote physical activity by increased active transport to work, school, and food sources.33, 35, 38, 94 Much research pertaining to the BE is focused on the importance of community level factors in explaining dietary choices or physical activity, and links to various health outcomes, including obesity. Studies examining aspects of the BE found that property disorder, nuisances, violent and total crime were all associated with increased weight, while factors such as housing damage, vacancy, bars on doors/windows, and warning signs had no association with childhood obesity.95, 96
Researchers have used various approaches to examine associations between BE factors and childhood obesity.95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107, 108 Evidence in the literature shows that improvements to the BE (e.g., places to exercise, green space, sidewalks, recreational areas, neighborhood safety, food access and healthier food options) will increase physical activity, potentially resulting in a decrease in obesity.94, 97, 98, 109 Decreased activity levels have been found when these services are difficult to access.103, 104, 110 Lower activity levels due to a poorly structured and unsafe BE are associated with an increase in obesity.99 Rahman et al.35 examined the decline in how children actively get to school by analyzing available data from the U.S. Department of Transportation. The analysis indicated a rapid decline in the percentage of children who walk or bike to school (42% in 1969 to 13% in 2001). In a 2009 survey, 84% of children either rode the bus or commuted in cars, largely due to a lack of neighborhood sidewalks and concerns about distance and traffic safety.35
Daycares and schools can potentially affect children’s health and well-being. Studies have indicated a higher BMI for children who received care from a center-/family-based facility or by a relative/nanny.101, 105 Although these studies found a correlation with care outside the home and increased BMI, no clear underlying causal linkages were identified. It was suggested that there may be a need for interventions to promote awareness of activity levels and better nutrition.101 On the other hand, since children spend a lot of their time in school, they obtain physical activity by participating in teams, lessons, and gym class, suggesting that neighborhood physical characteristics such as traffic, density, and land use may be less relevant.100
Neighborhood Food Outlets
Many food outlets readily accessible to children have poor dietary choices (e.g., fast-food restaurants, convenience stores, small food stores) with high fat, large portion sizes, and sugary drink options.111, 112, 113 Research shows that children who have easy access to poor quality food outlets tend to have a higher BMI and the potential to become overweight/obese when compared to children who do not have easy access to these food outlet types.107 In addition, the built environment plays a critical role in how adolescents perceive distances and safety to these various food outlets. Studies show that these types of stores, when in close proximity to neighborhoods or schools, increased the likelihood of purchases made at these food outlet types.107, 111, 112, 113, 114 Sanchez et al.111 indicated that minorities, specifically in neighborhoods that are predominantly Hispanic and African American, have a higher prevalence of obesity related to neighborhood food choices, and that 1/3 of all U.S. public middle and high schools have at least one fast food or convenience store within walking distance. In 2011, Leung et al114 suggested that residents with more produce vendors or farmers markets nearby had a lower risk of being overweight/obese. On the other hand, Shier et al.115 found no consistent evidence to support the idea that more grocery stores lowered youth BMI or that greater exposure to fast-food restaurants, convenience stores, and small food stores increased BMI.
Natural Environment
The natural environment refers to parks, trails, and green spaces that provide opportunities for physical activity. Aspects of the natural environment are often considered within BE research studies, where they examine accessibility to recreational facilities and schools.33, 94, 97, 102, 116, 117
The literature indicates that greenness has many beneficial factors when it comes to influencing childhood obesity and activity levels. In a study that followed 9–10 year olds for 8 years, increased green space was significantly associated with lower BMI z-scores, suggesting that greener neighborhoods led to higher activity levels or more time spent outside.116 This correlation between increased proximity to large amounts of green space and reduced BMI levels remained consistent throughout other studies.97, 102, 118 Other studies examining activity levels and green space found that children living closer to parks and green spaces were more likely to be active and more creative than those who lived further away.119, 120 While Potestio et al.121 found similar results with reduced risk of childhood obesity, once they incorporated other factors, the effect was reduced to non-significant, suggesting that findings may differ depending on the type of urban environment being studied.
A review article by McCurdy et al.122 provides more examples of how nature and outdoor activity can improve a child’s health. Studies included in their review suggested that for every additional hour spent outdoors, physical activity increased and the prevalence of children being overweight dropped.122 Increasing urbanization and population density can potentially reduce the green space available for active recreation, leaving school environments as the only available play areas, but if they are made of asphalt, they can only be used for certain types of activities. This limitation can be resolved with school playgrounds that maintain a greener landscape (trees, gardens, trails) that may stimulate activity.123
Environmental Chemical Stressors
An obesogen is an environmental pollutant that inappropriately stimulates the development of adipose tissue, stores fat in existing cells, disrupts the energy balance controlling systems, alters tissue sensitivity to some neurotransmitters, or disturbs the activity of the autonomous nervous system.124, 125 This term was coined in 2006 by Bruce Blumberg when he discovered that tin-based compounds given to pregnant laboratory mice predisposed offspring to weight gain.29 This finding suggested that humans who have been exposed to certain types of chemicals during sensitive windows of development may be pre-programmed to store increased amounts of fat, resulting in a lifelong struggle to maintain a healthy weight and exacerbating the deleterious effects of poor diet and inadequate exercise.126, 127 Endocrine disrupting chemicals interfere with the body’s endocrine system and may adversely affect the metabolic system in humans by targeting the nuclear hormone proliferator-activated receptor gamma (PPARγ). The number of environmental chemicals that may activate peroxisome PPARγ is unknown, but much research indicates that activating PPARγ is an important pathway for adipogenesis and obesity.128, 129, 130, 131, 132
Although correlations between obesogens and obesity have been found in animal studies, there is a knowledge gap regarding how obesogens affect children’s health. To overcome this gap, researchers use retrospective cross-sectional analyses to examine relationships, in addition to epidemiological studies specifically examining obesogens. Various chemicals have been studied, including flame retardants, polychlorinated biphenyls (PCBs), perfluorinated compounds (PFCs), bisphenol A, phthalates, pesticides and their degradation products, metals, and mixtures of air pollutants (Supplementary Table S4). However, the research community does not agree on whether chemicals should be considered obesogens. In the following sections, we explore the evidence surrounding these chemicals. For metals, bisphenol A, phthalates, and mixtures of air pollution, the evidence suggests linkages between exposures to these chemical classes and obesity; hence these chemical classes are described in the manuscript. For the others, the evidence is less clear, and those are described in the Supplementary Information.
Metals
Organotins, a suspected obesogenic chemical class, are the active ingredients in fungicides, miticides, wood preservatives, and heat stabilizers in polyolefin plastics, among many other uses.126, 133, 134 Organotins are considered ubiquitous and human exposure may occur from ingestion of drinking water and foods and direct contact with house dust.126, 134, 135 Although there is compelling evidence to suggest that organotins may be contributing to the obesity epidemic, there is a data gap concerning their effects on humans.126, 134
Toxic metals may contribute to obesity by potentially interfering with aspects of metabolism by substituting for essential metals and micronutrients or by causing oxidative stress.136 Toxic metals such as lead, cadmium, cobalt, antimony, barium, molybdenum, thallium, tungsten, and cesium have been examined for their role in causing obesity. Various studies have examined how metals may cause obesity during the early life course (prenatal exposure during pregnancy to early lifestage environmental exposures) through ingestion or inhalation of low levels in air or secondhand smoke.136, 137, 138 One study examined the association between BMI, waist circumference (WC) and toxic metals, adjusting for age, ethnicity, and gender, using participants aged 6–60 years from the National Health and Nutrition Examination Survey (NHANES). Of the nine metals examined (barium, cadmium, cobalt, cesium, molybdenum, lead, antimony, thallium, tungsten), for children aged 6–18 years, BMI and WC were negatively associated with cobalt and lead exposures and positively associated with barium exposures. The other metals had no significant associations with children’s weight or WC.136 In 2011, Afeiche et al.137 examined prenatal lead exposures and the weight of children from the age of 0–5 years and found that maternal bone lead concentrations were associated with lower weight among female children but not male children. Another study found a negative association between maternal cadmium exposure and birth weight for girls and little evidence of any effect for boys, suggesting that gender could be a factor for susceptibility.138 The inconsistent associations between metal exposure and weight suggest further research is needed.
Persistent Organic Pollutants
Persistent organic pollutants, including various flame retardants, PCBs, and PFCs, have been linked to childhood obesity in some research. These manmade compounds have been or are used in numerous products including many found in a child’s environment. In addition, exposures may occur as a result of the compounds’ distributions in food, water, air, and other environmental media. Because the results reported in the literature are inconsistent, these chemicals are further discussed in the Supplementary Information.
Biologically Non-persistent Pollutants
Bisphenol A (BPA), a monomer component of polycarbonate plastics, can be found in canned foods, food and beverage containers, and other consumer products.6, 8, 127 Research indicates that BPA has estrogenic properties, can interfere with endocrine signaling pathways, and may affect energy balance and glucose homeostasis.8, 127, 139
BPA was measured in spot urine samples collected from pregnant women and children at ages 5 and 9 years as part of the CHAMACOS study. Results indicated that prenatal urinary BPA concentrations were negatively associated with BMI at 9 years of age in girls but not boys. Urinary BPA concentrations measured at 9 years of age were positively associated with BMI, WC, fat mass, and overweight/obesity in both boys and girls.140 Contrary to these findings, in a different cohort, Yamano et al.141 found that after a five year follow-up, urinary BPA measurements had no association with BMI. Similar results were seen in other cross-sectional studies where older children had higher BPA concentrations that correlated to an increased risk of being overweight/obese, specifically in females.142, 143 A cross-sectional study examining NHANES data (2003–2008) found that urinary BPA concentrations were significantly associated with obesity in children and adolescents after controlling for several cofactors.6 Another study using the NHANES data reported that non-Hispanic white boys were more likely to be overweight/obese, independent of other major risk factors, as a result of exposure to BPA.144 To support these associations, more research is needed using longitudinal studies to confirm the findings observed in the available cross-sectional studies.6, 142, 143, 144
Phthalates are a group of man-made chemicals widely used in industrial and consumer product applications.8, 145, 146 Exposure to phthalates can occur from food sources, plastic containers, flooring materials and wall coverings, medical devices, personal care products, lacquers, varnishes, and coatings. Exposure for infants and children may also come from skin contact with surfaces and frequent mouthing of fingers and other objects (e.g., plastic toys), ingestion of breast milk, infant formula, cow’s milk, or food packaging, and through inhalation.146 Similar to the PFCs, phthalates activate the PPARγ pathways in 3T3-L1 murine pre-adipocytes,147, 148 thereby altering adipocyte differentiation, energy storage, and or homeostatic controls.149, 150
In 2008 (ref. 145) and 2010 (ref. 150), Hatch et al. examined the associations between various urinary phthalate metabolites and BMI values and WC from NHANES. For adolescent girls, they found that BMI values and WC increased with increases in urinary monoethyl phthalate (MEP) concentrations, but were inversely related to increases in urinary mono(2-ethylhexyl)phthalate (MEHP) concentrations. They found no associations between BMI, WC, and phthalate metabolites measured for children aged 6 to 11 years.145, 150 Trasande et al.151 performed stratified and whole-sample cross-sectional analyses for NHANES children aged 6–19 years (2003–2008) and found that low molecular weight phthalate metabolites were associated with increases in obesity in non-Hispanic black children.151 Other studies have reported similar results in spite of weak statistical evidence that phthalates increase a child’s weight.152, 153, 154 While most phthalate research has focused on children in the United States, one cross-sectional study was conducted in three primary and three middle schools in China. The Chinese researchers found that, when age and sex were included in the model, MEHP and MEP were positively associated with BMI and WC.155
Pesticides and Degradation Products
Studies examining prenatal exposures to various pesticides and children’s weight have documented inconsistent observations. A detailed discussion can be found in the Supplementary Information.
Air Pollution
Maternal smoking during pregnancy is associated with an increased risk of childhood overweight BMI. Less is known about the association between secondhand tobacco smoke (SHS) exposure and childhood BMI. Various studies examined children who were born to families with active and prenatal tobacco smoke exposure.85, 156, 157, 158, 159 These studies found that SHS was positively associated with increased BMI when exposed children were compared to unexposed children. These studies went on to suggest that exposure to SHS in a child’s environment, especially in the first year, appears to be a key risk factor for a child becoming overweight.85, 156, 157, 158, 159
Air pollution (such as PAHs, ozone, PM2.5 and PM10, carbon monoxide (CO), nitrogen oxides (NOx)) has been linked to various adverse health outcomes such as asthma, cardiovascular disease, inflammatory responses, neurocognitive difficulties, and obesity.160, 161 These adverse health outcomes may affect a child’s ability to engage in physical activities, which may impact his/her weight.162 Prenatal air pollution exposures (PAHs, NOx, CO, PM) associated with birthweight have found either no associations or low birthweight, except for one report by Rundle et al., where prenatal exposures to PAHs were associated with obesity at age 7 years.160, 161, 163, 164, 165, 166, 167, 168, 169, 170 Jerrett et al.162, 171 examined the relationship between BMI levels in children and traffic density, air pollutants, and proximity of roadways to residential areas. In both studies, exposure to air pollutants resulted in increased BMI levels.
Discussion
Obesity is a critical health issue affecting millions of children worldwide, with the number of overweight or obese pre-school aged children projected to increase to approximately 60 million by 2020 (ref. 2). This review illustrates the myriad plausible contributors to childhood obesity. Literature discussing various chemical and non-chemical stressors associated with childhood obesity was collected and categorized into individual, family, community, or chemical exposures from the total environment where children live, learn, and play. When studies of chemical and non-chemical stressors were compared, inconclusive or contradictory results were sometimes observed, suggesting that obesity is a complex problem and the scientific community is far from finding evidence to support a single stressor responsible for childhood obesity.172, 173
The vast majority of obesity research focuses on non-chemical stressors such as SES, diet, the built environment, and activity. Figure 2 illustrates that 67% of the articles in this literature review focused on non-chemical stressors and, in general, reported a causal link between one non-chemical stressor of interest and obesity. Some studies do account for non-chemical stressor confounders, but lack data on chemical exposures that occur within a particular environment. For instance, parental smoking status is measured in many studies of non-chemical stressors as a surrogate for chemical exposure. While exposure to the chemicals in cigarette smoke is important, they are certainly not the only chemical agents to which children are exposed across their life stages.
Over the last decade, much research has considered the role of obesogens in affecting childhood obesity. A common theme is that obesogens are ubiquitous, man-made chemicals with multiple routes of exposure, acting on the same metabolic pathways, suggesting the importance of cumulative exposures. Much of the research conducted for chemical responses employs animal studies, where outcomes and trans-generational responses can be observed in a relatively short period of time.127, 147, 148, 174, 175, 176 Based on the selection scheme detailed in this paper’s methodology, only 33% of the studies included in the literature review considered environmental chemical exposures that were not a consequence of prenatal cigarette smoke exposure or physician-prescribed medications. The most common chemical exposures included perfluorinated compounds, phthalates, and outdoor air pollution. As with much of the non-chemical stressor research, many of the chemical exposure studies included covariates such as SES, nutrition, and activity.
Research on chemical stressors as factors in childhood obesity is relatively new and new methods and technologies are being created to support this novel research. There are vast numbers of chemicals to which children are exposed in their total environment and the lack of information in the literature would suggest more research needs to focus on environmental exposures and childhood obesity. The literature focused on chemical exposures and obesity outcomes is limited, and often involves cross-sectional studies that are not necessarily designed to answer questions on cause and effect. Positive associations between exposures and obesity across all chemical and non-chemical stressors is difficult to observe due to inconsistencies in data collection, duration of exposure time, age at outcome measurement, study types, logistical challenges of early life exposures and measurements of similar stressors (diet, activity).
A few studies included more than one potential confounder and though they were able to capture a more complete picture of the exposure scenarios, the studies were cross-sectional by design.151 Complex cohort studies like CHAMACOS, the German Ulm Birth Cohort, and the Danish National Birth Cohort have used a more comprehensive approach, incorporating both chemical and non-chemical stressors such as diet, activity, television watching, SES, secondhand smoke, and maternal and infant weights. While this mix of chemical and non-chemical stressors is more indicative of real-world exposures, these studies examined chemical exposure only in utero or from cord blood—not continuously through the follow-up years.158, 177, 178, 179
Finding cumulative, multiplicative, and possibly antagonistic effects among the myriad stressors is daunting, but it is important whether we want to explore the argument that multiple stressors are at play in childhood obesity. It is important for researchers to obtain accurate and complete measurements to fill the data gaps so that we can better understand the onset of obesity and potential opportunities to evaluate interventions relevant to child-specific environments. Examining relationships among stressors may also determine which stressors could be easily modified by interventions to reduce obesogenic behaviors and exposures. Many stressors are interconnected, and their effects should be examined while accounting for multiple covariates, confounders, and effect modifiers.
To encourage integrated transdisciplinary thinking and hypothesis generation, researchers from diverse fields (such as medicine, genetics, behavioral and environmental sciences) need to work together. They should also join with those in economics, marketing, communications, and policy, alongside industry partners, urban planners, community leaders, and transportation designers to produce environments that are conducive to children’s health and well-being.180 Designing research to encompass all stressors related to obesity will be costly and logistically challenging, but robust prospective longitudinal and translational studies can begin to bridge existing knowledge gaps, and possibly find subtle causalities.
Many studies use cross-sectional designs because they are more cost effective, can take a snapshot of different population groups at a single point in time, and generate large amounts of data. However, these types of studies lack definite conclusions regarding cause and effect.46, 117, 140, 181, 182, 183, 184 On the other hand, while longitudinal studies follow children for a period of time, these types of studies may be limited by the use of retrospective data, parent-based reporting, limited measurements of other covariates, imperfect or missing measurements of confounders, gaps in data reporting, or selection bias.165, 185, 186 Cross-comparison studies may prove difficult due to varying study designs and ages of the children. This situation is especially likely for measurements made during adolescence, when the onset of puberty can modify responses.
If we hope to combat childhood obesity, large longitudinal cohort studies that begin in the prenatal period will be essential to understand the complex interrelationships between the myriad chemical and non-chemical stressors.187 Studies like the perinatal prospective longitudinal study at Duke University, the Newborn Epigenetic STudy (NEST), and the CHAMACOS study are representative of the types of longitudinal cohort studies needed to examine the effects of environmental exposures and nutrition, both in utero and during childhood.167, 188
Researchers suggest that childhood obesity may be an early adaptive response to epigenetic mechanisms that guide gene expression involved with energy balance through DNA methylation.189 Ongoing studies suggest that nutrition during pregnancy may affect the health status of children and may be passed down through generations. These epigenetic changes are thought to be explained by modifications in chemical markers that attach to susceptible loci in DNA.188, 189 Determination of these epigenetic pathways acting on genes and proteins and how they may encode could eventually lead to prevention and treatment options.190 When researchers propose this type of analysis, it will still be important to consider non-chemical stressors along with chemical agents to better understand epigenetic mechanisms that regulate metabolic states, and to determine if and when those factors incur changes leading to obesity.
This systematic review suggests that childhood obesity is multifactorial, where the interrelationships between chemical and non-chemical stressors may have an effect on a child’s health and well-being. An integrative approach needs to be implemented to examine the child from a holistic point of view. To accomplish this, researchers need to take into account the well-being of the child through various lifestages, from early child development through adolescence. Tulve et al.33 has designed a conceptual framework where a child’s health and well-being is influenced by all the components of the built, natural, and social environments. A child’s holistic health extends beyond a child’s physical and mental health to include emotions, learning and development, physical safety, relationships, and material well-being. Children’s health and well-being are ultimately the product of inherent factors (e.g., race, gender, genetics), as well as their activities and behaviors. However, as shown in this paper, health and well-being may be influenced by the interrelationships of chemical and non-chemical stressors found in the real-world environment where children live, learn, and play.
Limitations
A literature review of this magnitude is a tremendous undertaking, and while we strove to be systematic in our review, there is still the possibility that we missed relevant articles. For this review, the literature search was limited to PubMed and Google Scholar and covered the time period of January 2004 to July 2014. Only peer-reviewed literature was included in this review. Since our focus was on chemical and non-chemical stressors and the inter-relationship of these stressors in impacting child health and well-being (i.e., obesity), we did not include research published in non-life science fields (such as economics or political science).
Due to the vast number of stressors encountered in the literature, it was important to organize the stressors for easier reference. We recognize that other organizational methods could have been used to present the information but found separating them into four major categories that coincide with the conceptual framework outlined in Tulve et al.33 allowed for clear organization and focus to present the information. A holistic understanding of childhood obesity begins with the individual child and their behaviors/habits formed as a result of their surroundings. The second section contained family and social stressors that impact and influence children’s behaviors or exposures to which the child had no control. The third section incorporated the built and natural environments in the community, thereby encompassing their total environment. The final section incorporated all the environmental chemicals to which children may potentially be exposed.
Conclusions
This comprehensive review of the childhood obesity literature set out to examine the interrelationships between chemical and non-chemical stressors which may play a role in childhood obesity. This review shows how numerous chemical and non-chemical stressors are linked to childhood obesity, but does not claim to have exhausted the list of plausible contributors that are yet to be discovered or are in the early stages of being linked to the obesity epidemic. This review provides information on the complexities and possible interactions between chemical and non-chemical stressors in a child’s environment and how these impact childhood obesity. By better understanding the interactions of chemical and non-chemical stressors contributing to childhood overweight and obesity, we can begin to inform community decision makers on how to promote healthy, child-specific environments and provide solutions to improve overall community health and well-being.
References
World Health Organziation Global Status Report on Noncommunicable Diseases 2010. World Health Organization: Italy. 2010.
de Onis M, Blossner M, Borghi E . Global prevalence and trends of overweight and obesity among preschool children. Am J Clin Nutr 2010; 92 (5): 1257–1264.
Centers for Diesease Contol and Prevention. Overwight and obesity: childhood overweight and obesity 2012 [Division of Nutrition, Physical Activity, and Obesity, National Center for Chronic Disease Prevention and Health Promotion. Available at http://www.cdc.gov/obesity/childhood/index.html.
Ogden CL, Carroll MD, Curtin LR, McDowell MA, Tabak CJ, Flegal KM . Prevalence of overweight and obesity in the united states, 1999-2004. JAMA 2006; 295 (13): 1549–1555.
Ogden CL, Carroll MD, Kit BK, Flegal KM . Prevalence of obesity and trends in body mass index among us children and adolescents, 1999-2010. JAMA 2012; 307 (5): 483–490.
Trasande L, Attina TM, Blustein J . Association between urinary bisphenol A concentration and obesity prevalence in children and adolescents. JAMA 2012; 308 (11): 1113–1121.
La Merrill M, Birnbaum LS . Childhood obesity and environmental chemicals. Mount Sinai J Med N Y 2011; 78 (1): 22–48.
Grün F . Obesogens. Curr Opin Endocrinol Diabetes Obes 2010; 17 (5): 453–459.
Calarge CA, Acion L, Kuperman S, Tansey M, Schlechte JA . Weight gain and metabolic abnormalities during extended risperidone treatment in children and adolescents. J Child Adoles Psychopharmacol 2009; 19 (2): 101–109.
Fleten C, Nystad W, Stigum H, Skjaerven R, Lawlor DA, Davey Smith G et al. Parent-offspring body mass index associations in the Norwegian Mother and Child Cohort Study: a family-based approach to studying the role of the intrauterine environment in childhood adiposity. Am J Epidemiol 2012; 176 (2): 83–92.
Gubbels JS, van Assema P, Kremers SP . Physical activity, sedentary behavior, and dietary patterns among children. Curr Nutr Rep 2013; 2 (2): 105–112.
Ino T . Maternal smoking during pregnancy and offspring obesity: meta-analysis. Pediatr Int 2010; 52 (1): 94–99.
Kwon S, Janz KF, Burns TL, Levy SM . Effects of adiposity on physical activity in childhood: Iowa Bone Development Study. Med Sci Sports Exerc 2011; 43 (3): 443–448.
Whitaker RC, Phillips SM, Orzol SM, Burdette HL . The association between maltreatment and obesity among preschool children. Child Abuse Neglect 2007; 31 (11-12): 1187–1199.
Grün F . Obesogens. Curr Opin Endocrinol Diabetes Obes 2010; 17 (5): 453–459.
Thayer KA, Heindel JJ, Bucher JR, Gallo MA . Role of environmental chemicals in diabetes and obesity: a National Toxicology Program Workshop Review. Environ Health Perspect 2012; 120 (6): 779–789.
Biro FM, Wien M . Childhood obesity and adult morbidities. Am J Clin Nutr 2010; 91 (5): 1499S–1505SS.
Davison KK, Francis LA, Birch LL . Reexamining obesigenic families: parents' obesity-related behaviors predict girls' change in BMI. Obes Res 2005; 13 (11): 1980–1990.
Hesketh KD, Hinkley T, Campbell KJ . Children's physical activity and screen time: qualitative comparison of views of parents of infants and preschool children. Int J Behav Nutr Phys Activity 2012; 9: 152.
Kestens Y, Lebel A, Chaix B, Clary C, Daniel M, Pampalon R et al. Association between activity space exposure to food establishments and individual risk of overweight. PLoS ONE 2012; 7 (8): e41418.
Perez A, Hoelscher DM, Springer AE, Brown HS, Barroso CS, Kelder SH et al. Physical activity, watching television, and the risk of obesity in students, Texas, 2004-2005. Prev Chronic Dis 2011; 8 (3): A61.
Pradinuk M, Chanoine JP, Goldman RD . Obesity and physical activity in children. Canad Fam Phys Med Fam Can 2011; 57 (7): 779–782.
Wojcicki JM, Heyman MB . Let's move — childhood obesity prevention from pregnancy and infancy onward. N Engl J Med 2010; 362 (16): 1457–1459.
U.S. EPA. Framework for Cumulative Risk Assessment. U.S. Government Printing Office: Washington, DC. 2003.
Gundersen C, Mahatmya D, Garasky S, Lohman B . Linking psychosocial stressors and childhood obesity. Obes Rev 2011; 12 (5): e54–e63.
Lewis AS, Sax SN, Wason SC, Campleman SL . Non-chemical stressors and cumulative risk assessment: an overview of current initiatives and potential air pollutant interactions. Int J Environ Res Public Health 2011; 8 (6): 2020–2073.
Dancause KN, Laplante DP, Fraser S, Brunet A, Ciampi A, Schmitz N et al. Prenatal exposure to a natural disaster increases risk for obesity in 5(1/2)-year-old children. Pediatr Res 2012; 71 (1): 126–131.
Cheung WW, Mao P . Recent advances in obesity: genetics and beyond. ISRN Endocrinol 2012; 2012: 536905.
Holtcamp W . Obesogens: an environmental link to obesity. Environ Health Perspect 2012; 120 (2): a62–a68.
U.S. EPA. Guidance on selecting age groups for monitoring and assessing childhood exposures to environmental contaminants. In: National Center for Environmental Assessment, editor. Washington, DC, USA 2005.
Araujo R, Dyer RS, Fortmann RC, Fulk FA, Hauchman FS, Heggem DT et al A Conceptual Framework for U.S. EPA’s National Exposure Research Laboratory. Washington, DC: U.S. Environmental Protection Agency, 2009 Contract No.: EPA/600/R-09/003 (NTIS PB2010-103951).
U.S. EPA. Framework for Ecological Risk Assessment. U.S. Government Printing Office: Washington, DC. 1992.
Tulve NS, Ruiz JDC, Lichtveld K, Darney SP, Quackenboss JJ . Development of a conceptual framework depicting a child’s total (built, natural, social) environment in order to optimize health and well-being. J Environ Health Sci 2016; 2 (2): 1–8.
Grow HM, Cook AJ, Arterburn DE, Saelens BE, Drewnowski A, Lozano P . Child obesity associated with social disadvantage of children's neighborhoods. Soc Sci Med (1982) 2010; 71 (3): 584–591.
Rahman T, Cushing RA, Jackson RJ . Contributions of built environment to childhood obesity. Mount Sinai J Med N Y 2011; 78 (1): 49–57.
Aitsi-Selmi A, Batty GD, Barbieri MA, Silva AA, Cardoso VC, Goldani MZ et al. Childhood socioeconomic position, adult socioeconomic position and social mobility in relation to markers of adiposity in early adulthood: evidence of differential effects by gender in the 1978/79 Ribeirao Preto cohort study. Int J Obes (2005) 2013; 37 (3): 439–447.
Hallal PC, Clark VL, Assuncao MC, Araujo CL, Goncalves H, Menezes AM et al. Socioeconomic trajectories from birth to adolescence and risk factors for noncommunicable disease: prospective analyses. J Adolesc Health 2012; 51 (6 Suppl): S32–S37.
Qazi HA . Childhood obesity and parks and playgrounds: a review of issues of equality, gender and social support. J Res Med Sci 2011; 16 (4): 553–558.
Brophy S, Cooksey R, Lyons RA, Thomas NE, Rodgers SE, Gravenor MB . Parental factors associated with walking to school and participation in organised activities at age 5: analysis of the Millennium Cohort Study. BMC Public Health 2011; 11: 14.
Kendzor DE, Caughy MO, Owen MT . Family income trajectory during childhood is associated with adiposity in adolescence: a latent class growth analysis. BMC Public Health 2012; 12: 611.
Voorhees CC, Catellier DJ, Ashwood JS, Cohen DA, Rung A, Lytle L et al. Neighborhood socioeconomic status and non school physical activity and body mass index in adolescent girls. J Phys Activity Health 2009; 6 (6): 731–740.
Wells NM, Evans GW, Beavis A, Ong AD . Early childhood poverty, cumulative risk exposure, and body mass index trajectories through young adulthood. Am J Public Health 2010; 100 (12): 2507–2512.
Knai C, Lobstein T, Darmon N, Rutter H, McKee M . Socioeconomic patterning of childhood overweight status in Europe. Int J Environ Res Public Health 2012; 9 (4): 1472–1489.
Ruijsbroek A, Wijga AH, Kerkhof M, Koppelman GH, Smit HA, Droomers M . The development of socio-economic health differences in childhood: results of the Dutch longitudinal PIAMA birth cohort. BMC Public Health 2011; 11: 225.
Kleiser C, Schaffrath Rosario A, Mensink GB, Prinz-Langenohl R, Kurth BM . Potential determinants of obesity among children and adolescents in Germany: results from the cross-sectional KiGGS Study. BMC Public Health 2009; 9: 46.
Morgenstern M, Sargent JD, Hanewinkel R . Relation between socioeconomic status and body mass index: evidence of an indirect path via television use. Arch Pediatr Adolesc Med 2009; 163 (8): 731–738.
Morgen CS, Mortensen LH, Rasmussen M, Andersen AM, Sorensen TI, Due P . Parental socioeconomic position and development of overweight in adolescence: longitudinal study of Danish adolescents. BMC Public Health 2010; 10: 520.
Morrissey TW, Dunifon RE, Kalil A . Maternal employment, work schedules, and children's body mass index. Child Dev 2011; 82 (1): 66–81.
Chen AY, Escarce JJ . Family structure and childhood obesity, early childhood longitudinal study — kindergarten cohort. Prev Chronic Dis 2010; 7 (3): A50.
Weden MM, Brownell P, Rendall MS . Prenatal, perinatal, early life, and sociodemographic factors underlying racial differences in the likelihood of high body mass index in early childhood. Am J Public Health 2012; 102 (11): 2057–2067.
Wang Y, Zhang Q . Are American children and adolescents of low socioeconomic status at increased risk of obesity? Changes in the association between overweight and family income between 1971 and 2002. Am J Clin Nutr 2006; 84: 707–716.
Whitaker RC, Orzol SM . Obesity among US urban preschool children: relationships to race, ethnicity, and socioeconomic status. Arch Pediatr Adolesc Med 2006; 160 (6): 578–584.
Balistreri KS, Van Hook J . Socioeconomic status and body mass index among hispanic children of immigrants and children of natives. Am J Public Health 2009; 99 (12): 2238–2246.
de Hoog ML, van Eijsden M, Stronks K, Gemke RJ, Vrijkotte TG . Overweight at age two years in a multi-ethnic cohort (ABCD study): the role of prenatal factors, birth outcomes and postnatal factors. BMC Public Health 2011; 11: 611.
Singh GK, Kogan MD, Van Dyck PC, Siahpush M . Racial/ethnic, socioeconomic, and behavioral determinants of childhood and adolescent obesity in the united states: analyzing independent and joint associations. Ann Epidemiol. 2008; 18: 682–695.
Ingstrup KG, Schou Andersen C, Ajslev TA, Pedersen P, Sorensen TI, Nohr EA . Maternal distress during pregnancy and offspring childhood overweight. J Obes 2012; 2012: 462845.
Van Dijk AE, Van Eijsden M, Stronks K, Gemke RJ, Vrijkotte TG . The relation of maternal job strain and cortisol levels during early pregnancy with body composition later in the 5-year-old child: the ABCD study. Early Hum Dev 2012; 88 (6): 351–356.
Li J, Olsen J, Vestergaard M, Obel C, Baker JL, Sorensen TI . Prenatal stress exposure related to maternal bereavement and risk of childhood overweight. PLoS ONE 2010; 5 (7): e11896.
Suglia SF, Duarte CS, Chambers EC, Boynton-Jarrett R . Cumulative social risk and obesity in early childhood. Pediatrics 2012; 129 (5): e1173–e1179.
Parks EP, Kumanyika S, Moore RH, Stettler N, Wrotniak BH, Kazak A . Influence of stress in parents on child obesity and related behaviors. Pediatrics 2012; 130 (5): e1096–e1104.
Gundersen C, Lohman BJ, Garasky S, Stewart S, Eisenmann J . Food security, maternal stressors, and overweight among low-income US children: results from the National Health and Nutrition Examination Survey (1999-2002). Pediatrics 2008; 122 (3): e529–e540.
Koch FS, Sepa A, Ludvigsson J . Psychological stress and obesity. J Pediatr 2008; 153 (6): 839–844.
Moens E, Braet C, Bosmans G, Rosseel Y . Unfavourable family characteristics and their associations with childhood obesity: a cross-sectional study. Eur Eating Disord Rev 2009; 17 (4): 315–323.
Stenhammar C, Olsson G, Bahmanyar S, Hulting AL, Wettergren B, Edlund B et al. Family stress and BMI in young children. Acta Paediatr 2010; 99 (8): 1205–1212.
Lohman BJ, Stewart S, Gundersen C, Garasky S, Eisenmann JC . Adolescent overweight and obesity: links to food insecurity and individual, maternal, and family stressors. J Adolesc Health 2009; 45 (3): 230–237.
Giles LC, Whitrow MJ, Rumbold AR, Davies CE, de Stavola B, Pitcher JB et al. Growth in early life and the development of obesity by age 9 years: are there critical periods and a role for an early life stressor? Int J Obes 2013; 37 (4): 513–519.
Midei AJ, Matthews KA . Interpersonal violence in childhood as a risk factor for obesity: a systematic review of the literature and proposed pathways. Obes Rev 2011; 12 (5): e159–e172.
Garasky S, Stewart SD, Gundersen C, Lohman BJ, Eisenmann JC . Family stressors and child obesity. Soc Sci Res 2009; 38: 755–766.
Blaine B . Does depression cause obesity?: A meta-analysis of longitudinal studies of depression and weight control. J Health Psychol 2008; 13 (8): 1190–1197.
Sweeting H, Wright C, Minnis H . Psychosocial correlates of adolescent obesity, ‘slimming down’ and ‘becoming obese’. J Adolesc Health 2005; 37 (5): 409.
Vanaelst B, Michels N, Clays E, Herrmann D, Huybrechts I, Sioen I et al. The association between childhood stress and body composition, and the role of stress-related lifestyle factors-cross-sectional findings from the Baseline ChiBS Survey. Int J Behav Med 2013; 21 (2): 292–301.
van Jaarsveld CHM, Fidler JA, Steptoe A, Boniface D, Wardle J . Perceived stress and weight gain in adolescence: a longitudinal analysis. Obesity 2009; 17 (12): 2155–2161.
Rofey DL, Kolko RP, Iosif AM, Silk JS, Bost JE, Feng W et al. A longitudinal study of childhood depression and anxiety in relation to weight gain. Child Psychiatry Hum Dev 2009; 40 (4): 517–526.
Fraser A, Tilling K, Macdonald-Wallis C, Sattar N, Brion MJ, Benfield L et al. Association of maternal weight gain in pregnancy with offspring obesity and metabolic and vascular traits in childhood. Circulation 2010; 121 (23): 2557–2564.
Oken E, Taveras EM, Kleinman KP, Rich-Edwards JW, Gillman MW . Gestational weight gain and child adiposity at age 3 years. Am J Obstet Gynecol 2007; 196 (4): 322.e1–.e8.
Karaolis-Danckert N, Buyken AE, Kulig M, Kroke A, Forster J, Kamin W et al. How pre- and postnatal risk factors modify the effect of rapid weight gain in infancy and early childhood on subsequent fat mass development: results from the Multicenter Allergy Study 90. Am J Clin Nutr 2008; 87 (5): 1356–1364.
Smith GD, Steer C, Leary S, Ness A . Is there an intrauterine influence on obesity? Evidence from parent-child associations in the Avon Longitudinal Study of Parents and Children (ALSPAC). Arch Dis Childhood 2007; 92 (10): 876–880.
Matijasevich A, Brion MJ, Menezes AM, Barros AJD, Santos IS, Barros FC . Maternal smoking during pregnancy and offspring growth in childhood: 1993 and 2004 Pelotas cohort studies. Arch Dis Childhood 2011; 96 (6): 519–525.
Shi Y, De Groh M, Morrison H . Perinatal and early childhood factors for overweight and obesity in young canadian children. Can J Public Health Revue Can Sante Public 2013; 104 (1): e69–e74.
Gorog K, Pattenden S, Antova T, Niciu E, Rudnai P, Scholtens S et al. Maternal smoking during pregnancy and childhood obesity: results from the CESAR Study. Matern Child Health J 2011; 15 (7): 985–992.
Ino T, Shibuya T, Saito K, Ohtani T . Effects of maternal smoking during pregnancy on body composition in offspring. Pediatr Int 2011; 53 (6): 851–857.
Koshy G, Delpisheh A, Brabin BJ . Dose response association of pregnancy cigarette smoke exposure, childhood stature, overweight and obesity. Eur J Public Health 2011; 21 (3): 286–291.
Oken E, Huh SY, Taveras EM, Rich-Edwards JW, Gillman MW . Associations of maternal prenatal smoking with child adiposity and blood pressure. Obes Res 2005; 13 (11): 2021–2028.
Oken E, Levitan E, Gillman M . Maternal smoking during pregnancy and child overweight: systematic review and meta-analysis. Int J Obes 2008; 32 (2): 201–210.
Raum E, Kupper-Nybelen J, Lamerz A, Hebebrand J, Herpertz-Dahlmann B, Brenner H . Tobacco smoke exposure before, during, and after pregnancy and risk of overweight at age 6Obesity2011; 19 (12): 2411–2417.
Suzuki K, Sato M, Ando D, Kondo N, Yamagata Z . Differences in the effect of maternal smoking during pregnancy for childhood overweight before and after 5 years of age. J Obstetr Gynaecol Res 2013; 39 (5): 914–921.
Wen X, Shenassa ED, Paradis AD . Maternal smoking, breastfeeding, and risk of childhood overweight: findings from a national cohort. Matern Child Health J 2013; 17 (4): 746–755.
Leary SD, Smith GD, Rogers IS, Reilly JJ, Wells JC, Ness AR . Smoking during pregnancy and offspring fat and lean mass in childhood. Obesity 2006; 14 (12): 2284–2293.
Mamun AA, O'Callaghan MJ, Williams GM, Najman JM . Maternal smoking during pregnancy predicts adult offspring cardiovascular risk factors - evidence from a community-based large birth cohort study. PLoS ONE 2012; 7 (7): e41106.
Salsberry PJ, Reagan PB . Dynamics of early childhood overweight. Pediatrics 2005; 116 (6): 1329–1338.
Janjua NZ, Mahmood B, Islam MA, Goldenberg RL . Maternal and early childhood risk factors for overweight and obesity among low-income predominantly black children at age five years: a Prospective Cohort Study. J Obes 2012; 2012: 457173.
Howe LD, Matijasevich A, Tilling K, Brion MJ, Leary SD, Smith GD et al. Maternal smoking during pregnancy and offspring trajectories of height and adiposity: comparing maternal and paternal associations. Int J Epidemiol. 2012; 41 (3): 722–732.
von Kries R, Bolte G, Baghi L, Toschke AM . Parental smoking and childhood obesity—is maternal smoking in pregnancy the critical exposure? Int J Epidemiol 2008; 37 (1): 210–216.
Ferdinand AO, Sen B, Rahurkar S, Engler S, Menachemi N . The relationship between built environments and physical activity: a systematic review. Am J Public Health 2012; 102 (10): e7–e13.
Miranda ML, Edwards SE, Anthopolos R, Dolinsky DH, Kemper AR . The built environment and childhood obesity in Durham, North Carolina. Clin Pediatr 2012; 51 (8): 750–758.
Carter MA, Dubois L, Tremblay MS, Taljaard M, Jones BL . Trajectories of childhood weight gain: the relative importance of local environment versus individual social and early life factors. PLoS ONE 2012; 7 (10): e47065.
Bell JF, Wilson JS, Liu GC . Neighborhood greenness and 2-year changes in body mass index of children and youth. Am J Prevent Med 2008; 35 (6): 547–553.
Dulin-Keita A, Kaur Thind H, Affuso O, Baskin ML . The associations of perceived neighborhood disorder and physical activity with obesity among African American adolescents. BMC Public Health 2013; 13: 440.
Edwards KL, Clarke GP, Ransley JK, Cade J . The neighbourhood matters: studying exposures relevant to childhood obesity and the policy implications in Leeds, UK. J Epidemiol Commun Health 2010; 64 (3): 194–201.
Franzini L, Elliott MN, Cuccaro P, Schuster M, Gilliland MJ, Grunbaum JA et al. Influences of physical and social neighborhood environments on children's physical activity and obesity. Am J Public Health 2009; 99 (2): 271–278.
Geoffroy MC, Power C, Touchette E, Dubois L, Boivin M, Seguin JR et al. Childcare and overweight or obesity over 10 years of follow-up. J Pediatrics 2013; 162 (4): 753–8 e1.
Lovasi GS, Jacobson JS, Quinn JW, Neckerman KM, Ashby-Thompson MN, Rundle A . Is the environment near home and school associated with physical activity and adiposity of urban preschool children? J Urban Health Bull N Y Acad Med 2011; 88 (6): 1143–1157.
Singh GK, Siahpush M, Kogan MD . Neighborhood socioeconomic conditions, built environments, and childhood obesity. Health Affairs 2010; 29 (3): 503–512.
Wall MM, Larson NI, Forsyth A, Van Riper DC, Graham DJ, Story MT et al. Patterns of obesogenic neighborhood features and adolescent weight: a comparison of statistical approaches. Am J Prevent Med 2012; 42 (5): e65–e75.
Benjamin SE, Rifas-Shiman SL, Taveras EM, Haines J, Finkelstein J, Kleinman K et al. Early child care and adiposity at ages 1 and 3 years. Pediatrics 2009; 124 (2): 555–562.
Crawford DA, Timperio AF, Salmon JA, Baur L, Giles-Corti B, Roberts RJ et al. Neighbourhood fast food outlets and obesity in children and adults: the CLAN Study. Pediatr Obes 2008; 3 (4): 249–256.
Galvez MP, Hong L, Choi E, Liao L, Godbold J, Brenner B . Childhood obesity and neighborhood food store availability in an inner city community. Acad Pediatr 2009; 9 (5): 339–343.
Leatherdale ST, Pouliou T, Church D, Hobin E . The association between overweight and opportunity structures in the built environment: a multi-level analysis among elementary school youth in the PLAY-ON study. Int J Public Health 2011; 56 (3): 237–246.
Lovasi GS, Hutson MA, Guerra M, Neckerman KM . Built environments and obesity in disadvantaged populations. Epidemiol Rev 2009; 31: 7–20.
Pearce JR, Maddison R . Do enhancements to the urban built environment improve physical activity levels among socially disadvantaged populations? Int J Equity Health 2011; 10: 28.
Sánchez BN, Sanchez-Vaznaugh EV, Uscilka A, Baek J, Zhang L . Differential associations between the food environment near schools and childhood overweight across race/ethnicity, gender, and grade. Am J Epidemiol 2011; 175 (12): 1284–1293.
Osei-Assibey G, Dick S, Macdiarmid J, Semple S, Reilly JJ, Ellaway A et al. The influence of the food environment on overweight and obesity in young children: a systematic review. BMJ Open 2012; 2 (6): p e001538.
Laska MN, Hearst MO, Forsyth A, Pasch KE, Lytle L . Neighbourhood food environments: are they associated with adolescent dietary intake, food purchases and weight status? Public Health Nutr 2010; 13 (11): 1757–1763.
Leung CW, Laraia BA, Kelly M, Nickleach D, Adler NE, Kushi LH et al. The influence of neighborhood food stores on change in young girls' body mass index. Am J Prevent Med 2011; 41 (1): 43–51.
Shier V, An R, Sturm R . Is there a robust relationship between neighbourhood food environment and childhood obesity in the USA? Public Health 2012; 126 (9): 723–730.
Wolch J, Jerrett M, Reynolds K, McConnell R, Chang R, Dahmann N et al. Childhood obesity and proximity to urban parks and recreational resources: a longitudinal cohort study. Health Place 2011; 17 (1): 207–214.
Acharya K, Feese M, Franklin F, Kabagambe EK . Body mass index and dietary intake among head start children and caregivers. J Am Diet Assoc 2011; 111 (9): 1314–1321.
Epstein L, Raja S, Daniel T, Paluch R, Wilfley D, Saelens B et al. The built environment moderates effects of family-based childhood obesity treatment over 2 years. Ann Behav Med 2012; 44 (2): 248–258.
Roemmich JN, Epstein LH, Raja S, Yin L, Robinson J, Winiewicz D . Association of access to parks and recreational facilities with the physical activity of young children. Prevent Med 2006; 43 (6): 437–441.
Potwarka L, Kaczynski A, Flack A . Places to play: association of park space and facilities with healthy weight status among children. J Commun Health 2008; 33 (5): 344–350.
Potestio ML, Patel AB, Powell CD, McNeil DA, Jacobson RD, McLaren L . Is there an association between spatial access to parks/green space and childhood overweight/obesity in Calgary Canada? Int J Behav NutrPhys Activity 2009; 6: 77.
McCurdy LE, Winterbottom KE, Mehta SS, Roberts JR . Using nature and outdoor activity to improve children's health. Curr Problems Pediatr Adolesc Health Care 2010; 40 (5): 102–117.
Cleland V, Crawford D, Baur LA, Hume C, Timperio A, Salmon J . A prospective examination of children's time spent outdoors, objectively measured physical activity and overweight. Int J Obes 2008; 32 (11): 1685–1693.
Decherf S, Demeneix BA . The obesogen hypothesis: a shift of focus from the periphery to the hypothalamus. J Toxicol Environ Health B Crit Rev 2011; 14 (5-7): 423–448.
Blumberg B . Obesogens, stem cells and the maternal programming of obesity. J Dev Origins Health Dis 2011; 2 (1): 3–8.
Janesick A, Blumberg B . Obesogens, stem cells and the developmental programming of obesity. Int J Androl 2012; 35 (3): 437–448.
Grün F, Blumberg B . Perturbed nuclear receptor signaling by environmental obesogens as emerging factors in the obesity crisis. Rev Endocr Metab Disord 2007; 8 (2): 161–171.
Schug TT, Janesick A, Blumberg B, Heindel JJ . Endocrine disrupting chemicals and disease susceptibility. J Steroid Biochem Mol Biol. 2011; 127 (3-5): 204–215.
Akyurek N, Aycan Z, Cetinkaya S, Akyurek O, Yilmaz Agladioglu S, Ertan U . Peroxisome proliferator activated receptor (PPAR)-gamma concentrations in childhood obesity. Scand J Clin Lab Invest 2013.
Janesick A, Blumberg B . Minireview: PPAR gamma as the target of obesogens. J Steroid Biochem Mol Biol 2011; 127 (1-2): 4–8.
le Maire A, Grimaldi M, Roecklin D, Dagnino S, Vivat-Hannah V, Balaguer P et al. Activation of RXR-PPAR heterodimers by organotin environmental endocrine disruptors. EMBO Rep 2009; 10 (4): 367–373.
Rees WD, McNeil CJ, Maloney CA . The roles of PPARs in the fetal origins of metabolic health and disease. PPAR Res 2008; 2008: 459030.
Iguchi T, Katsu Y, Horiguchi T, Watanabe H, Blumberg B, Ohta Y . Endocrine disrupting organotin compounds are potent inducers of imposex in gastropods and adipogenesis in vertebrates. Mol Cell Toxicol 2007; 3 (1): 1–10.
Sousa AC, Pastorinho MR, Takahashi S, Tanabe S . History on organotin compounds, from snails to humans. Environ Chem Lett 2014; 12 (1): 117–137.
Kotake Y . Molecular mechanisms of environmental organotin toxicity in mammals. Biol Pharm Bull 2012; 35 (11): 1876–1880.
Padilla MA, Elobeid M, Ruden DM, Allison DB . An examination of the association of selected toxic metals with total and central obesity indices: NHANES 99-02. Int J Environ Res Public Health 2010; 7 (9): 3332–3347.
Afeiche M, Peterson KE, Sanchez BN, Cantonwine D, Lamadrid-Figueroa H, Schnaas L et al. Prenatal lead exposure and weight of 0- to 5-year-old children in mexico city. Environ Health Perspect 2011; 119 (10): 1436–1441.
Kippler M, Tofail F, Gardner R, Rahman A, Hamadani JD, Bottai M et al. Maternal cadmium exposure during pregnancy and size at birth: a prospective cohort study. Environ Health Perspect 2012; 120 (2): 284–289.
Alonso-Magdalena P, Nadal A Fetal and adult exposure to bisphenol-a as a contributing factor in the etiology of the metabolic syndrome. In: Bourguignon J-P, Jégou B, Kerdelhué B, Toppari J, Christen Y eds. Multi-System Endocrine Disruption. Research and Perspectives in Endocrine Interactions. Springer: Berlin Heidelberg. 2011 p 113–125.
Harley KG, Aguilar Schall R, Chevrier J, Tyler K, Aguirre H, Bradman A et al. Prenatal and postnatal bisphenol a exposure and body mass index in childhood in the CHAMACOS Cohort. Environ Health Perspect 2013; 121 (4): 514–520.
Yamano Y, Miyakawa S, Iizumi K, Itoh H, Iwasaki M, Tsugane S et al. Long-term study of urinary bisphenol A in elementary school children. Environ Health Prevent Med 2008; 13 (6): 332–337.
Li D-K, Miao M, Zhou Z, Wu C, Shi H, Liu X et al. Urine bisphenol-a level in relation to obesity and overweight in school-age children. PLoS ONE 2013; 8 (6): e65399.
Wang HX, Zhou Y, Tang CX, Wu JG, Chen Y, Jiang QW . Association between bisphenol A exposure and body mass index in Chinese school children: a cross-sectional study. Environ Health 2012; 11: 79.
Bhandari R, Xiao J, Shankar A . Urinary Bisphenol A and obesity in US children. Am J Epidemiol 2013; 177 (11): 1263–1270.
Hatch EE, Nelson JW, Qureshi MM, Weinberg J, Moore LL, Singer M et al. Association of urinary phthalate metabolite concentrations with body mass index and waist circumference: a cross-sectional study of NHANES data, 1999-2002. Environ Health 2008; 7: 27.
Meeker JD, Sathyanarayana S, Swan SH . Phthalates and other additives in plastics: human exposure and associated health outcomes. Philos Transac R Soc B Biol Sci 2009; 364 (1526): 2097–2113.
Ellero-Simatos S, Claus SP, Benelli C, Forest C, Letourneur F, Cagnard N et al. Combined transcriptomic-(1)H NMR metabonomic study reveals that monoethylhexyl phthalate stimulates adipogenesis and glyceroneogenesis in human adipocytes. J Proteome Res 2011; 10 (12): 5493–5502.
Hao C, Cheng X, Xia H, Ma X . The endocrine disruptor mono-(2-ethylhexyl) phthalate promotes adipocyte differentiation and induces obesity in mice. Biosci Rep 2012; 32: 619–629.
Grün F, Blumberg B . Perturbed nuclear receptor signaling by environmental obesogens as emerging factors in the obesity crisis. Rev Endocr Metab Disord 2007; 8: 161–171.
Hatch EE, Nelson JW, Stahlhut RW, Webster TF . Association of endocrine disruptors and obesity: perspectives from epidemiological studies. Int J Androl 2010; 33 (2): 324–332.
Trasande L, Attina TM, Sathyanarayana S, Spanier AJ, Blustein J . Race/Ethnicity-specific associations of urinary phthalates with childhood body mass in a nationally representative sample. Environ Health Perspect 2013; 121 (4): 501–506.
Philippat C, Mortamais M, Chevrier C, Petit C, Calafat AM, Ye X et al. Exposure to phthalates and phenols during pregnancy and offspring size at birth. Environ Health Perspect 2012; 120 (3): 464–470.
Teitelbaum SL, Mervish N, Moshier E, Vangeepuram N, Galvez MP, Calafat A et al. Associations between phthalate metabolite urinary concentrations and body size measures in New York City children. Environ Res 2012; 112: 186–193.
Wolff MS, Engel SM, Berkowitz GS, Ye X, Silva MJ, Zhu C et al. Prenatal phenol and phthalate exposures and birth outcomes. Environ Health Perspect 2008; 116 (8): 1092–1097.
Wang H, Zhou Y, Tang C, He Y, Wu J, Chen Y et al. Urinary phthalate metabolites are associated with body mass index and waist circumference in Chinese school children. PLoS ONE 2013; 8 (2): e56800.
Apfelbacher CJ, Loerbroks A, Cairns J, Behrendt H, Ring J, Kramer U . Predictors of overweight and obesity in five to seven-year-old children in Germany: results from cross-sectional studies. BMC Public Health 2008; 8: 171.
Braun JM, Daniels JL, Poole C, Olshan AF, Hornung R, Bernert JT et al. Prenatal environmental tobacco smoke exposure and early childhood body mass index. Paediatr Perinat Epidemiol 2010; 24 (6): 524–534.
Florath I, Kohler M, Weck MN, Brandt S, Rothenbacher D, Schottker B et al. Association of pre- and post-natal parental smoking with offspring body mass index: an 8-year follow-up of a birth cohort. Pediatr Obes 2013; 9 (2): 121–134.
Ittermann T, Thamm M, Schipf S, John U, Rettig R, Volzke H . Relationship of smoking and/or passive exposure to tobacco smoke on the association between serum thyrotropin and body mass index in large groups of adolescents and children. Thyroid 2013; 23 (3): 262–268.
Laurent O, Wu J, Li L, Chung J, Bartell S . Investigating the association between birth weight and complementary air pollution metrics: a cohort study. Environ Health 2013; 12 (1): 18.
van den Hooven EH, Jaddoe VW, de Kluizenaar Y, Hofman A, Mackenbach JP, Steegers EA et al. Residential traffic exposure and pregnancy-related outcomes: a prospective birth cohort study. Environ Health 2009; 8: 59.
Jerrett M, McConnell R, Chang CC, Wolch J, Reynolds K, Lurmann F et al. Automobile traffic around the home and attained body mass index: a longitudinal cohort study of children aged 10-18 years. Prevent Med 2010; 50 (Suppl 1): S50–S58.
Rundle A, Hoepner L, Hassoun A, Oberfield S, Freyer G, Holmes D et al. Association of childhood obesity with maternal exposure to ambient air polycyclic aromatic hydrocarbons during pregnancy. Am J Epidemiol 2012; 175 (11): 1163–1172.
Aguilera I, Guxens M, Garcia-Esteban R, Corbella T, Nieuwenhuijsen MJ, Foradada CM et al. Association between GIS-based exposure to urban air pollution during pregnancy and birth weight in the INMA Sabadell Cohort. Environ Health Perspect 2009; 117 (8): 1322–1327.
Bell ML, Belanger K, Ebisu K, Gent JF, Lee HJ, Koutrakis P et al. Prenatal exposure to fine particulate matter and birth weight: variations by particulate constituents and sources. Epidemiology 2010; 21 (6): 884–891.
Estarlich M, Ballester F, Aguilera I, Fernandez-Somoano A, Lertxundi A, Llop S et al. Residential exposure to outdoor air pollution during pregnancy and anthropometric measures at birth in a multicenter cohort in Spain. Environ Health Perspect 2011; 119 (9): 1333–1338.
Harley KG, Chevrier J, Aguilar Schall R, Sjodin A, Bradman A, Eskenazi B . Association of prenatal exposure to polybrominated diphenyl ethers and infant birth weight. Am J Epidemiol 2011; 174 (8): 885–892.
Mannes T, Jalaludin B, Morgan G, Lincoln D, Sheppeard V, Corbett S . Impact of ambient air pollution on birth weight in Sydney, Australia. Occup Environ Med 2005; 62 (8): 524–530.
Polanska K, Hanke W, Sobala W, Brzeznicki S, Ligocka D . Exposure to polycyclic aromatic hydrocarbons and newborn biometric indicators. Int J Occup Med Environ Health 2010; 23 (4): 339–346.
van den Hooven EH, Pierik FH, de Kluizenaar Y, Willemsen SP, Hofman A, van Ratingen SW et al. Air pollution exposure during pregnancy, ultrasound measures of fetal growth, and adverse birth outcomes: a prospective cohort study. Environ Health Perspect 2012; 120 (1): 150–156.
Jerrett M, McConnell R, Wolch J, Chang R, Lam C, Dunton G et al. Traffic-related air pollution and obesity formation in children: a longitudinal, multilevel analysis. Environ Health 2014; 13: 49.
Keith SW, Redden DT, Katzmarzyk PT, Boggiano MM, Hanlon EC, Benca RM et al. Putative contributors to the secular increase in obesity: exploring the roads less traveled. Int J Obes 2006; 30: 1585–1594.
Saunders T . Potential contributors to the Canadian pediatric obesity epidemic. ISRN Pediatrics 2011; 2011: 917684.
Irigaray P, Ogier V, Jacquenet S, Notet V, Sibille P, Méjean L et al. Benzo[a]pyrene impairs β-adrenergic stimulation of adipose tissue lipolysis and causes weight gain in mice. FASEB J 2006; 273 (7): 1362–1372.
Sun Q, Yue P, Deiuliis JA, Lumeng CN, Kampfrath T, Mikolaj MB et al. Ambient air pollution exaggerates adipose inflammation and insulin resistance in a mouse model of diet-induced obesity. Circulation 2009; 119: 538–546.
Xu X, Yavar Z, Verdin M, Ying Z, Mihai G, Kampfrath T et al. Effect of early particulate air pollution exposure on obesity in mice role of p47phox. Arterioscler Thromb Vasc Biol 2010; 30: 2518–2527.
Warner M, Aguilar Schall R, Harley KG, Bradman A, Barr D, Eskenazi B . In utero DDT and DDE exposure and obesity status of 7-year-old Mexican-American children in the CHAMACOS cohort. Environ Health Perspect 2013; 121 (5): 631–636.
Fei C, McLaughlin JK, Tarone RE, Olsen J . Fetal growth indicators and perfluorinated chemicals: a study in the Danish National Birth Cohort. Am J Epidemiol 2008; 168 (1): 66–72.
Halldorsson TI, Rytter D, Haug LS, Bech BH, Danielsen I, Becher G et al. Prenatal exposure to perfluorooctanoate and risk of overweight at 20 years of age: a Prospective Cohort Study. Environ Health Perspect 2012; 120 (5): 668–673.
NIH Strategic Plan for NIH Obesity Research: A Report of the NIH Obesity Task Force, In: National Institutes of Health USDoHaHS ed. NIH Publication: Bethesda, MD. 2011.
Björk J, Albin M, Grahn P, Jacobsson H, Ardö J, Wadbro J et al. Recreational values of the natural environment in relation to neighbourhood satisfaction, physical activity, obesity and wellbeing. J Epidemiol Community Health 2008; 62 (4): e2.
Carson V, Janssen I . Associations between factors within the home setting and screen time among children aged 0-5 years: a cross-sectional study. BMC Public Health 2012; 12: 539.
Lissner L, Lanfer A, Gwozdz W, Olafsdottir S, Eiben G, Moreno LA et al. Television habits in relation to overweight, diet and taste preferences in European children: the IDEFICS study. Eur J Epidemiol 2012; 27 (9): 705–715.
McDonald NC, Deakin E, Aalborg AE . Influence of the social environment on children's school travel. Prevent Med 2010; 50 (Supplement(0): S65–S68.
Birnbaum LS . Environmental chemicals: evaluating low-dose effects. Environ Health Perspect 2012; 120 (4): A143–A144.
McAllister EJ, Dhurandhar NV, Keith SW, Aronne LJ, Barger J, Baskin M et al. Ten putative contributors to the obesity epidemic. Crit Rev Food Sci Nutr. 2009; 49 (10): 868–913.
Trasande L, Cronk C, Durkin M, Weiss M, Schoeller DA, Gall EA et al. Environment and obesity in the National Children’s Study. Environ Health Perspect 2009; 117 (2): 159–166.
Hoyo C, Murtha AP, Schildkraut JM, Forman MR, Calingaert B, Demark-Wahnefried W et al. Folic acid supplementation before and during pregnancy in the Newborn Epigenetics STudy (NEST). BMC Public Health 2011; 11 (1): 46.
Perkins E, Murphy SK, Murtha AP, Schildkraut J, Jirtle RL, Demark-Wahnefried W et al. Insulin-like growth factor 2/H19 methylation at birth and risk of overweight and obesity in children. J Pediatr. 2012; 161 (1): 31–39.
US Department of Health and Human Services. Strategic plan for NIH obesity research: a report of the NIH Obesity Research Task Force. National Institutes of Health (NIH) Publication. 2004 Aug(04-5493):9..
Acknowledgements
This project was supported by an appointment to the Internship/Research Participation Program at the U.S. Environmental Protection Agency, Office of Research and Development, National Exposure Research Laboratory, Human Exposure and Atmospheric Sciences Division, Exposure Measurements and Analysis Branch, administered by the Oak Ridge Institute for Science and Education (ORISE) through an interagency agreement between the U.S. Department of Energy and EPA. It has been subjected to Agency administrative review and approved for publication. Mention of trade names or commercial products does not constitute endorsement or recommendation for use.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Journal of Exposure Science and Environmental Epidemiology website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Lichtveld, K., Thomas, K. & Tulve, N. Chemical and non-chemical stressors affecting childhood obesity: a systematic scoping review. J Expo Sci Environ Epidemiol 28, 1–12 (2018). https://doi.org/10.1038/jes.2017.18
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jes.2017.18
Keywords
This article is cited by
-
Early-life antibiotic exposure increases the risk of childhood overweight and obesity in relation to dysbiosis of gut microbiota: a birth cohort study
Annals of Clinical Microbiology and Antimicrobials (2022)
-
Distributions and determinants of time spent outdoors among school-age children in China
Journal of Exposure Science & Environmental Epidemiology (2022)
-
Exposure science in an infectious disease pandemic: who do we want to be?
Journal of Exposure Science & Environmental Epidemiology (2020)
-
Kynurenic acid as the neglected ingredient of commercial baby formulas
Scientific Reports (2019)
-
Joint Impact of Synthetic Chemical and Non-chemical Stressors on Children’s Health
Current Environmental Health Reports (2019)