Abstract
Phthalates and bisphenol A (BPA) are high production volume and ubiquitous chemicals that are quickly metabolized in the body. Traditionally, studies have relied on single spot urine analyses to assess exposure; ignoring variability in concentrations throughout a day or over a longer period of time. We compared BPA and phthalate metabolite results from urine samples collected at five different time points. Participants (n=80) were asked to collect all voids in a 24 h period on a weekday and then again on a weekend before 20 weeks of pregnancy. During the second and third trimesters and in the postpartum period, single spot urines were collected. Variability over time in urinary concentrations was assessed using intraclass correlation coefficients (ICCs) and the sensitivity to correctly classify a single sample as high or low versus the geometric mean (GM) of all samples was calculated. We found low reproducibility and sensitivity of BPA and all phthalate metabolites throughout pregnancy and into the postpartum period but much higher reproducibility within a day. Time of day when the urine was collected was a significant predictor of specific gravity adjusted exposure levels. We concluded that, if the interest is in average exposures across pregnancy, maternal/fetal exposure estimation may be more accurate if multiple measurements, collected across the course of the entire pregnancy, rather than a single spot measure, are performed.
Similar content being viewed by others
INTRODUCTION
Phthalates and bisphenol A (BPA) are high production volume man-made chemicals that are found in a number of industrial and consumer products and have widespread exposure in humans.1,2 BPA and some phthalates are suspected endocrine disruptors and have been associated with adverse reproductive and developmental effects in both animal3 and human4, 5, 6 studies.
Neither phthalates nor BPA bioaccumulate: they have very short half-lives in vivo, with most metabolites leaving the body within 24 h.7 Traditionally, urinary metabolite concentrations of phthalate metabolites and BPA have been measured from a single spot urine sample. Collecting a spot urine sample is relatively inexpensive and efficient, but ignores the diurnal and day-to-day variability of exposure to these ambient chemicals. In particular, during pregnancy there are both physiological and behavioral changes that could affect chemical concentrations and metabolism.8,9 To assess the potential reproductive toxicity of these chemicals, it is important to have valid and reliable measures of exposure. There are a few studies10, 11, 12, 13, 14, 15 that have examined the temporal variability of phthalate metabolites and BPA in pregnant women, but none have measured the variability within a day.
A major goal of this study was to examine intra-individual variability in urinary concentrations of BPA and phthalate metabolites, diurnally as well as throughout pregnancy, and during the post-partum period.
METHODS
Subjects
The Plastics and Personal-care Products use in Pregnancy (P4) study was approved by the research ethics boards at Health Canada and the Ottawa Hospital. After signing informed consent, 80 pregnant women were recruited prior to 20 weeks gestation from two obstetrical clinics in Ottawa, Canada between November 2009 and December 2010. Eligibility criteria included the ability to consent and communicate in English or French, age 18 years or older, gestation less than 20 weeks and planning on delivering at a local hospital. Women who had fetal abnormalities or major malformations in the current pregnancy, or had a history of medical complications (e.g., thyroid disorder, hypertension, diabetes and epilepsy), threat of spontaneous abortion or illicit drug use were excluded from the study.
Sampling Schedule
The sampling schedule is shown in Table 1. At the time of recruitment women were asked to collect all urine voids over a 24 h period, on a weekday (T1a) and/or a week-end day (T1b). Voids were collected in 120 ml Nalgene specimen cups and the time and date of each sample was recorded. Women were instructed to keep the urine cool at all times and were provided with the urine cups and a cooler bag with ice packs in order to avoid degradation of the target chemicals.16 A research assistant from the Ottawa Hospital visited the participants’ home to collect the urine samples and deliver them to the hospital lab where they were homogenized in a Vortex Mixer for 5 s and an aliquot removed and frozen at −80 °C within 36 h of collection. During the 2nd (T2; 24–28 weeks) and 3rd trimesters (T3; 32–36 weeks) and in the post-partum period (T5; 2-3 months), women were asked to provide a single spot urine sample (minimum 50 ml) at a regularly scheduled clinic visit or at home visit. These samples were also kept cool (refrigeration) and frozen within 36 h of collection. At each time point, participants completed questionnaires about their pregnancy, employment, smoking status and potential exposures. Samples of cups, storage vials and urine handling materials were evaluated prior to beginning the study and found not to be a source of sample contamination for any of the analytes being measured in this study.
Chemical Analysis
The P4 study chemical analysis was divided into two parts (see Table 2). Part I measured 11 different phthalate metabolites and BPA in urine collected at all study time points (T1a, T1b, T2, T3, T5) while Part II measured an additional 10 phthalate metabolites just in the 24 h serial spot urines (T1a, T1b) for a subset of participants who completed both visits (n=31). For urine measurements, three different methods were developed by the Laboratoire de toxicologie of the Institut national de santé publique du Québec (INSPQ). A gas chromatographic tandem mass spectrometric method was used for the measurement of BPA and ultra-performance liquid chromatography methods were used for the analysis of phthalate metabolites. The methods used isotope dilution standardization with radio-labeled or deuterated analogues for most of the compounds. The methods were fully validated using ISO 17025 guidelines. Limit of detection (LOD), limit of quantification (LOQ), linearity, accuracy, intra-day and inter-day precision, specificity and robustness assays were applied to the methods. Because of the ubiquity of the substances BPA and phthalate esters, labware and reagent chemicals were carefully treated to eliminate contamination during sample preparation and analysis. Labware was washed with organic solvent or baked (500 °C) while some reagents (e.g., carbonate buffer) were washed with organic solvents. Quality control samples (blank, low, medium and high concentration levels) were used for each analyte. Urine specific gravity (SG) was measured using a digital refractometer with automatic temperature compensation (Atago UG-alpha, #3464).
The determination of total BPA (free plus conjugated) was measured after enzymatic hydrolysis of urine samples. After derivatization with pentafluorobenzyl bromide, the samples were extracted with a mixture of hexane and dichloromethane and then analyzed using an Agilent 6890 gas chromatograph coupled with a Waters Quattro mass spectrometer equipped with a source operating in the negative chemical ionization mode and a detector in MRM mode.
For the phthalate analysis, urine samples were enzymatically hydrolyzed. The resulting phthalate monoesters were extracted either by an anion exchange solid phase for Part I of the study or by a liquid-liquid technique using a mixture of hexane and ethyl acetate for Part II. The dry extracts reconstituted with aqueous solvents were analysed using a Micromass Quattro Premier XE mass spectrometer (for all analytes of Part I and MCMHP of Part II) or a Waters Xevo TQ-S mass spectrometer (for other analytes of Part II) in MRM mode with an electrospray ion source in negative mode.
Statistical Analysis
Data analysis was performed using SAS Enterprise Guide (version 4.2). The numeric machine readings from the INSPQ lab were reported for phthalate metabolites and BPA for levels below the LOD. Given that urinary levels were not normally distributed, they were natural log-transformed. Metabolites with less than 70% detection were excluded from the analysis (mono-isononyl phthalate [(MiNP), mono-(7-hydroxy-methyloctyl) phthalate (MHiDP), mono-(7-carboxy-2,7-dimethylheptyl) phthalate (MCiNP), mono-(carboxy-isooctyl) phthalate (MCiOP)]. We conducted descriptive statistics to calculate frequency distributions of maternal characteristics, and the geometric mean and percentiles of each chemical by visit. Spearman correlations were calculated on SG adjusted metabolite levels between trimester single spot urine samples (T2, T3) and postpartum samples (T5). The SG-adjusted concentration used the following formula adapted from Just et al.:17 Pc=Pi [(SGm–1)/(SGi−1)] where Pc is the SG-adjusted metabolite concentration, Pi is the observed metabolite concentration, and SGi is the specific gravity of the urine sample and SGm is the median SG for the cohort. We examined time of day differences in metabolite levels for the 24 h serial spot urine samples (T1a, T1b). We reported geometric means (GMs) for each time period and tested for fixed effects using specific gravity adjusted log–transformed metabolite levels in mixed models with random subject effects.
The MEHP% was calculated by converting the concentrations of the five di-2-ethylhexyl phthalate (DEHP) metabolites [(monoethylhexyl phthalate (MEHP), mono-(2-ethyl-5-hydroxyhexyl) phthalate (MEHHP), mono-(2-ethyl-5-oxohexyl) phthalate (MEOHP), mono(2-ethyl-5-carboxy-pentyl)phthalate (MECPP), mono(2-carboxy-methylhexyl)phthalate (MCMHP)] into nmol/L and dividing the molar mass of MEHP by the mass of the sum of all five metabolites, then multiplying by 100 to give MEHP%. The MEHP% represents a person’s relative efficiency to form the more hydrophilic and potentially less biologically active secondary metabolites.10,18 We also calculated the mass concentration ratio of MECPP to MEHHP, which is a potential indicator of DEHP exposure timing.18,19
Intraclass correlation coefficients (ICC) were calculated using a one-way random effects model (Proc Mixed) to estimate the between- and within-subject variability within a day and throughout pregnancy. ICC measures the ratio of between-subject variance to total variance ranging from 0 (meaning no within person reproducibility) to 1 (meaning perfect reproducibility): 0.75 was defined as high; 0.40–0.75 as moderate; below 0.40 as poor reproducibility.20 We ran two different models: 1) unadjusted for specific gravity (SG); and, 2) SG- adjusted metabolite concentrations. We imputed 0.0001 for values of 0 in the log transformed urinary metabolites. To calculate ICCs across all study time points, we chose a random sample (proc survey select) for each participant from T1A and T1B where serial samples were collected over a 24 h period.
For the calculation of empirical distribution of sensitivity, we followed the methods given in Adibi et al.,10 which involved the following steps: (1) a single sample per woman was randomly selected from any visit and classified as high or low based on the Canadian Health Measures Survey (CHMS) geometric mean (GM) for women of reproductive age as the cutoff point for BPA21 and phthalates;22 (2) the “True” exposure was calculated from the GM of the woman’s remaining samples and was similarly classified as low or high based on the CHMS GM for that metabolite; (3) for each woman, the randomly selected sample was compared with the GM (“True” exposure) of all her remaining samples in terms of low vs. high exposure and the sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were calculated; and, (4) the process was repeated 1000 times and the median was reported.
RESULTS
Most of the women in the study were born in Canada and had a high household income of >$100,000 Canadian per year (see Table 3). Close to half of the women were primiparous (46%) and the mean maternal age was 32.4 years (median 32 years). Over 32% of the participants had tried smoking at some point in their life but only a few were current smokers (data not shown). Season of conception was nearly evenly spread across all seasons.
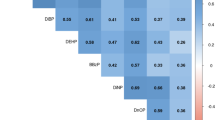
The chemical descriptive statistics are shown in Table 4. There was 100% detection for BPA, mono-n-butyl phthalate (MBP), MEHHP, monoethyl phthalate (MEP), mono-2-hydroxy-isobutyl phthalate (2OH-MiBP), MECPP and mono-iso-butyl phthalate (MiBP). The BPA geometric mean was quite consistent across pregnancy and into the postpartum period and ranged from 1.2–1.5 ug/l depending on the visit. The highest phthalate metabolite levels were those of MEP (GM 42.5 ug/l) followed by MBP (23 ug/l). The correlation between pregnancy spot urine samples (T2, T3) and the postpartum samples are shown in Table 5. The highest correlation (r=0.54) was seen among the MBP third trimester (T3) and postpartum (T5) spot urine samples. Within pregnancy there were significant but low correlations for BPA, MBP, MEHP, MEOHP, and MEP. We found significant differences in metabolite levels by time of day for several metabolites (see Table 6 and Figure 1). We found the highest levels in the evening (18:00–23:59) for BPA, MCPP, MHBP, MHiNP and 4 DEHP metabolites (MEHP, MEHHP, MEOHP, MECPP). Only MEP had the highest level in the morning samples (8:00–11:59), while MBzP, 2OH-MiBP, MCMHP and MiBP were highest between midnight and 7:59. Presumably one of the samples within this time period could have included the first morning void.
The ICCs for Part I and Part II of the study are shown on Tables 7 and 8 and displayed in Figures 2 and 3. SG-adjusted ICCs were consistently higher than the unadjusted ICCs for the within day samples (T1a and T1b) but had minimal effect or reduced the ICC across all time points. For Part I metabolites, within a day reproducibility was moderate for some phthalate metabolites (MEP, MBzP) and the ICCs were quite similar for the weekdays and the weekend days. However there was low reproducibility across all time points for all metabolites, including BPA. For Part II metabolites (Table 6), there was moderate reproducibility within a week-day or week-end day for 2OH-MiBP, MHiNP, mono-(2-propyl-6 oxo heptyl)phthalate (MOiDP), MiBP, MEHP% and MECPP/MEHHP ratio. There were often differences between the weekday (T1a) and weekend day (T1b) ICCs but this difference was not consistent across chemicals.
DISCUSSION
In general we found metabolite levels to vary by time of day, and ICCs were much higher for the within day samples (T1a and T1b) than across all time points. The ICCs were higher for metabolites of chemicals used in consumer products (e.g., MEP, MBzP),23 than for chemicals for which diet is likely to be the main source (e.g., BPA, DEHP metabolites). We found that all metabolites had low ICCs across pregnancy and into the postpartum period. The correlation coefficients across visits were generally low with some moderate correlations for only MBP and MEP.
BPA
The P4 study found low reproducibility of BPA within a weekday (0.33), a weekend day (0.31) and across pregnancy (0.07). In general, the literature suggests low reproducibility of BPA with reported ICCs ranging from 0.10–0.32 for studies looking at pregnant women.11,13,15,24, 25, 26, 27 The P4 study urinary concentrations were similar to other studies of pregnant women, with our geometric means ranging from 1.2–1.5 ug/l (see Supplementary Materials). In a CDC study with 8 participants who collected 427 samples over one week, the authors found high within person variance for spot urine, first morning voids and 24 h urine samples.28 There are 2 studies29,30 that have found moderate ICCs (0.40, 0.51) in men and children. Both these studies collected samples only days apart. Like other authors27 we found low correlation across pregnancy study visits for BPA. In our time of day analysis we found the highest BPA levels in the evening samples (18:00–23:59). If the main source of BPA is food31 it seems reasonable that the highest levels would be in the evening after consuming food all day. However Stahlhut et al.32 did not find a strong association between fasting times and BPA levels.
MEP and MBzP
Both MEP and MBzP had moderate reproducibility for samples collected within a day (T1a, T1b) (see Figure 2). We also found moderate correlation coefficients between trimester spot urines and the postpartum period (Table 5). The majority of other studies have also shown high to moderate reproducibility for MEP measured in repeated spot urine samples,11 and in repeated first morning voids.26,33, 34, 35, 36, 37, 38 Eight other studies have found moderate ICCs for MBzP with ICCs ranging from 0.41–0.6510,12,18,33, 34, 35, 36,38,39 (See Supplementary Table 2). However across all time points, we found low reproducibility for both MEP and MBzP. Adibi et al.10 reported low reproducibility for MEP for repeat spot urines over 6 weeks in pregnancy. Townsend et al.26 and Teitlbaum40 showed lower reproducibility of samples collected over a longer period of time (6 months to 3 years). The time of day analysis (Table 6) showed MEP to vary by time of day with the highest levels between 8:00–11:59. Preau et al.38 also showed significant variation of MEP within a day, among 8 volunteers who collected 427 samples over 7 consecutive days.
MCPP
Our study showed low reproducibility throughout pregnancy and within a day. Other studies have also shown low reproducibility18,40 for MCPP. In contrast, Adibi et al.10 found moderate reproducibility (0.44) in spot urine samples from pregnant women collected over 6 weeks, while Peck et al.36 found moderate ICCs (0.59) in samples from women who collected serial first morning voids over 2 months.
MBP, MiBP
Our study suggested lower reproducibility across the study period for MBP. However, we saw moderate reproducibility within a weekday (MBP, MiBP) and weekend day (MiBP). The literature suggests that the reproducibility of MBP appears to be moderate to high with the majority of studies showing adjusted ICCs above 0.40.10, 11, 12,34, 35, 36,39 For MiBP, other studies have reported ICCs that ranged from 0.2840 to as high as 0.54.10
DEHP Metabolites (MEHP, MEHHP, MEOHP, MECPP, MCMHP)
Our study showed low reproducibility across pregnancy for MEHP, MEHHP, MEOHP, which is in agreement with other studies.10, 11, 12 For some DEHP metabolites, within day reproducibility was moderate for a weekday (MEHHP, MECPP) and a weekend day (MEHP, MCMHP). Two other studies have shown moderate reproducibility of MEHP over the short term.34,35 Townsend et al.26 found moderate reproducibility in MECPP in samples from first morning voids over 1–3 years. Time of day was an important predictor for all of the DEHP metabolites measured with the highest levels for 4 of them (MEHP, MEHHP, MEOHP, MECPP) being in the evening. Cantonwine et al.12 also found significant variation for MECPP by time of day in their study.
Derived Exposures
The MEHP% calculation has been suggested as a way to calculate a person’s relative efficiency to metabolize the monoester (MEHP) to the more hydrophilic oxidative secondary metabolites (MEHHP, MEOHP, MECPP, MCMHP).10,18 In our study, MEHP% was more reproducible within a weekday (ICC=0.56) and weekend day (ICC=0.60) than any of the secondary metabolites alone. Three other studies10,12,18 have also shown higher ICCs for MEHP% than the secondary metabolites alone (see Supplementary Tables). This may suggest that despite the variability in excretion of the metabolites in urine, the metabolism (MEHP%) may be more reproducible over time. We also calculated the mass concentration ratio of MECPP to MEHHP, which is a potential indicator of timing between DEHP exposure and urine sample collection18,19 and found moderate reproducibility. Meeker et al.18 showed higher reproducibility in the ratio than the metabolites alone but it was still low (ICC=0.26). Cantonwine et al.12 showed only slightly higher reproducibility in the ratio compared with the metabolites alone.
Surrogate Analysis
The surrogate analysis is shown on Table 9 with sensitivity to correctly classify a participant in the ‘high’ category using a randomly chosen sample versus the GM of all the urine samples ranging from 0.64 (MHiNP, MECPP) to 1.00 (MiBP). BPA had a sensitivity of 65%, similar to that reported by Braun et al.11 (see Table 10). Our results were similar to Adibi et al.10 for the following phthalate metabolites: MBP, MBzP, MCPP and MEP; however, Braun et al.11 showed slightly lower sensitivities for MBP, MBzP and MEP.
Limitations and Considerations
Our study is limited by its somewhat low recruitment (11% acceptance) and bias towards highly educated, high-income Caucasian women. A recent study41 showed significant differences in urinary concentrations based on socioeconomic status and ethnicity; however this would only be an issue if urinary concentrations affect the variability of these biomarkers.
An ICC of 0.40 has been suggested as sufficient reproducibility in a biomarker to justify using it in an epidemiological study;42 however, this would still cause exposure misclassification and a reduction in the relative risk, as explained by de Klerk et al.43 and discussed by Adibi et al.10 In a simulation study, de Klerk et al.43 estimated that an ICC of 0.42 in the exposure variable would still result in a reduction in the median relative risk of 32% while an ICC of 0.72 would give a 17% reduction.
CONCLUSIONS
We found low reproducibility and sensitivity of BPA and all phthalate metabolites throughout pregnancy and into the postpartum period but, much higher reproducibility within a day. Time of day was also a significant predictor of exposure levels. Given this, it seems that for a few phthalate metabolites (MEP, MBzP) there is moderate reproducibility over a short period of time in pregnancy (days ) and, practically speaking, a single spot urine sample will quite accurately represent exposure to phthlatates; however, to accurately represent exposure over the course of the entire pregnancy, more than one measurement at different times of day is required to get a more accurate picture of exposure, particularly when diet is the main source of exposure to the chemicals of interest.
References
Haines DA, Murray J Human biomonitoring of environmental chemicals—early results of the 2007-2009 Canadian Health Measures Survey for males and females. Int J Hyg Environ Health 2012: 215: 133–137.
Woodruff TJ, Zota AR, Schwartz JM Environmental chemicals in pregnant women in the United States: NHANES 2003-2004. Environ Health Perspect 2011: 119: 878–885.
Saillenfait AM, Sabate JP, Robert A, Rouiller-Fabre V, Roudot AC, Moison D et al. Dose-dependent alterations in gene expression and testosterone production in fetal rat testis after exposure to di-n-hexyl phthalate. J Appl Toxicol 2013: 33: 1027–1035.
Swan SH, Main KM, Liu F, Stewart SL, Kruse RL, Calafat AM et al. Decrease in anogenital distance among male infants with prenatal phthalate exposure. Environ Health Perspect 2005: 113: 1056–1061.
Rochester JR Bisphenol A and human health: A review of the literature. Reprod Toxicol 2013: 42C: 132–155.
Braun JM, Yolton K, Dietrich KN, Hornung R, Ye X, Calafat AM et al. Prenatal bisphenol A exposure and early childhood behavior. Environ Health Perspect 2009: 117: 1945–1952.
ATSDR. Toxcological Profile Di(2-Ethylhexyl)Phthalate. Atlanta, Georgia: Department of Health and Human Services, Public Health Service. Agency of Toxic Substances and Disease Registry, 2002.
Dafnis E, Sabatini S The effect of pregnancy on renal function: physiology and pathophysiology. Am J Med Sci 1992: 303: 184–205.
Hytten F Blood volume changes in normal pregnancy. Clin Haematol 1985: 14: 601–612.
Adibi JJ, Whyatt RM, Williams PL, Calafat AM, Camann D, Herrick R et al. Characterization of phthalate exposure among pregnant women assessed by repeat air and urine samples. Environ Health Perspect 2008: 116: 467–473.
Braun JM, Smith KW, Williams PL, Calafat AM, Camann D, Herrick R et al. Variability of urinary phthalate metabolite and bisphenol A concentrations before and during pregnancy. Environ Health Perspect 2012: 120: 739–745.
Cantonwine DE, Cordero JF, Rivera-Gonzalez LO, Anzalota Del Toro LV, Ferguson KK, Mukherjee B et al. Urinary phthalate metabolite concentrations among pregnant women in Northern Puerto Rico: Distribution, temporal variability, and predictors. Environ Int 2014: 62: 1–11.
Meeker JD, Cantonwine DE, Rivera-Gonzalez LO, Ferguson KK, Mukherjee B, Calafat AM et al. Distribution, variability, and predictors of urinary concentrations of phenols and parabens among pregnant women in Puerto Rico. Environ Sci Technol 2013: 47: 3439–3447.
Philippat C, Mortamais M, Chevrier C, Petit C, Calafat AM, Ye X et al. Exposure to phthalates and phenols during pregnancy and offspring size at birth. Environ Health Perspect 2012: 120: 464–470.
Quiros-Alcala L, Eskenazi B, Bradman A, Ye X, Calafat AM, Harley K Determinants of urinary bisphenol A concentrations in Mexican/Mexican-American pregnant women. Environ Int 2013: 59: 152–160.
Ye X, Bishop AM, Reidy JA, Needham LL, Calafat AM Temporal stability of the conjugated species of bisphenol A, parabens, and other environmental phenols in human urine. J ExpoSciEnviron Epidemiol 2007: 17: 567–572.
Just AC, Adibi JJ, Rundle AG, Calafat AM, Camann DE, Hauser R et al. Urinary and air phthalate concentrations and self-reported use of personal care products among minority pregnant women in New York city. J Expo Sci Environ Epidemiol 2010: 20: 625–633.
Meeker JD, Calafat AM, Hauser R Urinary phthalate metabolites and their biotransformation products: predictors and temporal variability among men and women. J Expo Sci Environ Epidemiol 2012: 22: 376–385.
Lorber M, Koch HM, Angerer J A critical evaluation of the creatinine correction approach: can it underestimate intakes of phthalates? A case study with di-2-ethylhexyl phthalate. J Expo Sci Environ Epidemiol 2011: 21: 576–586.
Rosner B Fundamentals of Biostatistics. Duxburty: Belmont, CA, 2006.
Canada H, Report on Human Biomonitoring of Environmental Chemcials in Canada: Results of the Canadian Health Measures Survey Cycle 1 (2007-2009). Ottawa, ON, 2010.
Health Canada. Second Report on Human Biomonitoring of Environmental Chemicals in Canada: Results of the Canadian Health Measures Survey Cycle 2 (2009-2011). Ottawa, ON, 2013.
IPCS INCHEM. Concise International Chemical Assessment Document 17: Butyl benzyl phthalate http://www.inchem.org/documents/cicads/cicads/cicad17.htm#PartNumber:4. Accessed May 26, 2014.
Braun JM, Kalkbrenner AE, Calafat AM, Bernert JT, Ye X, Silva MJ et al. Variability and predictors of urinary bisphenol A concentrations during pregnancy. Environ Health Perspect 2011: 119: 131–137.
Philippat C, Wolff MS, Calafat AM, Ye X, Bausell R, Meadows M et al. Prenatal exposure to environmental phenols: concentrations in amniotic fluid and variability in urinary concentrations during pregnancy. Environ Health Perspect 2013: 121: 1225–1231.
Townsend MK, Franke AA, Li X, Hu FB, Eliassen AH Within-person reproducibility of urinary bisphenol A and phthalate metabolites over a 1 to 3 year period among women in the Nurses' Health Studies: a prospective cohort study. Environ Health 2013: 12: 80.
Jusko TA, Shaw PA, Snijder CA, Pierik FH, Koch HM, Hauser R et al. Reproducibility of urinary bisphenol A concentrations measured during pregnancy in the Generation R Study. J Expo Sci Environ Epidemiol 2014: 24: 532–536.
Ye X, Wong LY, Bishop AM, Calafat AM Variability of urinary concentrations of bisphenol A in spot samples, first morning voids, and 24-hour collections. Environ Health Perspect 2011: 119: 983–988.
Lassen TH, Frederiksen H, Jensen TK, Petersen JH, Main KM, Skakkebæk NE et al. Temporal variability in urinary excretion of bisphenol A and seven other phenols in spot, morning, and 24-h urine samples. Environ Res 2013: 126: 164–170.
Heffernan AL, Aylward LL, Samidurai AJ, Davies PS, Toms LM, Sly PD et al. Short term variability in urinary bisphenol A in Australian children. Environ Int 2014: 68: 139–143.
Vandenberg LN, Hauser R, Marcus M, Olea N, Welshons WV Human exposure to bisphenol A (BPA). Reprod Toxicol 2007: 24: 139–177.
Stahlhut RW, Welshons WV, Swan SH Bisphenol A data in NHANES suggest longer than expected half-life, substantial nonfood exposure, or both. Environ Health Perspect 2009: 117: 784–789.
Baird DD, Saldana TM, Nepomnaschy PA, Hoppin JA, Longnecker MP, Weinberg CR et al. Within-person variability in urinary phthalate metabolite concentrations: measurements from specimens after long-term frozen storage. J Expo Sci Environ Epidemiol 2010: 20: 169–175.
Hoppin JA, Brock JW, Davis BJ, Baird DD Reproducibility of urinary phthalate metabolites in first morning urine samples. Environ Health Perspect 2002: 110: 515–518.
Marcus M, Christensen KY, Manatunga A, Rudra CB, Brock JW, Small CM Variability of phthalate monoester levels in daily first-morning urine from adult women: a pilot study. Rev Environ Health 2010: 25: 359–368.
Peck JD, Sweeney AM, Symanski E, Gardiner J, Silva MJ, Calafat AM et al. Intra- and inter-individual variability of urinary phthalate metabolite concentrations in Hmong women of reproductive age. J Expo Sci Environ Epidemiol 2010: 20: 90–100.
Frederiksen H, Kranich SK, Jorgensen N, Taboureau O, Petersen JH, Andersson AM Temporal variability in urinary phthalate metabolite excretion based on spot, morning, and 24-h urine samples: considerations for epidemiological studies. Environ Sci Technol 2013: 47: 958–967.
Preau JLJ, Wong LY, Silva MJ, Needham LL, Calafat AM Variability over 1 week in the urinary concentrations of metabolites of diethyl phthalate and di(2-ethylhexyl) phthalate among eight adults: an observational study. Environ Health Perspect 2010: 118: 1748–1754.
Fromme H, Bolte G, Koch HM, Angerer J, Boehmer S, Drexler H et al. Occurrence and daily variation of phthalate metabolites in the urine of an adult population. IntJ HygEnviron Health 2007: 210: 21–33.
Teitelbaum SL, Britton JA, Calafat AM, Ye X, Silva MJ, Reidy JA et al. Temporal variability in urinary concentrations of phthalate metabolites, phytoestrogens and phenols among minority children in the United States. Environ Res 2008: 106: 257–269.
Kobrosly RW, Parlett LE, Stahlhut RW, Barrett ES, Swan SH Socioeconomic factors and phthalate metabolite concentrations among United States women of reproductive age. Environ Res 2012: 115: 11–17.
Rosner B Fundamentals of Biostatistics. Dusbury: Pacific Grove, CA, 2000.
de Klerk NH, English DR, Armstrong BK A review of the effects of random measurement error on relative risk estimates in epidemiological studies. Int J Epidemiol 1989: 18: 705–712.
Acknowledgements
We would like to give our sincere thanks to the participants of the P4 study and the OMNI Research Group. We would also like to express our gratitude to Antonia Calafat for her expert advice throughout the project and on this paper. This project was funded by Health Canada’s Chemical Management Plan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Journal of Exposure Science and Environmental Epidemiology website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Fisher, M., Arbuckle, T., Mallick, R. et al. Bisphenol A and phthalate metabolite urinary concentrations: Daily and across pregnancy variability. J Expo Sci Environ Epidemiol 25, 231–239 (2015). https://doi.org/10.1038/jes.2014.65
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jes.2014.65
Keywords
This article is cited by
-
Maternal urine phthalate metabolite exposure and miscarriage risk: a nested case–control study of the Zunyi Birth Cohort
Environmental Science and Pollution Research (2022)
-
Impact of perinatal environmental health education intervention on exposure to endocrine disruptors during pregnancy—PREVED study: study protocol for a randomized controlled trial
Trials (2021)
-
Prenatal exposures to mixtures of endocrine disrupting chemicals and children’s weight trajectory up to age 5.5 in the SELMA study
Scientific Reports (2021)
-
Fetal growth in environmental epidemiology: mechanisms, limitations, and a review of associations with biomarkers of non-persistent chemical exposures during pregnancy
Environmental Health (2019)
-
Biomonitoring and Nonpersistent Chemicals—Understanding and Addressing Variability and Exposure Misclassification
Current Environmental Health Reports (2019)