Abstract
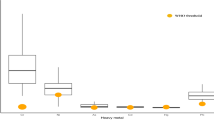
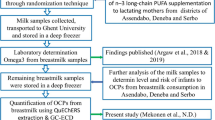
This study looked to identify determinants of exposure to dioxin in breast milk from breast-feeding women in a hot spot of dioxin exposure in Vietnam. Breast milk was collected from 140 mothers 1 month after delivery. The risk factors investigated included length of residency, drinking of well water and the frequency of animal food consumption. Cluster analysis was performed to identify dietary patterns of fish and meat portions, fish variety and egg variety. Residency, age and parity were clearly associated with increased dioxin levels. Drinking well water and the consumption of marine crab and shrimps were related to higher levels of furans in breast milk. The consumption of quail eggs also appeared to be associated with increased levels of some dioxin isomers in this area. Some mothers who ate no or less meat than fish and mothers who consumed more freshwater fish than marine fish had lower levels of dioxins in their breast milk. However, the type of water and the eating habits of mothers contributed only partly to the increased dioxin levels in their breast milk; the length of residency was the most important risk factor associated with increased dioxin body burdens of mothers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Mai TA, Doan TV, Tarradellas J, Felippe de Alencastro L, Grandjean D . Dioxin contamination in soils of Southern Vietnam. Chemosphere 2007; 67: 1802–1807.
Schecter A, Hoang TQ, Päpke O, Tung KC, Constable JD . Agent Orange, dioxins, and other chemicals of concern in Vietnam: Update 2006. J Occup Environ Med 2006; 48: 408–413.
Hatfield Consultants and the Office of National Steering Committee 33. Comprehensive Assessment of Dioxin Contamination in Da Nang Airport, Viet Nam: Environmental Levels, Human Exposure and Options for Mitigating Impacts. Final Report 2009. http://www.hatfieldgroup.com/wp-content/uploads/AgentOrangeReports/DANDI-II1450/Da%20Nang%202009%20Report.pdf.
Dwernychunk LW . Dioxin hot spots in Vietnam. Chemosphere 2005; 60: 998–999.
Tai PT, Nishijo M, Kido T, Nakagawa H, Maruzeni S, Anh NTN et al. Dioxin concentrations in breast milk of Vietnamese nursing mothers: A survey four decades after the herbicide spraying. Environ Sci Technol 2011; 45: 6625–6632.
WHO, IARC. Polychlorinated dibenzo-p-dioxins and polychlorinated dibenzo furans, in: IARC Monographs on the evaluation of carcinogenic risks to humans. Vol. 69. IARC: Lyon. 1997.
Sasamoto T, Horii S, Ibe A, Takada N, Shirota K . Concentration changes of PCDDs, PCDFs, and dioxin-like PCBs in human breast milk samples as shown by a follow-up survey. Chemosphere 2006; 64: 642–649.
Baars AJ, Bakker MI, Baumann RA, Boon PE, Freijer JI, Hoogenboom LAP et al. Dioxins, dioxin-like PCBs and nondioxin-like PCBs in foodstuffs: occurrence and dietary intake in the Netherlands. Toxicol Lett 2004; 151: 51–61.
Patandin S, Dagnelie PC, Mulder PG, Op de Coul E, Van der Veen JE, Weisglas-Kuperus N et al. Dietary exposure to polychlorinated biphenyls and dioxins from infancy until adulthood: a comparison between breast-feeding, toddler, and long-term exposure. Environ Health Perspect 1999; 107: 45–51.
Leng JH, Kayama F, Wuang PY, Nakamura M, Nakata T, Wuang Y . Levels of persistent organic pollutants in human milk in two Chinese coastal cities, Tianjin and Yantai: Influence of fish consumption. Chemosphere 2009; 75: 634–639.
Kvalem HE, Knutsen HK, Thomsen C, Haugen M, Stigum H, Brantsaeter AL . Role of dietary patterns for dioxin and PCB exposure. Mol Nutr Food Res 2009; 53: 1438–1451.
Arisawa K, Uemura H, Hiyoshi M, Kitayama A, Takami H, Sawachika F et al. Dietary patterns and blood levels of PCDDs, PCDFs, and dioxin-like PCBs in 1656 Japanese individuals. Chemosphere 2011; 82: 656–662.
Schecter A, Hoang TQ, Pavuk M, Päpke O, Malisch R, Constable JD . Food as a source of dioxin exposure in the residents of Bien Hoa city, Vietnam. J Occup Environ Med 2003; 45: 781–788.
Van den Berg M, Birnbaum LS, Denison M, Vito MD, Farland W, Feeley M et al. The 2005 World Health Organization Revolution of human and mammalian toxic equivalency factors for dioxins and dioxin-like compounds. Toxicol Sci 2006; 93: 223–241.
Nishijo M, Tai PT, Nakagawa H, Maruzeni S, Anh NTN, Luong HV et al. Impact of perinatal dioxin exposure on infant growth: a cross-sectional and longitudinal studies in dioxin-contaminated areas in Vietnam. PLoS One 2012; 7: e4027.
Traag W, Hoang Thi T, Murk T, Hoogenboom L . Dioxins in free range consumption eggs from Vietnam: levels and health risks. Organohalogen Compounds 2012; 74: 1373–1376.
Chen HL, Huang HY, Huang PC, Lee CC . Relationship of PCDD/F concentrations in duck-egg farmers and consumption of ranched duck eggs in central Taiwan. Environ Toxicol Chem 2010; 29: 2402–2408.
Ulaszewska MM, Zuccato E, Capri E, Iovine R, Colombo A, Rotella G et al. The effect of waste combustion on the occurrence of polychlorinated dibenzo-p-dioxins (PCDDs), polychlorinated dibenzo furans (PCDFs) and polychlorinated biphenyls (PCBs) in breast milk in Italy. Chemosphere 2011; 82: 1–8.
Nakatani T, Okazaki K, Ogaki S, Itano K, Fujita T, Kuroda K et al. Polychlorinated dibenzo-p-dioxins, polychlorinated dibenzofurans, and coplanar polychlorinated biphenyls in human milk in Osaka city, Japan. Arch Environ Contam Toxicol 2005; 49: 131–140.
Guan P, Tajimi M, Uehara R, Watanabe M, Oki I, Ojima T et al. Associations between dietary intake and breast milk dioxin levels in Tokyo, Japan. Pediat Int 2005; 47: 560–566.
Schecter A, Papke O, Lis A, Ball M, Ryan JJ, Olson JR et al. Decrease in milk and blood dioxin levels over two years in a mother nursing twins: estimates of decreased maternal and increased infant dioxin body burden from nursing. Chemosphere 1996; 32: 543–549.
Ritter L, Solomon K, Sibley P, Hall K, Keen P, Mattu G et al. Sources, pathways, and relative risks of contaminants in surface water and groundwater: a perspective prepared for the Walkerton inquiry. J Toxicol Environ Health A 2002; 65: 1–142.
Environmental Agency in Japan Handbook of dynamics model of dioxins. Division of environmental dioxin contamination control, Environmental Agency. Tokyo, Japan 2004. http://www.env.go.jp/chemi/dioxin/hand.
Wang IC, Wu YL, Lin LF, Chang-Chien GP . Human dietary exposure to polychlorinated dibenzo-p-dioxins and polychlorinated dibenzo furans in Taiwan. J Hazard Mater 2009; 164: 621–626.
Nakatani T, Ogaki S, Itano K, Fujita T, Mori Y, Endo G . Toxic equivalent (TEQ) levels and characteristics of PCDD/F and CoPCB concentrations in marine foods in Japan. Bull Environ Contam Toxicol 2006; 77: 237–244.
Kijlstra A, Traag W, Hoogenboom L . Effect of flock size on dioxin levels in eggs from chickens kept outside. Poultry Sci 2007; 86: 2042–2048.
Acknowledgements
We thank all mothers participating in this study. We are grateful to medical staffs in Health Department of DaNang city, Thanh Khe district hospital, and Commune Health Centers in Thanh Khe district for their collaboration in surveys. This work was supported partly by Project Research from JSPS Asian Core Program, and the Japan Society for the promotion of science (Grant-in-Aid for Scientific Research (B) (25305024) and (25290005)).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Journal of Exposure Science and Environmental Epidemiology website
Supplementary information
Rights and permissions
About this article
Cite this article
Anh, N., Nishijo, M., Tai, P. et al. Maternal risk factors associated with increased dioxin concentrations in breast milk in a hot spot of dioxin contamination in Vietnam. J Expo Sci Environ Epidemiol 24, 489–496 (2014). https://doi.org/10.1038/jes.2013.73
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jes.2013.73
Keywords
This article is cited by
-
Paternal Environmental Toxicant Exposure and Risk of Adverse Pregnancy Outcomes
Current Obstetrics and Gynecology Reports (2019)
-
Sex-specific effects of perinatal dioxin exposure on eating behavior in 3-year-old Vietnamese children
BMC Pediatrics (2018)