Abstract
In the course of optimizing pargamicin A production in Amycolatopsis sp. ML1-hF4, we discovered novel cyclic depsipeptide compounds in the broth and designated them valgamicins A, C, T and V. The structures of these molecules were determined by spectroscopic studies, advanced Marfey’s method and X-ray crystal structural analysis. Valgamicin C contains the extremely rare amino acid cleonine. To our knowledge, this is the first report of a cleonine-containing metabolite from a naturally isolated microorganism without any breeding or mutation treatment. None of the valgamicins showed potent antibacterial activity against either Gram-positive or -negative bacteria. Valgamicins A, C and T exhibited moderate cytotoxicity against human tumor cell lines.
Similar content being viewed by others
Introduction
Actinomycetes bacteria are an important medicine source as they can produce compounds with complex novel structures with useful biological activity.1, 2, 3 Streptomycin,4 kanamycin,5 bleomycin,6 avermectins7 and FK-5068 are well-known metabolites of Actinomycetes, which have contributed to considerable advances in the medical field. Thus, it is of interest to discover further interesting and useful compounds with new scaffolds and/or biological activity from the metabolites of Actinomycetes.
During optimization of the productivity of the anti-Gram positive, membrane-acting compound, pargamicin A produced by Amycolatopsis sp. ML1-hF4,9, 10 we discovered the related antibacterial compounds pargamicins B, C and D in the broth, as previously reported.11 Pargamicin A is a structurally unique cyclic peptide consisting of N-methyl-3-hydroxy valine, 4-hydroxy piperazic acid, sarcosine, phenylalanine, N-hydroxy isoleucine and piperazic acid.9
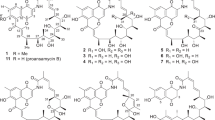
We have also discovered new cytotoxic cyclic depsipeptides in this broth and designated them valgamicins (VLGs) A, C, T and V, named after their different amino acids, alanine, cleonine, threonine and valine, respectively (Figure 1). VLG-C contains the extremely rare amino acid, cleonine, 2-amino-2-(1-hydroxycyclopropyl) acetic acid.12 Herein, we report the first example of a cleonine-containing bacterial metabolite from a naturally isolated organism without any breeding or mutation treatment. The fermentation, isolation, structural elucidation, antimicrobial activity, cytotoxicity and structure–activity relationship of the VLGs are described.
Results and discussion
Producing organism and fermentation
Amycolatopsis sp. ML1-hF4 strain was cultured for 3 days at 30 °C in liquid media consisting of 2% galactose, 2% dextrin hydrate, 1% soytone, 0.5% corn steep liquor, 0.2% ammonium sulfate and 1.0% calcium carbonate (pH 7.0 before sterilization). This seed culture was inoculated (3%) into media consisting of 0.33% galactose, 0.33% dextrin hydrate, 0.17% glycerol, 0.17% soytone, 0.083% corn steep liquor, 0.033% ammonium sulfate and 0.2% calcium carbonate (pH 7.0 before sterilization), and cultivated for 7 days at 27 °C with shaking (180 r.p.m.). This media was sixfold diluted compared with that used for pargamicin A discovery1 and the same as that used for discovery of pargamicins B, C and D.11
Isolation of VLG-A, C, T and V
The 2 l culture broth described above was then centrifuged (3000 g, 10 °C, 15 min) and the supernatant was applied to a Diaion HP-20 column (Mitsubishi Chemical Co., Tokyo, Japan, 700 ml wet volume). The column was washed with 2 l each of distilled water, 40% aqueous methanol, and 90% aqueous methanol and eluted with 90% aqueous acetone. The acetone-eluted fraction was dried to give crude material (390.6 mg) and subjected to silica gel column chromatography. The column was washed with EtOAc and eluted with EtOAc/MeOH (10:1) to give crude VLGs (219.6 mg) and was further purified by reverse-phase HPLC. The HPLC conditions were as follows: column, Shiseido CAPCELL PAK UG-120 (20 × 250 mm, Tokyo, Japan); flow rate, 10 ml min−1; solvent, 40–60% CH3CN aq + 0.01% trifluoroacetic acid, linear gradient; column oven, 50 °C; detection, 205 nm. VLG-T (1.6 mg), VLG-A (2.0 mg), VLG-C (1.9 mg) and VLG-V (5.6 mg) were eluted at 36–38, 52–54, 61–63 and 82–84 min, respectively.
Structural elucidation of VLGs-A, C, T and V
The physicochemical properties of VLG-A, C, T and V are summarized in Table 1. The molecular formulae of VLG-A, C, T and V were C29H48N4O8, C31H50N4O9, C30H50N4O9 and C31H52N4O8, respectively, as determined by HRESI-MS and NMR spectra. (1H and 13C NMR spectra; Supplementary Figures S1–S8, 2D-COSY spectra; Supplementary Figures S9–S12, HMQC spectra; Supplementary Figures S13–S16, heteronuclear multiple bond correlation (HMBC) spectra; Supplementary Figures S17–S20, F1-selective HMBC of VLG-C; Supplementary Figure S21, HRESI-MS; Supplementary Figures S22–S25). NMR data are summarized in Table 2. The IR spectra of the VLGs showed absorptions characteristic of peptide bonds (1658–1669 and 1542–1546 cm−1) and lactone linkages (1737–1745 cm−1). The IR and NMR spectral data for the VLGs indicated that they are related peptide compounds.
Structure of VLG-C
The planar structure of VLG-C was determined as follows. The relationships between the proton and carbon signals of VLG-C were established by 1H, 13C, DEPT and HMQC spectra. These spectra revealed the presence of seven quaternary carbons, including six carbonyls, 10 sp3 methines, five methylenes and nine methyl groups. Analyses of 1H–1H COSY, 1D-TOCSY, HMBC and F1-selective HMBC13 spectra were performed to elucidate the structure. Two characteristic high magnetic field methylene signals (δH 0.52, 0.61) correlated in the 1H–1H COSY and coupled with a hydroxyl-bonded quaternary carbon (δC 54.1) in the HMBC, revealed the presence of a hydroxyl-cyclopropane moiety. The COSY correlation from an NH (δH 8.18) to an α-methine proton (δH 4.96) and a long-range correlation from this α-methine to a carbonyl (δC 170.5) and the quaternary carbon of β-cyclopropane indicated the presence of 2-amino-2-(1-hydroxycyclopropyl) acetic acid, the unusual amino acid cleonine.12, 14 The 1H–1H spin network from an α-methine (δH 4.61) to δ-methylene (δH 3.54, 3.67) in the 1H–1H COSY and 1D-TOCSY experiments (see Supplementary Figures S26–S29), and an α-methine correlated to a carbonyl carbon (δC 169.9) revealed a proline moiety. The COSY spectrum indicated correlations between dimethyl protons (δH 0.85, 0.83) to NH (δH 8.15), and a long-range correlation from an α-methine (δH 4.36) to the carbonyl at δC 170.4, that indicated the presence of a valine moiety. The 1H–1H COSY and 1D-TOCSY correlations from an oxymethine (δH 4.94, δC 75.4) to the isopropyl residue (δH 2.04, 0.92, 0.93) and the HMBC correlation from the oxymethine to a carbonyl carbon (δC 169.4) suggested the presence of a 2-hydroxy-3-methyl-butanoic acid moiety (I). The COSY correlations from the dimethyl protons (δH 0.79, 0.94) to an α-methine (δH 4.67), together with the long-range couplings from the α-methine to the carbonyl at δC 169.7 and from an N-methyl group (δH 3.06) to an α-methine (δC 60.8) indicated the presence of an N-methylvaline moiety. Lastly, COSY correlations supported a connection between branched methyl protons (δH 0.84, 0.88) to an oxymethine (δH 5.23, δC 77.3) while a long-range correlation from the oxymethine to the carbonyl at δC 167.2 suggested another 2-hydroxy-3-methylbutanoic acid moiety (II). The connectivity between these residues was established by the HMBC correlations illustrated in Figure 2 as follows: the NH proton (δH 8.18) of cleonine to the carbonyl carbon (δC 167.2) of 2-hydroxy-3-methyl-butanoic acid (II) with an amide linkage; the oxymethine proton (δH 5.23) of 2-hydroxy-3-methyl-butanoic acid (II) to the carbonyl carbon (δC 169.7) of N-Me-valine with an ester linkage; the N-Me protons (δH 3.06) of N-Me-valine to the carbonyl carbon (δC 169.4) of 2-hydroxy-3-methyl-butanoic acid (I) with an amide linkage, which is opposite to cleonine; the oxymethine proton (δH 4.94) of 2-hydroxy-3-methyl-butanoic acid (I) to the carbonyl carbon (δC 170.4) of valine with an ester linkage; and the NH proton (δH 8.15) of valine to the carbonyl carbon (δC 169.9) of proline with an amide linkage. In addition, the correlation between the α-methine proton of proline and the carbonyl carbon of cleonine was determined by selective inverse multiple bond analysis (SIMBA, see Supplementary Figure S30), which was supported by a 1D-NOE observed between the α-methine of cleonine and the δ-methylene of proline (see Supplementary Figure S31). The structure of VLG-C elucidated from these results is a novel cyclic hexadepsipeptide consisting of cleonine, two molecules of 2-hydroxy-3-methyl-butanoic acid, N-methylvaline, proline and valine (Figure 1).
Structures of VLG-A, V and T
The 1H and 13C NMR data of the VLGs are shown in Table 2. Since the 1H and 13C NMR spectra of VLG-A, T and V indicated that their structures were closely related to each other, they were determined in a similar manner to VLG-C (Supplementary Figure S32). The 1H–1H COSY correlation in VLG-A from an NH (δH 8.03) to an aliphatic methyl group (δH 1.15) and the long-range coupling from an α-methine (δH 4.61) to an amide carbon (δC 172.1) revealed the presence of an alanine moiety. The long-range couplings from the NH of alanine to the carbonyl carbon (δC 167.2) of 2-hydroxy-3-methyl-butanoic acid (II) and the α-methine (δH 4.60) of proline to the carbonyl carbon of alanine revealed VLG-A has an alanine residue replacing the cleonine present in VLG-C.
The 1H–1H COSY correlation in VLG-T from an NH (δH 8.67) to an aliphatic methyl group (δH 1.08), combined with the downfield shift of the carbon signal (δC 66.6) of the β-methine (δH 3.87) indicated a threonine moiety. The long-range coupling from the NH of threonine to the carbonyl carbon (δC 166.5) of 2-hydroxy-3-methyl-butanoic acid (II) and from the α-methine (δH 4.58) of proline to the amide carbon (δC 172.3) of threonine revealed that the structure of VLG-T has a threonine replacing the cleonine in VLG-C.
The 1H–1H COSY correlation in VLG-V from an NH (δH 8.36) to the aliphatic dimethyl group (δH 0.82, 0.82) and the HMBC correlation from an α-methine (δH 4.40) to the carbonyl at δC 170.4 suggested the presence of a valine moiety. The long-range couplings from the NH of valine to the carbonyl carbon (δC 166.9) of 2-hydroxy-3-methyl-butanoic acid (II) and the α-methine (δH 4.63) of proline to the carbonyl carbon of valine revealed that the structure of VLG-V has a valine replacing the cleonine in VLG-C. These correlations provided evidence that VLG-A, T and V have the identical cyclic hexadepsipeptide planar structure of VLG-C, except for the cleonine moiety. Hence, VLGs A, C, T and V are a family of novel cyclic depsipeptides, each possessing one different amino acid, alanine, cleonine, threonine and valine, respectively.
The absolute structures of the VLGs were determined by advanced Marfey’s analysis15 and X-ray crystal structural analysis.
The stereochemistry of the constituent amino acids of VLG-A, C, T and V were determined by the advanced Marfey’s method using L-FDLA15 (see Supplementary Figures S33–S38). The acid hydrolysates of the VLGs, and authentic D- and L-amino acids, were derivatized by treatment with L-FDLA, and analyzed using extracted ion chromatograms by LC-HRESI-MS (Supplementary Table S1). It was revealed that VLG-A contained L-Pro, D-Ala and D-Val; VLG-C contained L-Pro and D-Val; and VLG-T contained L-Pro, D-allo-Thr and D-Val; and VLG-V contained L-Pro and D-Val. The absolute structure of cleonine in VLG-C was determined from the elution patterns of the D- and L-FDLA-coupled derivatives of the VLG-C hydrolysates (see Supplementary Figure S38). 2,4-dinitrophenyl-5-d-Leucineamide (D-DLA)-cleonine (6.42 min) eluted faster than L-DLA-cleonine (7.84 min), indicating that the stereochemistry of cleonine is R.
The absolute configuration of VLG-A was determined by a single-crystal X-ray diffraction analysis (Supplementary Figure S39). A crystal of VLG-A was obtained in H2O/acetonitrile (1:1) at 4 °C for 5 days. The crystal structure revealed that the absolute configuration of the two 2-hydroxy-3-methyl-butanoic acids is S, and the absolute configuration of N-Me-valine is R. Marfey’s derivatives of N-Me-valine in hydrolysates of VLGs-A, C, T and V exhibited the same retention time in advanced Marfey’s analysis indicating that they have the same stereochemistry (see Supplementary Figures S34–S37). The stereochemistry of the other constituent amino-acid residues was in accordance with the results of advanced Marfey’s analysis. The absolute structures of the VLGs as determined are shown in Figure 1.
Biological activity
The antimicrobial activities of the VLGs are summarized together with that of vancomycin in Table 3. None of the VLGs or vancomycin exhibited antibacterial activity against Gram-negative bacteria. VLG-A and T exhibited weak antibacterial activity against Gram-positive bacteria, including Staphylococcus, Enterococcus and Bacillus sp., which was much lower than vancomycin. VLG-C and V did not exhibit any antibacterial activity against the Gram-positive bacteria tested.
The cytotoxicities of the VLGs against human tumor cell lines are summarized in Table 4. VLG-T exhibited the highest cytotoxicity of the VLGs against the tested cell lines with IC50 values from 6.6 to 21.6 μM. VLG-A and C also exhibited cytotoxicity but this was weaker than for VLG-T, IC50 values of 46.6–98.6 and 92.9–167.4 μM, respectively. VLG-V demonstrated almost no cytotoxicity with IC50 values from 187.0 to >250 μM. Although VLG-A and T exhibited similar antibacterial activity, VLG-T exhibited much more potent cytotoxicity against the tested cell lines than VLG-A. Despite having almost no antibacterial activity, VLG-C had moderate cytotoxicity against human tumor cell lines. The fact that the VLGs were more effective against human tumor cell lines than bacterial strains indicates that the VLGs might target mammalian-specific enzymes or molecules.
We isolated and determined the absolute structures of four novel metabolites designated VLG-A, C, T and V from the broth of the soil bacterium Amycolatopsis sp. ML1-hF4. These compounds differ structurally only by one amino acid; the cleonine moiety in VLG-C is replaced by different amino acids in the other molecules. However, despite the structural similarities, the cytotoxicity of the VLGs showed remarkable differences. These differences suggest that a hydrophobic amino acid at the position occupied by cleonine in VLG-C is a disadvantage for interaction with human cell lines.
To our knowledge, this is the first report of a cleonine-containing metabolite from a naturally isolated microorganism without any breeding or mutation treatment. Before discovery of VLG-C, cleomycin, which is produced from a UV-irradiated mutant strain of bleomycin-producing Actinomycetes, was the only known cleonine-containing metabolite.14 The stereochemistry of cleonine in cleomycin was proposed to be S because of the stereochemistry of L-β-hydroxyvaline in the bleomycin analog, YA-56.16, 17 In contrast, the stereochemistry of cleonine in VLG-C was revealed to be R according to the results of advanced Marfey’s analysis. Thus, this is the first discovery of D-cleonine in a natural product. The biosynthesis mechanism of VLG-C and new amino acid, cleonine, is quite interesting.
Experimental procedures
General experimental procedures
The optical rotations of the purified compounds were measured using a P-1030 polarimeter (JASCO, Tokyo, Japan). The IR spectra were recorded with an FT/IR-4100 Fourier transform infrared spectrometer (JASCO). The 1H and 13C NMR spectra were measured with an AVANCE III 500 spectrometer (Bruker, Billerica, MA, USA) and an ECZ600R spectrometer (JEOL RESONANCE, Tokyo, Japan) at 25 °C. All chemical shifts were referenced to the residual solvent peak for DMSO-d6: 2.49 ppm for 1H and 39.5 ppm for 13C. The mass spectra were recorded using a LTQ Orbitrap XL mass spectrometer (Thermo Fisher Scientific, San Jose, CA, USA).
Advanced Marfey’s analysis
VLG-A, C, T and V were treated with 6 M HCl at 110 °C for 18 h. For the analysis of cleonine, VLG-C was treated with 6 M HCl at 80 °C for 24 h. The resulting hydrolysates were dried and coupled with 1-fluoro-2,4-dinitrophenyl-5-L- and D-leucineamide (L- and D-FDLA, Tokyo Chemical Industry Co. Ltd., Tokyo, Japan) as per the manufacturer’s instructions.
LC-HRESIMS was performed using a Capcell Pak C18 MG, 2.1 mm i.d. × 150 mm, 5 μm column (Shiseido Co. Ltd., Tokyo, Japan) at 50 °C, with linear-gradient elution from 20% aqueous acetonitrile containing 0.1% formic acid to 80% aqueous acetonitrile containing 0.1% formic acid over 15 min and maintained at 80% until 20 min at a flow rate of 0.4 ml min−1. The mass spectra were obtained in ESI positive mode (m/z 100 to m/z 1000) and FDLA derivatives were monitored using extracted ion chromatograms (5 ppm tolerance) of the calculated exact mass for each protonated molecule.
Evaluations of antimicrobial activity and cytotoxicity
Minimum inhibitory concentrations against bacteria were determined as previously reported.18 Cytotoxicity was assayed using human cancer cell lines provided by the RIKEN BRC (Tsukuba, Japan) through the National Bio-Resource Project of the MEXT, Japan. The cell lines GSS and NB16 were plated on 96-well microplates at 5.4 × 104 and 1.4 × 105 cells per 0.1 ml per well, respectively, and other cell lines were plated at 2.7 × 104 cells per 0.1 ml per well in Dulbecco’s modified Eagle medium with the supplements described previously.19 After culturing at 37 °C for 24 h, the cells were treated with the VLGs and further cultured for 48 h. The cell viability was assayed by 3-(4,5- dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide assay.20
Single-crystal X-ray analysis
Single-crystal X-ray data were collected on a Rigaku VariMax with a RAPID imaging plate area detector with graphite-monochromated Cu-Kα radiation. Data collection was conducted at 93 K. The structure was solved by direct methods and refined by using full-matrix least-squares in SHELX. All non-hydrogen atoms were refined anisotropically. All hydrogen atoms were placed in standard calculated positions, and were refined isotropically.
Data availability
The X-ray crystallographic data for VLG-A have been deposited at the Cambridge Crystallographic Data Center (CCDC) under the deposition number CCDC 1537061.
Dedication
This article is dedicated to Prof. Hamao Umezawa on the occasion of the 60th anniversary of worldwide marketing of kanamycin.
References
Sharma, M. et al . Actinomycetes: source, identification, and their applications . Int. J. Curr. Microbiol. App. Sci. 3, 801 – 832 ( 2014 ).
Newman, D. J. & Cragg, G. M. Natural products as sources of new drugs from 1981 to 2014 . J. Nat. Prod. 79, 629 – 661 ( 2016 ).
Butler, M. S., Blaskovich, M. A. & Cooper, M. A. Antibiotics in the clinical pipeline at the end of 2015 . J. Antibiot. 70, 3 – 24 ( 2017 ).
Schatz, A., Bugie, E. & Waksman, S. A. Streptomycin, a substance exhibiting antibiotic activity against gram-positive and gram-negative bacteria . Proc. Soc. Exp. Biol. Med. 55, 66 – 69 ( 1944 ).
Umezawa, H. et al . Production and isolation of a new antibiotic: kanamycin . J. Antibiot. 10, 181 – 188 ( 1957 ).
Umezawa, H. Bleomycin and other antitumor antibiotics of high molecular weight . Antimicrob. Agents Chemother. 5, 1079 – 1085 ( 1965 ).
Burg, R. W. et al . Avermectins, new family of potent anthelmintic agents: producing organism and fermentation . Antimicrob. Agents Chemother. 15, 361 – 367 ( 1979 ).
Kino, T. et al . FK-506, a novel immunosuppressant isolated from a Streptomyces. I. Fermentation, isolation, and physico-chemical and biological characteristics . J. Antibiot. 40, 1249 – 1255 ( 1987 ).
Igarashi, M. et al . Pargamicin A, a novel cyclic peptide antibiotic from Amycolatopsis sp . J. Antibiot. 61, 387 – 393 ( 2008 ).
Hashizume, H., Adachi, H., Igarashi, M., Nishimura, Y. & Akamatsu, Y. Biological activities of pargamicin A, a novel cyclic peptide antibiotic from Amycolatopsis sp . J. Antibiot. 63, 279 – 283 ( 2010 ).
Hashizume, H., Sawa, R., Yamashita, K., Nishimura, Y. & Igarashi, M. Structure and antibacterial activities of new cyclic peptide antibiotics, pargamicins B, C and D, from Amycolatopsis sp. ML1-hF4 . J. Antibiot. 70, 699 – 704 ( 2017 ).
Kato, K., Takita, T. & Umezawa, H. Synthesis of cleonine, amino(1-hydroxycyclopropyl)acetic acid, a novel amino acid contained in cleomycin . Tetrahedron Lett. 21, 4925 – 4926 ( 1980 ).
Gaillet, C., Lequart, C., Debeire, P. & Nuzillard, J. M. Band-selective HSQC and HMBC experiments using excitation sculpting and PFGSE . J Magn Reson. 139, 454 – 459 ( 1999 ).
Umezawa, H., Muraoka, Y., Fujii, A., Naganawa, H. & Takita, T. Chemistry of bleomycin. XXVII. Cleomycin, a new family of bleomycin-phleomycin group . J. Antibiot. 33, 1079 – 1082 ( 1980 ).
Harada, K. et al . Application of D, L-FDLA derivatization to determination of absolute configuration of constituent amino acids in peptide by advanced Marfey's method . Tetrahedron Lett. 37, 3001 – 3004 ( 1996 ).
Ito, Y. et al . The antibiotic YA-56 complex: isolation, purification and physicochemical properties of the main components . J. Antibiot. 26, 77 – 83 ( 1973 ).
Ohashi, Y., Abe, H. & Ito, Y. β-Hydroxy-L-valine and 4-amino-3,6-dihydroxy-2-methylhexanoic acid, constitutional amino acids of the antibiotic YA-56 . Agr. Biol. Chem. 37, 2283 – 2287 ( 1973 ).
Japan Society of Chemotherapy Method of MIC determination . Chemotherapy 29, 77 – 79 ( 1981 ).
Wada, S. et al . Novel autophagy inducers lentztrehaloses A, B and C . J. Antibiot. 68, 521 – 529 ( 2015 ).
Mosmann, T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays . J. Immunol. Methods 65, 55 – 63 ( 1983 ).
Acknowledgements
The authors are grateful to the following researchers working at the Institute of Microbial Chemistry (BIKAKEN): Ms Yumiko Kubota for the measurement of NMR spectra; Ms Yuko Shibuya for the measurement of MICs; Ms Shigeko Harada for her technical assistance; Ms Miho Nagayoshi for the measurement of cytotoxicity; and Dr Yoshio Nishimura and Dr Hayamitsu Adachi for valuable discussions. This work was supported by the Japan Society for the Promotion of Science KAKENHI, grant number 16K08338.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on The Journal of Antibiotics website
Supplementary information
Rights and permissions
About this article
Cite this article
Hashizume, H., Iijima, K., Yamashita, K. et al. Valgamicin C, a novel cyclic depsipeptide containing the unusual amino acid cleonine, and related valgamicins A, T and V produced by Amycolatopsis sp. ML1-hF4. J Antibiot 71, 129–134 (2018). https://doi.org/10.1038/ja.2017.135
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ja.2017.135
This article is cited by
-
Suertides A–C: selective antibacterial cyclic hexapeptides from Amycolatopsis sp. MST-135876v3
The Journal of Antibiotics (2022)
-
Integrating vectors for genetic studies in the rare Actinomycete Amycolatopsis marina
BMC Biotechnology (2019)