Abstract
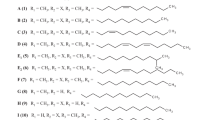
Novel antibiotics, active against acid-fast bacteria, caprazamycins, were isolated from the culture broth of Streptomyces sp. MK730-62F2. The planar structures of the compounds were determined by 2D NMR spectroscopic study. Furthermore, the absolute structure of caprazamycin B (2) was established by NMR spectroscopy and X-ray crystallography of its degradation products and by total synthesis of the 5-amino-5-deoxy-D-ribose moiety. In the course of degradation studies of 2 under alkaline and acidic conditions, we obtained the two core components, caprazene (11) and caprazol (14), respectively, in high yield.Structurally, caprazamycins belong to a family of lipo-uridyl antibiotics, which have been discovered as specific inhibitors of a bacterial translocase.
Similar content being viewed by others
Article PDF
References
Igarashi M, Nakagawa N, Hattori S, Doi N, Masuda T, Yamazaki T, Miyake T, Naganawa H, Ishizuka M, Shomura T, Omoto S, Yano I, Hamada M, Takeuchi T . Caprazamycins A∼F, novel anti-TB antibiotics, from Streptomyces sp., 42nd Interscience Conference on Antimicrobial Agents and Chemotherapy (San Diego), Abstracts F-2031, Sept. 27–30 ( 2002)
Igarashi M, Nakagawa N, Doi N, Hattori S, Naganawa H, Hamada M . Caprazamycin B, Novel Anti-TB Antibiotics, from Streptomyces sp. J Antibiot 56: 580–583 ( 2003)
Igarashi M, Nakagawa N, Hattori S, Kinoshita N, Masuda T, Hamada M, Akamatsu Y . Caprazamycins, novel lipo-nucleoside antibiotics, from Streptomyces sp. I. Taxonomy, production, isolation and biological properties. J Antibiot, in preparation.
Doi N, Igarashi M, Masuda T, Hattori S, Nakagawa N, Hamada M, Yano I, Akamatsu Y . Antimicrob Agents Chemother, in preparation.
Ubukata M, Kimura K, Isono K, Nelson CC, Gregson JM, Mcclosky JA . Structure elucidation of liposidomycins, a class of complex lipid nucleoside antibiotics. J Org Chem 57: 6392–6403 ( 1992)
Drautz H, Reuschenbach P, Zähner H, Rohr J, Zeeck A . Metabolic products of microorganisms. 225 Elloramycin, a new anthracycline-like antibiotic from Streptomyces olivaceus. J Antibiot 38: 1291–1301 ( 1985)
Toman R, Karácsonyi S, Palovcík R . New syntheses of mono- and di-O-methyl derivatives of methyl α-L-rhamnopyranoside. Carbohydr Res 56: 191–194 ( 1977)
Haverkamp J, De Bie MJA, Vliegenthart JFG . 13C- and 1H-NMR spectroscopy of permethylated gluco-, galacto-, and manno-pyranoses and their 6-deoxy analogues. Carbohydr Res 39: 201–211 ( 1975)
Bock K, Lundt I, Pedersen C . Assignment of anomer structure to carbohydrates through geminal 13C-H coupling constants. Tetrahedron Lett 13: 1037–1040 ( 1973)
Ewing DF, Goethals G, Mackenzie G, martin P, Ronco G, Vanbaelinghem L, Villa P . Novel reversed cyclonucleoside analogues with a D-ribofiranose glycone. Carbohydr Res 321: 190–196 ( 1999)
Sarabia-García F, López-Herrera FJ . Studies on the synthesis of tunicamycin. The preparation of 7-deoxy-2-deamino-6-hydroxy tunicamine and related products. Tetrahedron 52: 4757–4768 ( 1996)
Mcdonald L, Barbieri L, Carter G, Lenoy E, Lotvin J, Petersen P, Siegel M, Singh G, Williamson R . Structures of the muraymycins, novel peptidoglycan biosynthesis inhibitors. J Am Chem Soc 124: 10260–10261 ( 2002)
Yamaguchi H, Sato S, Yoshida S, Takada K, Itoh M, Seto H, Otake N . Capuramycin, a new nucleoside antibiotic. Taxonomy, fermentation, isolation and characterization. J Antibiot 39: 1047–1053 ( 1986)
Muramatsu Y, Muramatsu A, Ohnuki T, Ishii M, Kizuka M, Enokita R, Tsutsumi S, Arai M, Ogawa Y, Suzuki T, Takatsu T, Inukai M . Studies on novel bacterial translocase I inhibitors, A-500359s. I. Taxonomy, fermentation, isolation, physico-chemical properties and structure elucidation of A-500359 A, C, D and G. J Antibiot 56: 243–252 ( 2003)
Muramatsu Y, Miyakoshi S, Ogawa Y, Ohnuki T, Ishii M, Takatsu T, Inukai M . Studies on novel bacterial translocase I inhibitors, A-500359s. III. Deaminocaprolactam derivatives of capuramycin: A-500359 E, F, H; M-1 and M-2. J Antibiot 56: 259–267 ( 2003)
Altomare A, Cascarano M, Giacovazzo G, Guagliardi A, Burla MC, Polidori G, Camalli M . SIR92- a program for automatic solution of crystal structures by direct methods. J Appl Cryst 27: 435 ( 1994)
Beurskens PT, Admiraal G, Beurskens G, Bosman WP, de Gelder R, Israel R, Smits JMM . The DIRDIF-94 program system, Technical Report of the Crystallography Laboratory, University of Nijmegen, The Netherlands ( 1994)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Igarashi, M., Takahashi, Y., Shitara, T. et al. Caprazamycins, Novel Lipo-nucleoside Antibiotics, from Streptomyces sp.. J Antibiot 58, 327–337 (2005). https://doi.org/10.1038/ja.2005.41
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ja.2005.41
Keywords
This article is cited by
-
Application of antifungal metabolites from Streptomyces philanthi RL-1-178 for maize grain coating formulations and their efficacy as biofungicide during storage
World Journal of Microbiology and Biotechnology (2023)
-
Efficacy of the antifungal metabolites of Streptomyces philanthi RL-1-178 on aflatoxin degradation with its application to prevent aflatoxigenic fungi in stored maize grains and identification of the bioactive compound
World Journal of Microbiology and Biotechnology (2023)
-
Origin of the 3-methylglutaryl moiety in caprazamycin biosynthesis
Microbial Cell Factories (2022)
-
Synthesis of macrocyclic nucleoside antibacterials and their interactions with MraY
Nature Communications (2022)
-
Peculiarities of promiscuous l-threonine transaldolases for enantioselective synthesis of β-hydroxy-α-amino acids
Applied Microbiology and Biotechnology (2021)