Abstract
Background/Objectives:
The aim of this study was to characterize the effects of Maresin 1 (MaR1) in obesity-related liver steatosis and the mechanisms involved.
Methods:
MaR1 effects on fatty liver disease were tested in ob/ob (2–10 μg kg−1 i.p., 20 days) and in diet-induced obese (DIO) mice (2 μg kg−1, i.p., or 50 μg kg−1, oral gavage for 10 days), as well as in cultured hepatocytes.
Results:
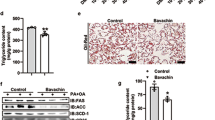
In ob/ob mice, MaR1 reduced liver triglycerides (TG) content, fatty acid synthase (FAS) and stearoyl-CoA desaturase-1 protein expression, while increased acetyl-CoA carboxylase (ACC) phosphorylation and LC3II protein expression, in parallel with a drop in p62 levels. Similar effects on hepatic TG, ACC phosphorylation, p62 and LC3II were observed in DIO mice after MaR1 i.p. injection. Interestingly, oral gavage of MaR1 also decreased serum transaminases, reduced liver weight and TG content. MaR1-treated mice exhibited reduced hepatic lipogenic enzymes content (FAS) or activation (by phosphorylation of ACC), accompanied by upregulation of carnitine palmitoyltransferase (Cpt1a), acyl-coenzyme A oxidase (Acox1) and autophagy-related proteins 5 and 7 (Atg5–7) gene expression, along with increased number of autophagic vacuoles and reduced p62 protein levels. MaR1 also induced AMP-activated protein kinase (AMPK) phosphorylation in DIO mice and in primary hepatocytes, and AMPK inhibition completely blocked MaR1 effects on Cpt1a, Acox1, Atg5 and Atg7 expression.
Conclusions:
MaR1 ameliorates liver steatosis by decreasing lipogenic enzymes, while inducing fatty acid oxidation genes and autophagy, which could be related to AMPK activation. Thus, MaR1 may be a new therapeutic candidate for reducing fatty liver in obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
WHO. New WHO report: deaths from noncommunicable diseases on the rise, with developing world hit hardest. Cent Eur J Public Health 2011; 19: 114–120.
Lazo M, Clark JM . The epidemiology of nonalcoholic fatty liver disease: a global perspective. Semin Liver Dis 2008; 28: 339–350.
Jornayvaz FR, Shulman GI . Diacylglycerol activation of protein kinase Cepsilon and hepatic insulin resistance. Cell Metab 2012; 15: 574–584.
Van De Wier B, Koek GH, Bast A, Haenen GR . The potential of flavonoids in the treatment of non-alcoholic fatty liver disease. Crit Rev Food Sci Nutr 2017; 57: 834–855.
Polyzos SA, Kountouras J, Zavos C, Deretzi G . Nonalcoholic fatty liver disease: multimodal treatment options for a pathogenetically multiple-hit disease. J Clin Gastroenterol 2012; 46: 272–284.
Tilg H, Moschen AR . Evolution of inflammation in nonalcoholic fatty liver disease: the multiple parallel hits hypothesis. Hepatology 2010; 52: 1836–1846.
Musso G, Gambino R, Cassader M . Recent insights into hepatic lipid metabolism in non-alcoholic fatty liver disease (NAFLD). Prog Lipid Res 2009; 48: 1–26.
Singh R, Cuervo AM . Lipophagy: connecting autophagy and lipid metabolism. Int J Cell Biol 2012; 2012: 282041.
Singh R, Kaushik S, Wang Y, Xiang Y, Novak I, Komatsu M et al. Autophagy regulates lipid metabolism. Nature 2009; 458: 1131–1135.
Liu HY, Han J, Cao SY, Hong T, Zhuo D, Shi J et al. Hepatic autophagy is suppressed in the presence of insulin resistance and hyperinsulinemia: inhibition of FoxO1-dependent expression of key autophagy genes by insulin. J Biol Chem 2009; 284: 31484–31492.
Yang L, Li P, Fu S, Calay ES, Hotamisligil GS . Defective hepatic autophagy in obesity promotes ER stress and causes insulin resistance. Cell Metab 2010; 11: 467–478.
Gonzalez-Periz A, Claria J . Resolution of adipose tissue inflammation. ScientificWorldJournal 2010; 10: 832–856.
White PJ, St-Pierre P, Charbonneau A, Mitchell PL, St-Amand E, Marcotte B et al. Protectin DX alleviates insulin resistance by activating a myokine-liver glucoregulatory axis. Nat Med 2014; 20: 664–669.
Serhan CN . Pro-resolving lipid mediators are leads for resolution physiology. Nature 2014; 510: 92–101.
Titos E, Claria J . Omega-3-derived mediators counteract obesity-induced adipose tissue inflammation. Prostaglandins Other Lipid Mediat 2013; 107: 77–84.
Neuhofer A, Zeyda M, Mascher D, Itariu BK, Murano I, Leitner L et al. Impaired local production of proresolving lipid mediators in obesity and 17-HDHA as a potential treatment for obesity-associated inflammation. Diabetes 2013; 62: 1945–1956.
Gonzalez-Periz A, Horrillo R, Ferre N, Gronert K, Dong B, Moran-Salvador E et al. Obesity-induced insulin resistance and hepatic steatosis are alleviated by omega-3 fatty acids: a role for resolvins and protectins. FASEB J 2009; 23: 1946–1957.
Hellmann J, Tang Y, Kosuri M, Bhatnagar A, Spite M . Resolvin D1 decreases adipose tissue macrophage accumulation and improves insulin sensitivity in obese-diabetic mice. FASEB J 2011; 25: 2399–2407.
Martinez-Fernandez L, Gonzalez-Muniesa P, Laiglesia LM, Sainz N, Prieto-Hontoria PL, Escote X et al. Maresin 1 improves insulin sensitivity and attenuates adipose tissue inflammation in ob/ob and diet-induced obese mice. FASEB J 2017; 31: 2135–2145.
Laiglesia LM, Lorente-Cebrian S, Lopez-Yoldi M, Lanas R, Martinez JA, Moreno-Aliaga MJ . Maresin 1 inhibits TNF-alpha-induced lipolysis and autophagy in 3T3-L1 adipocytes. J Cell Physiol 2017; e-pub ahead of print 13 July 2017; doi: 10.1002/jcp.26096.
Rius B, Titos E, Moran-Salvador E, Lopez-Vicario C, Garcia-Alonso V, Gonzalez-Periz A et al. Resolvin D1 primes the resolution process initiated by calorie restriction in obesity-induced steatohepatitis. FASEB J 2014; 28: 836–848.
Li R, Wang Y, Zhao E, Wu K, Li W, Shi L et al. Maresin 1, a proresolving lipid mediator, mitigates carbon tetrachloride-induced liver injury in mice. Oxid Med Cell Longev 2016; 2016: 9203716.
Friedewald WT, Levy RI, Fredrickson DS . Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972; 18: 499–502.
Folch J, Lees M, Sloane Stanley GH . A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 1957; 226: 497–509.
Bejarano E, Yuste A, Patel B, Stout RF Jr, Spray DC, Cuervo AM . Connexins modulate autophagosome biogenesis. Nat Cell Biol 2014; 16: 401–414.
Rodriguez-Ortigosa CM, Celay J, Olivas I, Juanarena N, Arcelus S, Uriarte I et al. A GAPDH-mediated trans-nitrosylation pathway is required for feedback inhibition of bile salt synthesis in rat liver. Gastroenterology 2014; 147: 1084–1093.
Nevado C, Valverde AM, Benito M . Role of insulin receptor in the regulation of glucose uptake in neonatal hepatocytes. Endocrinology 2006; 147: 3709–3718.
Klionsky DJ, Abdelmohsen K, Abe A, Abedin MJ, Abeliovich H, Acevedo Arozena A et al. Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy 2016; 12: 1–222.
Lopez-Yoldi M, Fernandez-Galilea M, Laiglesia LM, Larequi E, Prieto J, Martinez JA et al. Cardiotrophin-1 stimulates lipolysis through the regulation of main adipose tissue lipases. J Lipid Res 2014; 55: 2634–2643.
Laiglesia LM, Lorente-Cebrian S, Prieto-Hontoria PL, Fernandez-Galilea M, Ribeiro SM, Sainz N et al. Eicosapentaenoic acid promotes mitochondrial biogenesis and beige-like features in subcutaneous adipocytes from overweight subjects. J Nutr Biochem 2016; 37: 76–82.
Ghislat G, Aguado C, Knecht E . Annexin A5 stimulates autophagy and inhibits endocytosis. J Cell Sci 2012; 125 (Pt 1): 92–107.
Yamada E, Singh R . Mapping autophagy on to your metabolic radar. Diabetes 2012; 61: 272–280.
Borgeson E, Johnson AM, Lee YS, Till A, Syed GH, Ali-Shah ST et al. Lipoxin A4 Attenuates Obesity-Induced Adipose Inflammation and Associated Liver and Kidney Disease. Cell Metab 2015; 22: 125–137.
Perez-Echarri N, Perez-Matute P, Marcos-Gomez B, Marti A, Martinez JA, Moreno-Aliaga MJ . Down-regulation in muscle and liver lipogenic genes: EPA ethyl ester treatment in lean and overweight (high-fat-fed) rats. J Nutr Biochem 2009; 20: 705–714.
de Castro GS, Cardoso JF, Calder PC, Jordao AA, Vannucchi H . Fish oil decreases hepatic lipogenic genes in rats fasted and refed on a high fructose diet. Nutrients 2015; 7: 1644–1656.
de Castro GS, Deminice R, Simoes-Ambrosio LM, Calder PC, Jordao AA, Vannucchi H . Dietary docosahexaenoic acid and eicosapentaenoic acid influence liver triacylglycerol and insulin resistance in rats fed a high-fructose diet. Mar Drugs 2015; 13: 1864–1881.
Bargut TC, Frantz ED, Mandarim-de-Lacerda CA, Aguila MB . Effects of a diet rich in n-3 polyunsaturated fatty acids on hepatic lipogenesis and beta-oxidation in mice. Lipids 2014; 49: 431–444.
Scapagnini G, Davinelli S, Kaneko T, Koverech G, Koverech A, Calabrese EJ et al. Dose response biology of resveratrol in obesity. J Cell Commun Signal 2014; 8: 385–391.
Azhar Y, Parmar A, Miller CN, Samuels JS, Rayalam S . Phytochemicals as novel agents for the induction of browning in white adipose tissue. Nutr Metab (Lond) 2016; 13: 89.
Dobrzyn P, Dobrzyn A, Miyazaki M, Cohen P, Asilmaz E, Hardie DG et al. Stearoyl-CoA desaturase 1 deficiency increases fatty acid oxidation by activating AMP-activated protein kinase in liver. Proc Natl Acad Sci USA 2004; 101: 6409–6414.
Ceddia RB . The role of AMP-activated protein kinase in regulating white adipose tissue metabolism. Mol Cell Endocrinol 2013; 366: 194–203.
Monsenego J, Mansouri A, Akkaoui M, Lenoir V, Esnous C, Fauveau V et al. Enhancing liver mitochondrial fatty acid oxidation capacity in obese mice improves insulin sensitivity independently of hepatic steatosis. J Hepatol 2012; 56: 632–639.
Orellana-Gavalda JM, Herrero L, Malandrino MI, Paneda A, Sol Rodriguez-Pena M, Petry H et al. Molecular therapy for obesity and diabetes based on a long-term increase in hepatic fatty-acid oxidation. Hepatology 2011; 53: 821–832.
Codogno P, Meijer AJ . Autophagy: a potential link between obesity and insulin resistance. Cell Metab 2010; 11: 449–451.
Gonzalez-Rodriguez A, Mayoral R, Agra N, Valdecantos MP, Pardo V, Miquilena-Colina ME et al. Impaired autophagic flux is associated with increased endoplasmic reticulum stress during the development of NAFLD. Cell Death Dis 2014; 5: e1179.
Chen Y, Xu C, Yan T, Yu C, Li Y . omega-3 Fatty acids reverse lipotoxity through induction of autophagy in nonalcoholic fatty liver disease. Nutrition 2015; 31: 1423–1429 e2.
Lopez-Vicario C, Alcaraz-Quiles J, Garcia-Alonso V, Rius B, Hwang SH, Titos E et al. Inhibition of soluble epoxide hydrolase modulates inflammation and autophagy in obese adipose tissue and liver: role for omega-3 epoxides. Proc Natl Acad Sci USA 2015; 112: 536–541.
Alers S, Loffler AS, Wesselborg S, Stork B . Role of AMPK-mTOR-Ulk1/2 in the regulation of autophagy: cross talk, shortcuts, and feedbacks. Mol Cell Biol 2012; 32: 2–11.
Fernandez-Galilea M, Perez-Matute P, Prieto-Hontoria PL, Sainz N, Lopez-Yoldi M, Houssier M et al. Alpha-lipoic acid reduces fatty acid esterification and lipogenesis in adipocytes from overweight/obese subjects. Obesity (Silver Spring) 2014; 22: 2210–2215.
Zhou G, Myers R, Li Y, Chen Y, Shen X, Fenyk-Melody J et al. Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest 2001; 108: 1167–1174.
Jelenik T, Rossmeisl M, Kuda O, Jilkova ZM, Medrikova D, Kus V et al. AMP-activated protein kinase alpha2 subunit is required for the preservation of hepatic insulin sensitivity by n-3 polyunsaturated fatty acids. Diabetes 2010; 59: 2737–2746.
Jung TW, Kim HC, Jeong JH . Protectin DX suppresses hepatic gluconeogenesis through AMPK-HO-1-mediated inhibition of ER stress. Cell Signal 2017; 34: 133–140.
Acknowledgements
This work was supported by the Ministry of Economy and Competitiveness of the Government of Spain (grant numbers BFU2012-36089 to MJM-A, BFU2015-65937-R (MINECO/FEDER)) to MJM-A and SL-C; Department of Health of the Navarra Government (grant number 67-2015) to MJM-A; CIBER Physiopathology of Obesity and Nutrition (CIBERobn), Carlos III Health Research Institute (grant number CB12/03/30002). LML is supported by a predoctoral fellowship from Asociación de Amigos (Universidad de Navarra) and from ‘la Caixa’ Banking Foundation. LMF is supported by an FPI predoctoral fellowship (Formación de Personal Investigador). We would like to thank Asunción Redín, María Zabala, Javier García, Eukene Vélaz, David García-Ros and Sara Arcelus for their valuable technical support on this project.
Author contributions
MJM-A and SL-C designed the research study; LML, LM-F, NS and PLP-H conducted the animal experiments; LML and NS performed mRNA expression and protein experiments; LML and MAB analyzed histological images; LML and CMR-O carried out the primary hepatocytes isolation and culture; LML, MJM-A and SL-C wrote the main manuscript; and JAM revised the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Laiglesia, L., Lorente-Cebrián, S., Martínez-Fernández, L. et al. Maresin 1 mitigates liver steatosis in ob/ob and diet-induced obese mice. Int J Obes 42, 572–579 (2018). https://doi.org/10.1038/ijo.2017.226
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2017.226
This article is cited by
-
One-anastomosis gastric bypass modulates the serum levels of pro- and anti-inflammatory oxylipins, which may contribute to the resolution of inflammation
International Journal of Obesity (2022)
-
Brown adipose tissue-derived MaR2 contributes to cold-induced resolution of inflammation
Nature Metabolism (2022)
-
Low serum Maresin-1 levels are associated with non-alcoholic fatty liver disease: a cross-sectional study
Lipids in Health and Disease (2021)
-
Maresin 1 regulates insulin signaling in human adipocytes as well as in adipose tissue and muscle of lean and obese mice
Journal of Physiology and Biochemistry (2021)
-
Special Issue: 2019 Consortium for Trans-Pyrenean Investigations on Obesity and Diabetes
Journal of Physiology and Biochemistry (2021)