Abstract
Background:
Growth trajectories have shown to be related to obesity and metabolic risks in later life, however body mass index (BMI) trajectories according to the presence or absence of metabolic syndrome (MS) and its parameters in adulthood are scarce in literature.
Objectives:
To investigate BMI trajectories during childhood in relation to MS and its parameters in adult age.
Methods:
A total of 1919 subjects (43.4% male, 20–60 y) participated in this retrospective cohort study. Height, weight, waist circumference (WC), blood glucose, high-density lipoprotein cholesterol, triglycerides and blood pressure were measured at adulthood. Childhood weight and height were collected retrospectively from health booklets. Differences between BMI growth curves of subjects with and without MS were assessed using mixed models for correlated data.
Results:
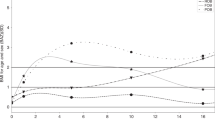
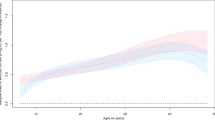
BMI trajectories differed according to the presence or not of MS at adulthood, from the age of 4 years forward (all P<0.05), to the presence or not of hypertriglyceridemia from 1.5 years forward (all P<0.05), and to WC>94 cm (men) / 80 cm (women) compared to lower WC, at all ages (all P<0.05).
Conclusions:
BMI growth curves differ according to the presence or not of MS at adulthood, but differences only appeared after the age of 4 years. Changes vary according to the MS parameters considered. Deviation of the MS-associated BMI curve from normal pattern could correspond to alteration in body composition. These differences in BMI trajectories during childhood support the theory of an early origin of the MS, justifying early prevention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
World Health Organization (WHO). Global Status Report on Noncommunicable Diseases 2014. World Health Organization: Geneva, Switzerland. 2014, p 302..
Dulloo AG, Antic V, Yang Z, Montani JP . Propellers of growth trajectories to obesity and the metabolic syndrome. Int J Obes 2006; 30 (Suppl 4): S1–S3.
Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009; 120: 1640–1645.
Owen CG, Whincup PH, Orfei L, Chou QA, Rudnicka AR, Wathern AK et al. Is body mass index before middle age related to coronary heart disease risk in later life? Evidence from observational studies. Int J Obes 2009; 33: 866–877.
Bays HE . Adiposopathy is ‘sick fat’ a cardiovascular disease? J Am Coll Cardiol 2011; 57: 2461–2473.
Börnhorst C, Tilling K, Russo P, Kourides Y, Michels N, Molnár D et al. Associations between early body mass index trajectories and later metabolic risk factors in European children: the IDEFICS study. Eur J Epidemiol 2015; 31: 513–525.
Rochlani Y, Pothineni NV, Mehta JL . Metabolic syndrome: does it differ between women and men? Cardiovasc Drugs Ther 2015; 29: 329–338.
Eriksson JG, Osmond C, Kajantie E, Forsén TJ, Barker DJ . Patterns of growth among children who later develop type 2 diabetes or its risk factors. Diabetologia 2006; 49: 2853–2858.
Hales CN, Barker DJ . The thrifty phenotype hypothesis. Br Med Bull 2001; 60: 5–20.
Rolland-Cachera MF, Deheeger M, Maillot M, Bellisle F . Early adiposity rebound: causes and consequences for obesity in children and adults. Int J Obes 2006; 30 (Suppl 4): S11–S17.
Hanley B, Dijane J, Fewtrell M, Grynberg A, Hummel S, Junien C et al. Metabolic imprinting, programming and epigenetics - a review of present priorities and future opportunities. Br J Nutr 2010; 104 (Suppl 1): S1–25.
Rolland-Cachera MF, Sempé M, Guilloud-Bataille M, Patois E, Péquignot-Guggenbuhl F, Fautrad V . Adiposity indices in children. Am J Clin Nutr 1982; 36: 178–184.
Rolland-Cachera MF, Akrout M, Péneau S. History and meaning of the body mass index. Interest of other anthropometric measurements. In: Frelut ML (ed.). The ECOG’s eBook on Child and Adolescent Obesity, European Childhood Obesity Group (ECOG). Belgium. 2015. Available at: ecog-obesity.eu/chapter-growth-charts-body-composition/history-meaning-body-mass-index-interest-anthropometric-measurements/..
Eriksson JG . Early growth and coronary heart disease and type 2 diabetes: findings from the Helsinki Birth Cohort Study (HBCS). Am J Clin Nutr 2011; 94 (6 Suppl): 1799S–1802S.
Rolland-Cachera MF, Deheeger M, Bellisle F, Sempé M, Guilloud-Bataille M, Patois E . Adiposity rebound in children: a simple indicator for predicting obesity. Am J Clin Nutr 1984; 39: 129–135.
Sovio U, Kaakinen M, Tzoulaki I, Das S, Ruokonen A, Pouta A et al. How do changes in body mass index in infancy and childhood associate with cardiometabolic profile in adulthood? Findings from the Northern Finland Birth Cohort 1966 Study. Int J Obes 2014; 38: 53–59.
Koyama S, Ichikawa G, Kojima M, Shimura N, Sairenchi T, Arisaka O . Adiposity rebound and the development of metabolic syndrome. Pediatrics 2014; 133: e114–e119.
Péneau S, González-Carrascosa R, Gusto G, Goxe D, Lantieri O, Fezeu L et al. Age at adiposity rebound: determinants and association with nutritional status and the metabolic syndrome at adulthood. Int J Obes 2016; 40: 1150–1156.
Hales CN, Barker DJP . Type 2 (non-insulin-dependent) diabetes mellitus: the thrifty phenotype hypothesis. Diabetologia 1992; 35: 595–601.
Eriksson JG, Forsén T, Tuomilehto J, Osmond C, Barker DJ . Early growth and coronary heart disease in later life: longitudinal study. BMJ 2001; 322: 949–953.
Bhargava SK, Sachdev HS, Fall CH, Osmond C, Lakshmy R, Barker DJ et al. Relation of serial changes in childhood body-mass index to impaired glucose tolerance in young adulthood. N Engl J Med 2004; 350: 865–875.
Ekelund U, Ong KK, Linné Y, Neovius M, Brage S, Dunger DB et al. Association of weight gain in infancy and early childhood with metabolic risk in young adults. J Clin Endocrinol Metab 2007; 92: 98–103.
Howe LD, Tilling K, Benfield L, Logue J, Sattar N, Ness AR et al. Changes in ponderal index and body mass index across childhood and their associations with fat mass and cardiovascular risk factors at age 15. PLoS ONE 2010; 5: e15186.
Liem ET, van Buuren S, Sauer PJ, Jaspers M, Stolk RP, Reijneveld SA . Growth during infancy and childhood, and adiposity at age 16 years: ages 2 to 7 years are pivotal. J Pediatr 2013; 162: 287–92.e2.
Ventura AK, Loken E, Birch LL . Developmental trajectories of girls' BMI across childhood and adolescence. Obesity (Silver Spring) 2009; 17: 2067–2074.
Huang RC, de Klerk NH, Smith A, Kendall GE, Landau LI, Mori TA et al. Lifecourse childhood adiposity trajectories associated with adolescent insulin resistance. Diabetes Care 2011; 34: 1019–1025.
Ziyab AH, Karmaus W, Kurukulaaratchy RJ, Zhang H, Arshad SH . Developmental trajectories of body mass index from infancy to 18 years of age: prenatal determinants and health consequences. J Epidemiol Community Health 2014; 68: 934–941.
Fall CH, Sachdev HS, Osmond C, Lakshmy R, Biswas SD, Prabhakaran D et alNew Delhi Birth Cohort. Adult metabolic syndrome and impaired glucose tolerance are associated with different patterns of BMI gain during infancy: data from the New Delhi Birth Cohort. Diabetes Care 2008; 31: 2349–2356.
Bihan H, Laurent S, Sass C, Nguyen G, Huot C, Moulin JJ et al. Association among individual deprivation, glycemic control, and diabetes complications: the EPICES score. Diabetes Care 2005; 28: 2680–2685.
Bulik CM, Wade TD, Heath AC, Martin NG, Stunkard AJ, Eaves LJ . Relating body mass index to figural stimuli: population-based normative data for Caucasians. Int J Obes Relat Metab Disord 2001; 25: 1517–1524.
Lohman TG, Roche AF, Martorell R . Anthropometric standardization reference manual. Human Kinetics Books: Champaign, IL, USA p 177 1988.
Friedewald WT, Levy RI, Fredrickson DS . Estimation of the concentration of low density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972; 18: 499–502.
Ylihärsilä H, Kajantie E, Osmond C, Forsén T, Barker DJ, Eriksson JG . Body mass index during childhood and adult body composition in men and women aged 56-70 y. Am J Clin Nutr 2008; 87: 1769–1775.
Rolland-Cachera MF, Péneau S . Growth trajectories associated with adult obesity. World Rev Nutr Diet 2013; 106: 127–134.
Knittle JL, Timmers K, Ginsberg-Fellner F, Brown RE, Katz DP . The growth of adipose tissue in children and adolescents. Cross-sectional and longitudinal studies of adipose cell number and size. J Clin Invest 1979; 63: 239–246.
Bouhours-Nouet N, Dufresne S, de Casson FB, Mathieu E, Douay O, Gatelais F et al. High birth weight and early postnatal weight gain protect obese children and adolescents from truncal adiposity and insulin resistance: metabolically healthy but obese subjects? Diabetes Care 2008; 31: 1031–1036.
Whitaker RC, Pepe MS, Wright JA, Seidel KD, Dietz WH . Early adiposity rebound and the risk of adult obesity. Pediatrics 1998; 101: E5.
Rolland-Cachera MF, Maillot M, Deheeger M, Souberbielle JC, Péneau S, Hercberg S . Association of nutrition in early life with body fat and serum leptin at adult age. Int J Obes 2013; 37: 1116–1122.
Michaelsen KF, Jørgensen MH . Dietary fat content and energy density during infancy and childhood: the effect on energy intake and growth. Eur J Clin Nutr 1995; 49: 467–483.
Chivers P, Hands B, Parker H, Bulsara M, Beilin LJ, Kendall GE et al. Body mass index, adiposity rebound and early feeding in a longitudinal cohort (Raine Study). Int J Obes 2010; 34: 1169–1176.
Rolland-Cachera MF, Scaglioni S. Role of nutrients in promoting adiposity development. In: Frelut ML (ed.). The ECOG’s eBook on Child and Adolescent Obesity, European Childhood Obesity Group (ECOG). Belgium. 2015. Available at: ebook.ecog-obesity.eu/chapter-nutrition-food-choices-eating-behavior/role-nutrients-promoting-adiposity-development/..
Michaelsen KF, Greer FR . Protein needs early in life and long-term health. Am J Clin Nutr 2014; 99: 718S–722S.
Singhal A, Lucas A . Early origins of cardiovascular disease: is there a unifying hypothesis? Lancet 2004; 363: 1642–1645.
Barker DJ, Osmond C, Kajantie E, Eriksson JG . Growth and chronic disease: findings in the Helsinki Birth Cohort. Ann Hum Biol 2009; 36: 445–458.
Dulloo AG, Jacquet J, Montani JP . Pathways from weight fluctuations to metabolic diseases: focus on maladaptive thermogenesis during catch-up fat. Int J Obes Relat Metab Disord 2002; 26 (Suppl 2): S46–S57.
Monteiro PO, Victora CG . Rapid growth in infancy and childhood and obesity in later life – a systematic review. Obes Rev 2005; 6: 143–154.
Leunissen RW, Kerkhof GF, Stijnen T, Hokken-Koelega A . Timing and tempo of first-year rapid growth in relation to cardiovascular and metabolic risk profile in early adulthood. JAMA 2009; 301: 2234–2242.
Skinner AC, Mayer ML, Flower K, Perrin EM, Weinberger M . Using BMI to determine cardiovascular risk in childhood: how do the BMI cutoffs fare? Pediatrics 2009; 124: e905–e912.
Taylor RW, Goulding A, Lewis-Barned NJ, Williams SM . Rate of fat gain is faster in girls undergoing early adiposity rebound. Obes Res 2004; 12: 1228–1230.
Falaschetti E, Hingorani AD, Jones A, Charakida M, Finer N, Whincup P et al. Adiposity and cardiovascular risk factors in a large contemporary population of pre-pubertal children. Eur Heart J 2010; 31: 3063–3072.
Czernichow S, Vergnaud AC, Maillard-Teyssier L, Peneau S, Bertrais S, Méjean C et al. Trends in the prevalence of obesity in employed adults in central-western France: a population-based study, 1995-2005. Prev Med 2009; 48: 262–266.
Acknowledgements
We are indebted to the participants for their involvement in the study. We thank Véronique Gourlet for data management and statistical analyses.
Author contributions
KVG supervised statistical analyses, interpreted data and drafted the manuscript. M-FR-C designed the protocol, interpreted the data and critically revised the manuscript. GG, DG and OL collected the data and critically revised the manuscript. SH designed the protocol and critically revised the manuscript. SP designed the protocol, supervised statistical analyses, interpreted data and critically revised the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Giudici, K., Rolland-Cachera, MF., Gusto, G. et al. Body mass index growth trajectories associated with the different parameters of the metabolic syndrome at adulthood. Int J Obes 41, 1518–1525 (2017). https://doi.org/10.1038/ijo.2017.119
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2017.119
This article is cited by
-
Cow’s milk fat and child adiposity: a prospective cohort study
International Journal of Obesity (2021)
-
Resolving early obesity leads to a cardiometabolic profile within normal ranges at 23 years old in a two-decade prospective follow-up study
Scientific Reports (2021)
-
Do body mass index and waist-to-height ratio over the preceding decade predict retinal microvasculature in 11–12 year olds and midlife adults?
International Journal of Obesity (2020)
-
Infant Growth and Long-term Cardiometabolic Health: a Review of Recent Findings
Current Nutrition Reports (2019)