Abstract
Background and objectives:
Obesity is a global epidemic which increases the risk of the metabolic syndrome. Cathelicidin (LL-37 and mCRAMP) is an antimicrobial peptide with an unknown role in obesity. We hypothesize that cathelicidin expression correlates with obesity and modulates fat mass and hepatic steatosis.
Materials and methods:
Male C57BL/6 J mice were fed a high-fat diet. Streptozotocin was injected into mice to induce diabetes. Experimental groups were injected with cathelicidin and CD36 overexpressing lentiviruses. Human mesenteric fat adipocytes, mouse 3T3-L1 differentiated adipocytes and human HepG2 hepatocytes were used in the in vitro experiments. Cathelicidin levels in non-diabetic, prediabetic and type II diabetic patients were measured by enzyme-linked immunosorbent assay.
Results:
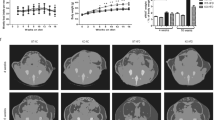
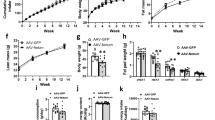
Lentiviral cathelicidin overexpression reduced hepatic steatosis and decreased the fat mass of high-fat diet-treated diabetic mice. Cathelicidin overexpression reduced mesenteric fat and hepatic fatty acid translocase (CD36) expression that was reversed by lentiviral CD36 overexpression. Exposure of adipocytes and hepatocytes to cathelicidin significantly inhibited CD36 expression and reduced lipid accumulation. Serum cathelicidin protein levels were significantly increased in non-diabetic and prediabetic patients with obesity, compared with non-diabetic patients with normal body mass index (BMI) values. Prediabetic patients had lower serum cathelicidin protein levels than non-diabetic subjects.
Conclusions:
Cathelicidin inhibits the CD36 fat receptor and lipid accumulation in adipocytes and hepatocytes, leading to a reduction of fat mass and hepatic steatosis in vivo. Circulating cathelicidin levels are associated with increased BMI. Our results demonstrate that cathelicidin modulates the development of obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ogden CL, Carroll MD, Kit BK, Flegal KM . Prevalence of childhood and adult obesity in the United States, 2011-2012. JAMA 2014; 311: 806–814.
Finkelstein EA, Trogdon JG, Cohen JW, Dietz W . Annual medical spending attributable to obesity: payer-and service-specific estimates. Health Aff 2009; 28: w822–w831.
Lackey DE, Olefsky JM . Regulation of metabolism by the innate immune system. Nat Rev Endocrinol 2016; 12: 15–28.
Chen L, Chen R, Wang H, Liang F . Mechanisms linking inflammation to insulin resistance. Int J Endocrinol 2015; 2015: 508409.
Khodabandehloo H, Gorgani-Firuzjaee S, Panahi G, Meshkani R . Molecular and cellular mechanisms linking inflammation to insulin resistance and beta-cell dysfunction. Transl Res 2016; 167: 228–256.
Ho S, Pothoulakis C, Koon HW . Antimicrobial peptides and colitis. Curr Pharm Des 2013; 19: 40–47.
Rivas-Santiago B, Trujillo V, Montoya A, Gonzalez-Curiel I, Castaneda-Delgado J, Cardenas A et al. Expression of antimicrobial peptides in diabetic foot ulcer. J Dermatol Sci 2012; 65: 19–26.
Gonzalez-Curiel I, Castaneda-Delgado J, Lopez-Lopez N, Araujo Z, Hernandez-Pando R, Gandara-Jasso B et al. Differential expression of antimicrobial peptides in active and latent tuberculosis and its relationship with diabetes mellitus. Hum Immunol 2011; 72: 656–662.
Park HY, Kim JH, Jung M, Chung CH, Hasham R, Park CS et al. A long-standing hyperglycaemic condition impairs skin barrier by accelerating skin ageing process. Exp Dermatol 2011; 20: 969–974.
Carretero M, Escamez MJ, Garcia M, Duarte B, Holguin A, Retamosa L et al. In vitro and in vivo wound healing-promoting activities of human cathelicidin LL-37. J Invest Dermatol 2008; 128: 223–236.
Masarone M, Federico A, Abenavoli L, Loguercio C, Persico M . Non alcoholic fatty liver. Epidemiology and natural history. Rev Recent Clin Trials 2014; 9: 126–133.
Yoon HJ, Cha BS . Pathogenesis and therapeutic approaches for non-alcoholic fatty liver disease. World J Hepatol 2014; 6: 800–811.
Kobyliak N, Abenavoli L . The Role of liver biopsy to assess non-alcoholic fatty liver disease. Rev Recent Clin Trials 2014; 9: 159–169.
Zezos P, Renner EL . Liver transplantation and non-alcoholic fatty liver disease. World J Gastroenterol 2014; 20: 15532–15538.
Karagiannides I, Kokkotou E, Tansky M, Tchkonia T, Giorgadze N, O'Brien M et al. Induction of colitis causes inflammatory responses in fat depots: evidence for substance P pathways in human mesenteric preadipocytes. Proc Natl Acad Sci USA 2006; 103: 5207–5212.
Karagiannides I, Bakirtzi K, Kokkotou E, Stavrakis D, Margolis KG, Thomou T et al. Role of substance P in the regulation of glucose metabolism via insulin signaling-associated pathways. Endocrinology 2011; 152: 4571–4580.
Karagiannides I, Thomou T, Tchkonia T, Pirtskhalava T, Kypreos KE, Cartwright A et al. Increased CUG triplet repeat-binding protein-1 predisposes to impaired adipogenesis with aging. J Biol Chem 2006; 281: 23025–23033.
Hara T, Kimura I, Inoue D, Ichimura A, Hirasawa A . Free fatty acid receptors and their role in regulation of energy metabolism. Rev Physiol Biochem Pharmacol 2013; 164: 77–116.
Larter CZ, Yeh MM, Van Rooyen DM, Teoh NC, Brooling J, Hou JY et al. Roles of adipose restriction and metabolic factors in progression of steatosis to steatohepatitis in obese, diabetic mice. J Gastroenterol Hepatol 2009; 24: 1658–1668.
Turcotte LP, Raney MA, Todd MK . ERK1/2 inhibition prevents contraction-induced increase in plasma membrane FAT/CD36 content and FA uptake in rodent muscle. Acta Physiol Scand 2005; 184: 131–139.
Pop-Busui R, Ang L, Holmes C, Gallagher K, Feldman EL . Inflammation as a therapeutic target for diabetic neuropathies. Curr Diab Rep 2016; 16: 29.
Febbraio M, Hajjar DP, Silverstein RL . CD36: a class B scavenger receptor involved in angiogenesis, atherosclerosis, inflammation, and lipid metabolism. J Clin Invest 2001; 108: 785–791.
Bonen A, Campbell SE, Benton CR, Chabowski A, Coort SL, Han XX et al. Regulation of fatty acid transport by fatty acid translocase/CD36. Proc Nutr Soc 2004; 63: 245–249.
Martin C, Chevrot M, Poirier H, Passilly-Degrace P, Niot I, Besnard P . CD36 as a lipid sensor. Physiol Behav 2011; 105: 36–42.
Christiaens V, Van Hul M, Lijnen HR, Scroyen I . CD36 promotes adipocyte differentiation and adipogenesis. Biochim Biophys Acta 2012; 1820: 949–956.
Pinheiro da Silva F, Gallo RL, Nizet V . Differing effects of exogenous or endogenous cathelicidin on macrophage toll-like receptor signaling. Immunol Cell Biol 2009; 87: 496–500.
Bost F, Aouadi M, Caron L, Even P, Belmonte N, Prot M et al. The extracellular signal-regulated kinase isoform ERK1 is specifically required for in vitro and in vivo adipogenesis. Diabetes 2005; 54: 402–411.
Miquilena-Colina ME, Lima-Cabello E, Sanchez-Campos S, Garcia-Mediavilla MV, Fernandez-Bermejo M, Lozano-Rodriguez T et al. Hepatic fatty acid translocase CD36 upregulation is associated with insulin resistance, hyperinsulinaemia and increased steatosis in non-alcoholic steatohepatitis and chronic hepatitis C. Gut 2011; 60: 1394–1402.
Clugston RD, Yuen JJ, Hu Y, Abumrad NA, Berk PD, Goldberg IJ et al. CD36-deficient mice are resistant to alcohol- and high-carbohydrate-induced hepatic steatosis. J Lipid Res 2014; 55: 239–246.
Jiao P, Feng B, Li Y, He Q, Xu H . Hepatic ERK activity plays a role in energy metabolism. Mol Cell Endocrinol 2013; 375: 157–166.
Andersen H . Motor dysfunction in diabetes. Diabetes Metab Res Rev 2012; 28: 89–92.
Schemmel KE, Padiyara RS, D'Souza JJ . Aldose reductase inhibitors in the treatment of diabetic peripheral neuropathy: a review. J Diabetes Complications 2010; 24: 354–360.
Krause MP, Riddell MC, Hawke TJ . Effects of type 1 diabetes mellitus on skeletal muscle: clinical observations and physiological mechanisms. Pediatr Diabetes 2011; 12: 345–364.
Cotter MA, Cameron NE, Robertson S, Ewing I . Polyol pathway-related skeletal muscle contractile and morphological abnormalities in diabetic rats. Exp Physiol 1993; 78: 139–155.
Cameron NE, Cotter MA, Robertson S . Changes in skeletal muscle contractile properties in streptozocin-induced diabetic rats and role of polyol pathway and hypoinsulinemia. Diabetes 1990; 39: 460–465.
Cameron NE, Cotter MA, Basso M, Hohman TC . Comparison of the effects of inhibitors of aldose reductase and sorbitol dehydrogenase on neurovascular function, nerve conduction and tissue polyol pathway metabolites in streptozotocin-diabetic rats. Diabetologia 1997; 40: 271–281.
Sacerdote P, Franchi S, Trovato AE, Valsecchi AE, Panerai AE, Colleoni M . Transient early expression of TNF-alpha in sciatic nerve and dorsal root ganglia in a mouse model of painful peripheral neuropathy. Neurosci Lett 2008; 436: 210–213.
Zhang LJ, Guerrero-Juarez CF, Hata T, Bapat SP, Ramos R, Plikus MV et al. Innate immunity. Dermal adipocytes protect against invasive Staphylococcus aureus skin infection. Science 2015; 347: 67–71.
Acknowledgements
This work was supported by CCFA (#2691), NIH K01(DK084256), and NIH R03 (DK103964) grants to HWK, CCFA (#287244, #3831 & #324000) to MC, SH and DHNTran, and United States PHS grant DK046763 to DQS. Clinical data and specimens were provided by the MIRIAD Biobank. MIRIAD is currently supported by F Widjaja Foundation Inflammatory Bowel and Immunobiology Research Institute, NIH grant P01DK046763, The European Union Grant 305479, NIDDK Grant DK062413, U54 DE023798 and the Leona M and Harry B Helmsley Charitable Trust.
Author contributions
DHYT, DH-NT and HWK acquired the data and drafted the manuscript. TS, LR, ECL, CO, SH, EF, CS, JEL, AS, MV, TCH, KB, MC, BS, IL and IK acquired the data and revised the manuscript. SAM, RLG, ZL, DQS, PF, DPBMG and SRT provided provision of materials, acquired data and revised the manuscript. HWK approved the final version of manuscript and study supervision.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Hoang-Yen Tran, D., Hoang-Ngoc Tran, D., Mattai, S. et al. Cathelicidin suppresses lipid accumulation and hepatic steatosis by inhibition of the CD36 receptor. Int J Obes 40, 1424–1434 (2016). https://doi.org/10.1038/ijo.2016.90
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2016.90
This article is cited by
-
Elafin inhibits obesity, hyperglycemia, and liver steatosis in high-fat diet-treated male mice
Scientific Reports (2020)
-
Dysregulated liver lipid metabolism and innate immunity associated with hepatic steatosis in neonatal BBdp rats and NOD mice
Scientific Reports (2019)
-
Antimicrobial proteins: intestinal guards to protect against liver disease
Journal of Gastroenterology (2019)
-
Upregulated expression of human cathelicidin LL-37 in hypercholesterolemia and its relationship with serum lipid levels
Molecular and Cellular Biochemistry (2018)