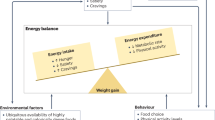
Abstract
Despite the increasing number of medications recently approved to treat obesity among adults, few agents have been formally evaluated in children or adolescents for this indication. Moreover, there is a paucity of guidance in the literature addressing best practices with regard to pediatric obesity pharmacotherapy clinical trial design, and only general recommendations have been offered by regulatory agencies on this topic. The purposes of this article are to (1) offer a background of the current state of the field of pediatric obesity medicine, (2) provide a brief review of the literature summarizing pediatric obesity pharmacotherapy clinical trials, and (3) highlight and discuss some of the unique aspects that should be considered when designing and conducting high-quality clinical trials evaluating the safety and efficacy of obesity medications in children and adolescents. Suggestions are offered in the areas of target population and eligibility criteria, clinical trial end-point selection, trial duration, implementation of lifestyle modification therapy and recruitment and retention of participants. Efforts should be made to design and conduct trials appropriately to ensure that high-quality evidence is generated on the safety and efficacy of various medications used to treat pediatric obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Skinner AC, Skelton JA . Prevalence and trends in obesity and severe obesity among children in the United States, 1999-2012. JAMA Pediatr 2014; 168: 561–566.
Kelly AS, Barlow SE, Rao G, Inge TH, Hayman LL, Steinberger J et al. Severe obesity in children and adolescents: identification, associated health risks, and treatment approaches: a scientific statement from the American Heart Association. Circulation 2013; 128: 1689–1712.
Skinner AC, Perrin EM, Moss LA, Skelton JA . Cardiometabolic risks and severity of obesity in children and young adults. N Engl J Med 2015; 373: 1307–1317.
Danielsson P, Kowalski J, Ekblom O, Marcus C . Response of severely obese children and adolescents to behavioral treatment. Arch Pediatr Adolesc Med 2012; 166: 1103–1108.
Knop C, Singer V, Uysal Y, Schaefer A, Wolters B, Reinehr T . Extremely obese children respond better than extremely obese adolescents to lifestyle interventions. Pediatr Obes 2015; 10: 7–14.
Johnston CA, Tyler C, Palcic JL, Stansberry SA, Gallagher MR, Foreyt JP . Smaller weight changes in standardized body mass index in response to treatment as weight classification increases. J Pediatr 2011; 158: 624–627.
Kalarchian MA, Levine MD, Arslanian SA, Ewing LJ, Houck PR, Cheng Y et al. Family-based treatment of severe pediatric obesity: randomized, controlled trial. Pediatrics 2009; 124: 1060–1068.
Ochner CN, Tsai AG, Kushner RF, Wadden TA . Treating obesity seriously: when recommendations for lifestyle change confront biological adaptations. Lancet Diabetes Endocrinol 2015; 3: 232–234.
Leibel RL, Rosenbaum M, Hirsch J . Changes in energy expenditure resulting from altered body weight. N Engl J Med 1995; 332: 621–628.
MacLean PS, Bergouignan A, Cornier MA, Jackman MR . Biology's response to dieting: the impetus for weight regain. Am J Physiol Regul Integr Comp Physiol 2011; 301: R581–R600.
Sumithran P, Prendergast LA, Delbridge E, Purcell K, Shulkes A, Kriketos A et al. Long-term persistence of hormonal adaptations to weight loss. N Engl J Med 2011; 365: 1597–1604.
Schwimmer JB . Clinical trials for adolescent obesity: cooking up an alphabet stew of what to do. JAMA Pediatr 2013; 167: 391–393.
Sherafat-Kazemzadeh R, Yanovski SZ, Yanovski JA . Pharmacotherapy for childhood obesity: present and future prospects. Int J Obes (Lond) 2013; 37: 1–15.
Freemark M . Pharmacotherapy of childhood obesity: an evidence-based, conceptual approach. Diabetes Care 2007; 30: 395–402.
McDonagh MS, Selph S, Ozpinar A, Foley C . Systematic review of the benefits and risks of metformin in treating obesity in children aged 18 years and younger. JAMA Pediatr 2014; 168: 178–184.
Park MH, Kinra S, Ward KJ, White B, Viner RM . Metformin for obesity in children and adolescents: a systematic review. Diabetes Care 2009; 32: 1743–1745.
Rogovik AL, Chanoine JP, Goldman RD . Pharmacotherapy and weight-loss supplements for treatment of paediatric obesity. Drugs 2010; 70: 335–346.
Mauras N, DelGiorno C, Hossain J, Bird K, Killen K, Merinbaum D et al. Metformin use in children with obesity and normal glucose tolerance—effects on cardiovascular markers and intrahepatic fat. J Pediatr Endocrinol Metab 2012; 25: 33–40.
Kendall D, Vail A, Amin R, Barrett T, Dimitri P, Ivison F et al. Metformin in Obese Children and Adolescents: The MOCA Trial. J Clin Endocrinol Metab 2013; 98: 322–329.
Kelly AS, Rudser KD, Nathan BM, Fox CK, Metzig AM, Coombes BJ et al. The effect of glucagon-like Peptide-1 receptor agonist therapy on body mass index in adolescents with severe obesity: a randomized, placebo-controlled, clinical trial. JAMA Pediatr 2013; 167: 355–360.
Yanovski SZ, Yanovski JA . Long-term drug treatment for obesity: a systematic and clinical review. JAMA 2014; 311: 74–86.
Karres J, Tomasi P, Saint RA . The development of pharmacological treatment of obesity in children. A European regulatory perspective. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 2011; 54: 570–576.
Oude LH, Baur L, Jansen H, Shrewsbury VA, O'Malley C, Stolk RP et al. Interventions for treating obesity in children. Cochrane Database Syst Rev 2009; CD001872.
Whitlock EP, O'Connor EA, Williams SB, Beil TL, Lutz KW . Effectiveness of weight management interventions in children: a Targeted Systematic Review for the USPSTF. Pediatrics 2010; 125: e396–e418.
Wilfley DE, Stein RI, Saelens BE, Mockus DS, Matt GE, Hayden-Wade HA et al. Efficacy of maintenance treatment approaches for childhood overweight: a randomized controlled trial. JAMA 2007; 298: 1661–1673.
Juonala M, Magnussen CG, Berenson GS, Venn A, Burns TL, Sabin MA et al. Childhood adiposity, adult adiposity, and cardiovascular risk factors. N Engl J Med 2011; 365: 1876–1885.
Berkowitz RI, Fujioka K, Daniels SR, Hoppin AG, Owen S, Perry AC et al. Effects of sibutramine treatment in obese adolescents: a randomized trial. Ann Intern Med 2006; 145: 81–90.
Chanoine JP, Hampl S, Jensen C, Boldrin M, Hauptman J . Effect of orlistat on weight and body composition in obese adolescents: a randomized controlled trial. JAMA 2005; 293: 2873–2883.
Colman E . Food and Drug Administration's obesity drug guidance document: a short history. Circulation 2012; 125: 2156–2164.
Woo JG . Using body mass index Z-score among severely obese adolescents: a cautionary note. Int J Pediatr Obes 2009; 4: 405–410.
Flegal KM, Wei R, Ogden CL, Freedman DS, Johnson CL, Curtin LR . Characterizing extreme values of body mass index-for-age by using the 2000 Centers for Disease Control and Prevention growth charts. Am J Clin Nutr 2009; 90: 1314–1320.
Gulati AK, Kaplan DW, Daniels SR . Clinical tracking of severely obese children: a new growth chart. Pediatrics 2012; 130: 1136–1140.
Cole TJ, Bellizzi MC, Flegal KM, Dietz WH . Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ 2000; 320: 1240–1243.
Cole TJ, Faith MS, Pietrobelli A, Heo M . What is the best measure of adiposity change in growing children: BMI, BMI %, BMI z-score or BMI centile? Eur J Clin Nutr 2005; 59: 419–425.
Paluch RA, Epstein LH, Roemmich JN . Comparison of methods to evaluate changes in relative body mass index in pediatric weight control. Am J Hum Biol 2007; 19: 487–494.
Bryant M, Ashton L, Nixon J, Jebb S, Wright J, Roberts K et al. Framework of outcome measures recommended for use in the evaluation of childhood obesity treatment interventions: the CoOR framework. Pediatr Obes 2014; 9: e116–e131.
Little RJ, D'Agostino R, Cohen ML, Dickersin K, Emerson SS, Farrar JT et al. The prevention and treatment of missing data in clinical trials. N Engl J Med 2012; 367: 1355–1360.
Evia-Viscarra ML, Rodea-Montero ER, Apolinar-Jimenez E, Munoz-Noriega N, Garcia-Morales LM, Leanos-Perez C et al. The effects of metformin on inflammatory mediators in obese adolescents with insulin resistance: controlled randomized clinical trial. J Pediatr Endocrinol Metab 2012; 25: 41–49.
Rezvanian H, Hashemipour M, Kelishadi R, Tavakoli N, Poursafa P . A randomized, triple masked, placebo-controlled clinical trial for controlling childhood obesity. World J Pediatr 2010; 6: 317–322.
Burgert TS, Duran EJ, Goldberg-Gell R, Dziura J, Yeckel CW, Katz S et al. Short-term metabolic and cardiovascular effects of metformin in markedly obese adolescents with normal glucose tolerance. Pediatr Diabetes 2008; 9: 567–576.
Freemark M, Bursey D . The effects of metformin on body mass index and glucose tolerance in obese adolescents with fasting hyperinsulinemia and a family history of type 2 diabetes. Pediatrics 2001; 107: E55.
Love-Osborne K, Sheeder J, Zeitler P . Addition of metformin to a lifestyle modification program in adolescents with insulin resistance. J Pediatr 2008; 152: 817–822.
Srinivasan S, Ambler GR, Baur LA, Garnett SP, Tepsa M, Yap F et al. Randomized, controlled trial of metformin for obesity and insulin resistance in children and adolescents: improvement in body composition and fasting insulin. J Clin Endocrinol Metab 2006; 91: 2074–2080.
Wiegand S, l'Allemand D, Hubel H, Krude H, Burmann M, Martus P et al. Metformin and placebo therapy both improve weight management and fasting insulin in obese insulin-resistant adolescents: a prospective, placebo-controlled, randomized study. Eur J Endocrinol 2010; 163: 585–592.
Yanovski JA, Krakoff J, Salaita CG, McDuffie JR, Kozlosky M, Sebring NG et al. Effects of metformin on body weight and body composition in obese insulin-resistant children: a randomized clinical trial. Diabetes 2011; 60: 477–485.
Lavine JE, Schwimmer JB, Van Natta ML, Molleston JP, Murray KF, Rosenthal P et al. Effect of vitamin E or metformin for treatment of nonalcoholic fatty liver disease in children and adolescents: the TONIC randomized controlled trial. JAMA 2011; 305: 1659–1668.
Wilson DM, Abrams SH, Aye T, Lee PD, Lenders C, Lustig RH et al. Metformin extended release treatment of adolescent obesity: a 48-week randomized, double-blind, placebo-controlled trial with 48-week follow-up. Arch Pediatr Adolesc Med 2010; 164: 116–123.
Maahs D, de Serna DG, Kolotkin RL, Ralston S, Sandate J, Qualls C et al. Randomized, double-blind, placebo-controlled trial of orlistat for weight loss in adolescents. Endocr Pract 2006; 12: 18–28.
Kelly AS, Metzig AM, Rudser KD, Fitch AK, Fox CK, Nathan BM et al. Exenatide as a weight-loss therapy in extreme pediatric obesity: a randomized, controlled pilot study. Obesity (Silver Spring) 2012; 20: 364–370.
Acknowledgements
KDR is supported in part by NCATS award UL1TR000114. JRR is supported in part by NHLBI award: F32HL12785. The authors are grateful to Dr Charles Billington for reviewing and commenting on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
ASK serves as a consultant for Takeda Pharmaceuticals and Novo Nordisk Pharmaceuticals and is the signatory author for a pediatric obesity clinical trial sponsored by Novo Nordisk Pharmaceuticals; he does not accept personal or professional income for his services. ASK also receives research support from Astra Zeneca Pharmaceuticals in the form of drug/placebo. CKF is a site principal investigator for a pediatric obesity clinical trial sponsored by Novo Nordisk Pharmaceuticals.The remainig authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kelly, A., Fox, C., Rudser, K. et al. Pediatric obesity pharmacotherapy: current state of the field, review of the literature and clinical trial considerations. Int J Obes 40, 1043–1050 (2016). https://doi.org/10.1038/ijo.2016.69
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2016.69
This article is cited by
-
Long-term weight loss after bariatric procedures for morbidly obese adolescents and youth: a single-institution analysis with up to 19-year follow-up
Surgical Endoscopy (2023)
-
Strategies in the Management of Adolescent Obesity
Current Pediatrics Reports (2020)
-
Severe Obesity in the Pediatric Population: Current Concepts in Clinical Care
Current Obesity Reports (2019)
-
Factors associated with long-term weight-loss maintenance following bariatric surgery in adolescents with severe obesity
International Journal of Obesity (2018)
-
Working toward precision medicine approaches to treat severe obesity in adolescents: report of an NIH workshop
International Journal of Obesity (2018)