Abstract
Aim:
To provide a systematic review, of published data, to compare weight losses following very low calorie (<800 kcal per day VLCD) or low-energy liquid-formula (>800 kcal per day LELD) diets, in people with and without type 2 diabetes mellitus (T2DM).
Methods:
Systematic electronic searches of Medline (1946–2015) and Embase (1947–2015) to identify published studies using formula total diet replacement diets (VLCD/LELD). Random effects meta-analysis using weighted mean difference (WMD) in body weight between groups (with and without diabetes) as the summary estimate.
Results:
Final weight loss, in the five included studies, weighted for study sizes, (n=569, mean BMI=35.5–42.6 kg/m2), was not significantly different between participants with and without T2DM: −1.2 kg; 95% CI: −4.1 to 1.6 kg). Rates of weight loss were also similar in the two groups −0.6 kg per week (T2DM) and 0.5 kg per week (no diabetes), and for VLCD (<800 kcal per day) and LELD (>800 kcal per day).
Conclusions:
Weight losses with liquid-formula diets are very similar for VLCD and LELD and for obese subjects with or without T2DM. They can potentially achieve new weight loss/ maintenance targets of >15–20% for people with severe and medically complicated obesity.
Similar content being viewed by others
Introduction
As populations become more obese, there is increasing concern about the rapidly rising categories of severe obesity, which demand greater weight loss to control secondary medical consequences. Type 2 diabetes mellitus (T2DM) is closely linked to obesity, and is the main contributor to rising healthcare costs of obesity.1, 2 Most individuals with T2DM have a BMI >25 kg/m2 and about half have a BMI >30 kg/m2.2, 3, 4 With a BMI over 35 kg/m2, 20% of all men and 11% of women have known diabetes.5
There is overwhelming evidence that modest sustained weight loss, for example, current target of 5–10%, prevents the onset of most new T2DM in people with pre-diabetes,6 and that it improves all aspects of diabetes control (glycaemia, blood pressure, lipids and microvascular damage7 with reductions in drug doses for medications used to treat hypertension, diabetes and dyslipidaemia.8 Although previous guidelines have retained a 5–10% weight-loss target, the 2010 Scottish Intercollegiate (SIGN) Obesity guideline, recognising changes in obesity prevalence, and also recent evidence for more aggressive interventions, has set a new national weight loss/maintenance target of >15–20% for those with BMI >35 kg/m2 or >30 kg/m2 with serious medical complications such as T2DM.9 In routine UK National Health Service diabetes care, few people achieve a weight loss of >15 kg (or >15%). Bariatric surgery is recommended for obese people with T2DM by both the UK National Institute for Care Excellence (NICE)10 and SIGN,9 and has been shown to reverse the metabolic abnormalities of T2DM rapidly following surgery, at least in people up to 6 years after diagnosis of T2DM.11 Surgery carries immediate and long-term risks, and is not acceptable to all people. The metabolic benefits of bariatric surgery have been reproduced by negative energy balance alone, using a low-energy liquid-formula diets, but these are seldom offered as part of diabetes care.12
With conventional dietary advice, obese people with T2DM usually lose less weight than people without diabetes, partly because they have already lost what they can manage, and partly because many anti-diabetic drugs are obesogenic.13 Here, we provide a systematic review assessing published studies to compare weight losses in people with T2DM and people without diabetes prescribed ‘Total Diet Replacement’ using modern commercial nutrient-replete very-low calorie diets (VLCD,<800 kcal per day) or low-energy liquid-formula diets (LELD, >800 kcal per day).
Methods
This review was conducted following the PRISMA guidelines on systematic reviews.14
Data sources and searches
Systematic electronic searches of Medline (1946–2015), Embase (1947–2015) and CINAHL (1946–2015) were performed to identify published studies using formula Total Diet Replacement diets. Key search terms used were: (weight adj2 (loss or lost or losing or reduce* or change*)), *Obesity/; (type 2 diabetes or non-insulin-dependent diabetic or non-insulin-dependent diabetes or T2DM); (low-energy liquid diet or low-energy diet or very-low calorie diet or liquid-formula diet or calorie restriction or very-low calorie diet). Hand searching of reference lists of retrieved studies was also conducted. The search was limited to English language, human and adults.
Study selection
Articles were selected on the basis of title and abstract. Inclusion was dependent on the following eligibility criteria: non-randomised or randomised controlled trial with a primary purpose of weight management, adult participants (>18 years), weight change reported quantitatively, and a comparison of weight change in patients with and without T2DM made. Studies identified by the database searches and review of potentially relevant studies against inclusion/exclusion criteria were carried out independently by two reviewers (WL and LH).
Quality assessment
The quality and possible sources of bias of included studies was independently assessed by two reviewers (WL and LH) using the Cochrane Risk of Bias Assessment tool for Non-Randomised Studies of Interventions (ACROBAT-NRSI).15
Data analysis
A random effects meta-analysis was conducted using Comprehensive Meta-analysis (CMA; Version 3.0 for Windows). The weighted mean difference (WMD) in body weight between participants with T2DM and non-diabetic subjects was used as the summary estimate. Cochrane’s Q statistic was used to assess heterogeneity, with the I2 statistic used to measure the degree of heterogeneity.
Because studies varied in duration, which necessarily affects total weight loss, a comparison was made between the rates of weight loss. Mean weekly rate of weight loss was calculated for each study by dividing mean weight loss by number of study weeks. Mean rate of weight loss for all studies was calculated by multiplying mean weekly weight loss for each study by number of study participants, and then the sum of these figures was divided by the total number of participants in the group. Where weight change was reported with s.e.m., this was converted to s.d. (s.e.m. × √n) to provide consistency in the reported variability.
Results
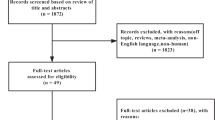
The systematic search identified 151 potential studies. After duplicates were removed 121 titles and abstracts were screened. Obviously irrelevant studies were excluded for the reasons illustrated in Figure 1. Full texts of 17 studies were retrieved and reviewed for eligibility against the inclusion/exclusion. Five studies fulfilled our criteria and were included in the review.
Study characteristics
Setting
The studies reported results from USA16, 17 Australia,18 UK19 and Sweden.20 The settings for studies varied. In one study the intervention was delivered on an inpatient basis,16 one was set in primary care19 an endocrine clinic,18 obesity clinic17 and surgical unit.20
Participant demographics
All studies recruited both males and females classified as obese (BMI >30 kg/m2). One study (16) included some people with BMI >25 kg/m2. Mean age ranged from 43 to 55 years.
Exclusion criteria
Exclusion criteria were not described in two studies.19, 20 Use of anti-obesity medications was an exclusion criterion in one study,18 and insulin use in another.16
Medications
Medications used in the management of T2DM were detailed in three studies18, 19, 20 and included metformin, sulphonylurea and insulin. The prescribed oral hypoglycaemic agents, was not specified in one study.16 Medications in addition to those for diabetes management were detailed in two studies.18, 19 One study did not detail medications at all.17
Energy content and duration of formula diets and total diet replacement
Daily energy provision from the formula diets varied among the included studies and ranged from 300 to 1000 kcal per day (Table 1). The durations of VLCD/LELD reported in studies ranged from 4 to 52 weeks. In one study17 participants transitioned to include a meal of ~300 kcal at different time points.
Quality of included studies
All five included studies were non-randomised (as it is impossible to randomise into T2DM or non-diabetes categories); two case–control studies,16, 17 one in which participants were allocated to the treatment of their preference,19 and two cohort studies; one retrospective17 and one prospective.20 As the studies were dietary interventions, none included blinding of participants.
Confounding was considered the main possible source of potential moderate or serious bias in all included studies (Table 2). Age, gender, baseline weight, and co-morbid conditions are potential confounders in the achievement of weight loss. Only one study included adjustments for these factors.17 However in this study the transition from total meal replacement to partial meal replacement varied among participants, with a potential influence on weight loss, and was not adjusted for in analysis. Participant selection was considered unlikely to have introduced significant bias, as in all studies those with T2DM were compared with those without diabetes from the same or similar population. Bias in the measurement of interventions was low, as in all studies the intervention was well defined. Outcomes were ascertained using objective measurements in all studies and therefore the risk of bias was considered low.
Attrition was reported in two of the five studies,18 and weight change data were reported for completers only (excluding dropouts). One study was carried out on an inpatient basis16 and all subjects were included in weight change analyses. In one study,19 the study population was small, no dropouts were reported, and we have assumed all subjects were followed up and included in the analysis. In the remaining study,20 all subjects completed the study and were included in the weight change analyses. Risk of bias from missing data was therefore considered low.
Weight losses
Baseline weights ranged from 99–126 kg (BMI: 30–42.6 kg/m2). Total weight loss ranged from 8.9 to 15 kg in those with T2DM and 7.9–21 kg in those without diabetes, over treatment durations of 4–-52 weeks.
The greatest rates of weight loss were mostly in studies with more restrictive VLCDs.16, 19 Study duration did not appear to influence overall weight loss. Mean weight loss in the study of longest duration17 was similar, or greater, than that observed in studies of shorter duration (Table 1).
Meta-analysis
Data allowing meta-analysis (mean weight change (s.d. or s.e.m.) were available for four studies (Figure 2). Analysis showed almost identical weight changes (post intervention) in those with and without diabetes (WMD: −1.2 kg; 95% CI: −4.1 to 1.6 kg). Substantial heterogeneity was observed (I2= 79%).
Safety
Transient events including constipation, dizziness, postural hypotension, dry skin and bad breath were reported in two studies.16, 18 Serious adverse events were reported in one study. Severe hypoglycaemia was experienced by one diabetic patient using VLCD, and one patient in each group suffered a non-fatal myocardial infarction.19 The remaining two studies reported that the intervention/study was completed without any adverse events.17, 20
Discussion
Substantial mean weight losses were achieved in all studies following VLCD/LELD, varying from 8 to 21 kg in total over 4–52 weeks. That total depended on a variety of factors, including duration of diet, and clinical characteristics of groups, such as age, gender and status of health. In all the studies, the mean rate of weight loss fell below that that predicted for 100% compliance with the estimated energy deficit.
Weight losses were similar for participants with T2DM and participants without diabetes, with the exception of one study,16 where baseline weight was significantly greater in participants without diabetes (126.1±8.5 vs 99.7±3.7 kg, P<0.05), which is likely to have contributed to greater weight loss in the non-diabetic group.
The data generated on weight loss per week, as studies varied widely in duration, also showed similar rates of weight loss in those with T2DM and those without. As with overall weight loss this was with the exception of the study by Henry et al.16 in which a greater rate of weight loss was observed in those without diabetes, again reflecting the higher baseline weight.
The mean weight loss in people with T2DM reported here, in studies which compared people with T2DM and those without T2DM, is similar to a previous meta-analysis of VLED in T2DM, which reported substantial weight loss of 9.6% by people with T2DM over a 6 week period, at a rate of around 1.6 kg per week.13 Studies using similar diets among only non-diabetic participants report similar mean weight losses.21, 22, 23
Similar weight losses with VLED and LELD have been reported previously in the study by Christensen et al.21 which compared VLED and LELD, with weight losses of around 11 kg was observed in both study groups over 8 weeks. Greater energy restriction also conferred no additional benefit in the study by Lin et al.22 with a mean weight loss of 8 kg observed in both groups (450 vs 800 kcal per day) after 12 weeks. Wikstrand et al.23 reported mean weight losses of 20 and 16 kg in free living non-diabetic, male and female participants respectively.
Amongst the studies included in the present review, rates of weight loss were consistent, at 1.2–3.2 kg per week except for much lower rates in studies by Li et al.17 0.3 kg per week and Baker et al.18 0.7 kg per week. The study by Li et al. was over a much longer period of 52 weeks, and the duration of the exclusive formula diet phase was variable amongst participants over the 1 year period, as they could opt to include a meal of ~300 kcal at different time points, which may have contributed to the lower rate of weight loss. Lower weight loss in comparison to other studies was acknowledged by Baker and colleagues. Factors suggested by the authors to explain this were a ‘calendar effect’ and also that the study population were well established patients at a tertiary referral centre and may have represented a more ‘difficult to treat group’ than that of other studies.
The metabolic benefits of bariatric surgery appear to be reproducible by VLCD/LELD. The Newcastle Counterpoint study12 achieved reversal of T2DM with mean weight loss of 15.3 kg in 11 people with T2DM within 4 years of diagnosis, using a 600 kcal per day low-energy liquid diet. The normalisation of fasting plasma glucose persisted for up to 3 months after return to normal diet. Normalisation of HbA1c, or fasting blood glucose, following VLCD was reported in one study included in the present review, Paisey et al.19 with mean absolute weight loss similar to that achieved by Lim et al.12 However normalisation of metabolic status relates both to ongoing energy restriction and on overall weight loss. Acute energy restriction has been shown to improve plasma glucose values and insulin sensitivity even before significant weight loss occurs.12, 13
Formula diets, with a period of total diet replacement, are widely used and popular outside healthcare settings. There is some published evidence that they are successful, with little or no evidence for serious safety problems,24 although large-scale safety testing has not been undertaken. The serious adverse events reported in one study included in the present review were not considered directly related to the diet. Despite clear evidence for efficacy and lack of safety concerns, there is a continuing reluctance by medical staff and in clinical guidelines to support the use of formula diets. Guidelines published in the USA and Australia25, 26 advocate the use of very low calorie/energy diets for weight management, but recommend these should be delivered in medical settings, with close medical monitoring.
At present formula diets are not funded directly within routine UK NHS care, whereas bariatric surgery is. In the 1970 s there was concern about safety of ill-designed modified fasting diets.27 Beliefs that more intensive interventions and rapid weight loss lead to greater weight regain, are not supported by evidence from controlled trials. Comparison of weight regains, following either a 12 week rapid weight loss programme or a 36 week gradual programme, found no difference in the proportion of weight regained at 144 week follow-up.28 Effective strategies for long-term weight-loss maintenance have been described.29, 30 A study conducted within routine NHS primary care29 found that an 810 kcal per day LELD was well accepted by 91 severely obese participants (mean BMI 48 kg/m2), and by GP’s and nurses. Those who attended had a mean weight loss of around 17 kg in 12 weeks. Critically around 33% of all 91 entrants maintained >15 kg weight loss at 12 months. The study was in people without diabetes, but the present review would suggest that results in those with T2DM would not differ greatly. One year follow-up of patients with knee osteoarthritis also found that a structured weight maintenance programme resulted in good maintenance of weight loss.30
It is estimated that by 2025, without new interventions for prevention of weight gain and obesity, five million people in the UK will have diabetes, mostly T2DM (DiabetesUK.org). Despite the recommendations in current clinical guidelines, there is no realistic prospect of bariatric surgery being offered to most obese people because of surgical and follow-up resource limitations. In addition, many people will not agree to surgery. New weight management approaches are required for obese people with T2DM that can be implemented in routine care, where most of these people are managed. The present results suggest that the weight loss phase of weight management can be achieved by a period of ‘Total Diet Replacement’ (TDR), using micronutrient-complete formula diets. This approach towards the SIGN weight loss target of >15 kg (enough to reverse most recent onset T2DM), in many cases is more cost-effectiveness than bariatric surgery.29
Most published studies do not adequately describe whether adverse events were sought systematically, or simply reported on an ad-hoc basis. Future research should include systematic data collection. At present there is no evidence for serious adverse events, and no specific medical monitoring is necessary except for withdrawal or dose reduction of hypoglycaemic, diuretic or anti-hypertensive drugs.
Limitations
This systematic review was limited by the very small number of published studies, using a range of TDR methods, which directly compared weight losses in people with and without T2DM. There was substantial heterogeneity in the studies included, although due to the limited number of studies subgroup analysis could not be conducted to explore heterogeneity. Differences in diets (that is, different calorie provisions across VLCD and LELD) and in particular the very different study durations probably explain much of the statistical heterogeneity. We therefore expressed the results as kg lost per week, to reduce the effect of study duration. There was no clear pattern to suggest different rates of weight loss between VLED and LELD, despite different prescribed energy deficits, indicating differences in compliance. This supports the conclusion of Christensen et al.21 that weight losses are similar on VLED or LELD. Heterogeneity may also have arisen from differences in prescribed medications and other aspects of diabetes care between the studies included. Despite the heterogeneity, the results from the different studies remain rather similar both within and between the patient groups, which provides some confidence that there is no important difference in the weight loss responses of T2DM and non-diabetic people when treated with a formula ‘Total Diet Replacement’. Future studies should include more complete data on clinical details (for example, medications) and complete data on the outcomes for all entered subjects. The extent of data among people with T2DM does not permit assessment of any diabetes-specific safety issues.
Conclusion
These data demonstrate that weight losses with VLCD or LELD are very similar, and that weight losses, following interventions of 4–52 weeks duration, are no different for obese people with or without T2DM. This result is novel, as most published studies of diet and lifestyle interventions have shown less weight loss among subjects with diabetes. It provides a firm baseline on which the use of LELD may be further developed to achieve new weight-loss targets of >15–20% for those with severe and medically complicated obesity. Further research is warranted to evaluate the delivery of more intensive, and better sustained, non-surgical weight-management approaches for routine NHS application, which could offer great benefit to people with T2DM.
Change history
14 March 2017
This article was originally published under a standard license, but has now been made available under a CC BY-NC-ND 4.0 license. The pdf and html versions of the paper have been modified accordingly.
References
Walker A . The Cost of Doing Nothing: The Economics of Obesity in Scotland. Robertson Centre for Biostatistics, University of Glasgow: Glasgow, 2003.
Chan JM, Rimm EB, Colditz GA, Stampfer MJ, Willett WC . Obesity, fat distribution and weight gain as risk factors for clinical diabetes in men. Diabetes Care 1994; 17: 961–969.
UKPDS UK . Prospective Diabetes Study (UKPDS) VIII. Study, design, progress and performance. Diabetology 1991; 34: 877–890.
Lean ME, Powrie JK, Anderson AS, Garthwaite PH . Obesity, weight loss and prognosis in type 2 diabetes. Diabet Med 1990; 7: 228–233.
Scottish Health Survey. Available from: http://www.scotland.gov.uk/Publications/2009/09/28102003/0, 2008.
Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walkder EA et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002; 346: 393–403.
Rowe R, Cowx M, Poole C, McEwan P, Morgan C, Walker M . The effects of orlistat in patients with diabetes: improvement in glycaemic control and weight loss. Curr Med Res Opin 2005; 21: 1885–1890.
Redmon JB, Bertoni AG, Connelly S, Feeney PA, Glasser SP, Glick H et al. Effect of the look AHEAD study intervention on medication use and related cost to treat cardiovascular disease risk factors in individuals with type 2 diabetes. Diabetes Care 2010; 33: 1153–1158.
SIGN. Management of obesity no. 115. Available from: http://www.sign.ac.uk/pdf/sign115.pdf.
NICE. Obesity: identification, assessment and management. Available from: http://www.nice.org.uk/guidance/cg189, 2014.
Guidone C, Manco M, Valera-Mora E, Iaconelli A, Gniuli D, Mari A et al. Mechanisms of recovery from type 2 diabetes after malabsorptive bariatric surgery. Diabetes 2006; 55: 2025–2031.
Lim EL, Hollingsworth KG, Aribisala BS, Chen MJ, Mathers JC, Taylor R . Reversal of type 2 diabetes: normalisation of beta cell function in association with decreased pancreas and liver triacylglycerol. Diabetology 2011; 54: 2506–2514.
Anderson JW, Cyril WC, Kendall CWC, David JA, Jenkins DJA . Importance of weight management in type 2 diabetes. J Am Coll Nutr 2003; 22: 331–339.
Moher D, Liberati A, Tetzlaff J, Altman DG . Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Brit Med J 2009; 339: 332–336.
Cochrane A . Risk of bias assessment tool for non-randomised studies of interventions (ACROBAT-NRS). Availbale from: https://sites.google.com/site/riskofbiastool/.
Henry RR, Wiest-Kent TA, Scheaffer L, Kolterman OG, Olefsky JM . Metabolic Consequences of very-low-calorie diet therapy in obese non-insulin-dependent diabetic and nondiabetic subjects. Diabetes 1986; 35: 155–164.
Li Z, Tseng C-h, Deng ML, Wang M, Heber D . Clinical efficacy of a medically supervised outpatient high-protein, low-calorie diet program is equivalent in prediabetic, diabetic and normoglycaemic obese patients. Nutr Diabetes 2014; 4: e105.
Baker ST, Jerumsa G, Prendergast LA, Panagiotopoulos S, Strauss BJ, Proietto J . Less fat reduction per unit weight loss in type 2 diabetic compared with nondiabetic obese individuals completing a very-low-calorie diet program. Metab Clin Exp 2012; 61: 873–882.
Paisey RB, Harvey P, Rice S, Belka I, Bower L, Dunn M et al. An intensive weight loss programme in established type 2 diabetes and controls: effects on weight and atherosclerosis risk factors at 1 year. Diabet Med 1998; 15: 73–79.
Noren E, Forsell H . Very low calorie diet without aspartame in obese subjects: improved metabolic control after 4 weeks treatment. Nutr J 2014; 13: 77.
Christensen P, Bliddal H, Riecke BF, Leeds AR, Astrup A, Christensen R . Comparison of a low-energy diet and a very low-energy diet in sedentary obese individuals:a pragmatic randomized controlled trial. Clin Obes 2011; 1: 31–40.
Lin WY, Wu CH, Chu NF, Chang CJ . Efficacy and safety of very-low-calorie diet in Taiwanese: a multicenter randomized, controlled trial. Nutrition 2009; 25: 1129–1136.
Wikstrand I, Torgerson J, Bengtsson Boström K . Very low calorie diet (VLCD) followed by a randomized trial of corset treatment for obesity in primary care. Scand J Primary Health Care 2010; 28: 89–94.
Rolland C, Hession M, Murray S, Wise A, Broom I . Randomized clinical trial of standard dietary treatment versus a low-carbohydrate/high-protein diet or the LighterLife Programme in the management of obesity. J Diabetes 2009; 1: 207–217.
The Royal Australian College of General Practitioners and Diabetes Australia. General practice management of type 2 diabetes: 2014–2015. Available from: https://static.diabetesaustralia.com.au/s/fileassets/diabetes-australia/5ed214a6-4cff-490f-a283-bc8279fe3b2f.pdf.
American Diabetes Association. Standards of medical care in diabetes-2016. J Clin Appl Res Educ 2016; 39. Available from: http://care.diabetesjournals.org/content/suppl/2015/12/21/39.Supplement_1.DC2/2016-Standards-of-Care.pdf.
Isner JM, Sours HE, Paris AL, Ferrans VJ, Roberts WC . Sudden, unexpected death in avid dieters using the liquid-protein-modified-fast diet. Observations in 17 patients and the role of the prolonged QT interval. Circulation 1979; 60: 1401–1412.
Purcell K, Sumithrab P, Prendergast LA, Bouniu CJ, Delbridge E, Proietta J . The effect of rate of weight loss on long-term weight management: randomised controlled trial. Lancet Diabetes Endocrinol 2014; 12: 954–962.
Lean M, Brosnahan N, McLoone P, McCombie L, Bell Higgs A, Ross H et al. Feasibility and indicative results from a 12-month low-energy liquid diet treatment and maintenance programme for sever obesity. Br J Gen Pract 63: e115–e124 2013.
Christensen R, Henriksen M, Leeds AR, Gudbergson H, Christensen P, Sorensen TJ et al. The effect of weight maintenance on symptoms of knee osteoarthritis in obese patients: 12 month randomized controlled trial. Arthritis Care Res 2014; 67: 640–650.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
WSL, MEJL and RT are currently engaged on the Diabetes Remission Clinical Trial (DiRECT) funded by Diabetes UK with additional support in kind from Cambridge Weight Plan. MEJL and RT have received support related to the use of formula diet products from Cambridge Weight Plan and from Nestle.
Additional information
Author contributions
MEJL and RT conceived of the idea. WSL and LH collected and analysed the data. WSL drafted the paper. All authors reviewed and revised the paper, and approved the final version.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Leslie, W., Taylor, R., Harris, L. et al. Weight losses with low-energy formula diets in obese patients with and without type 2 diabetes: systematic review and meta-analysis. Int J Obes 41, 96–101 (2017). https://doi.org/10.1038/ijo.2016.175
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2016.175
This article is cited by
-
Empfehlungen zur Ernährung von Personen mit Typ-2-Diabetes mellitus
Die Diabetologie (2024)
-
Evaluate the Effects of Different Types of Preoperative Restricted Calorie Diets on Weight, Body Mass Index, Operation Time and Hospital Stay in Patients Undergoing Bariatric Surgery: a Systematic Review and Meta Analysis Study
Obesity Surgery (2024)
-
The effects of a low carbohydrate diet combined with partial meal replacement on obese individuals
Nutrition & Metabolism (2023)
-
Impact of energy density on energy intake in children and adults: a systematic review and meta-analysis of randomized controlled trials
European Journal of Nutrition (2023)
-
Empfehlungen zur Ernährung von Personen mit Typ-2-Diabetes mellitus
Die Diabetologie (2023)