Abstract
Background:
Cross-sectional studies show that white adipose tissue hypertrophy (few, large adipocytes), in contrast to hyperplasia (many, small adipocytes), associates with insulin resistance and increased risk of developing type 2 diabetes. We investigated if baseline adipose cellularity could predict improvements in insulin sensitivity following weight loss.
Methods:
Plasma samples and subcutaneous abdominal adipose biopsies were examined in 100 overweight or obese individuals before and 10 weeks after a hypocaloric diet (7±3% weight loss) and in 61 obese subjects before and 2 years after gastric by-pass surgery (33±9% weight loss). The degree of adipose tissue hypertrophy or hyperplasia (termed the morphology value) in each individual was calculated on the basis of the relationship between fat cell volume and total fat mass. Insulin sensitivity was determined by homeostasis model assessment-estimated insulin resistance (HOMAIR).
Results:
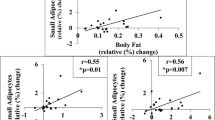
In both cohorts at baseline, subjects with hypertrophy displayed significantly higher fasting plasma insulin and HOMAIR values than subjects with hyperplasia (P<0.0001), despite similar total fat mass. Plasma insulin and HOMAIR were normalized in both cohorts following weight loss. The improvement (delta insulin or delta HOMAIR) was more pronounced in individuals with hypertrophy, irrespective of whether adipose morphology was used as a continuous (P=0.0002–0.027) or nominal variable (P=0.002–0.047). Absolute adipocyte size associated (although weaker than morphology) with HOMAIR improvement only in the surgery cohort. Anthropometric measures at baseline (fat mass, body mass index, waist-to-hip ratio or waist circumference) showed no significant association with delta insulin or delta HOMAIR.
Conclusions:
In contrast to anthropometric variables or fat cell size, subcutaneous adipose morphology predicts improvement in insulin sensitivity following both moderate and pronounced weight loss in overweight/obese subjects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hirsch J, Batchelor B . Adipose tissue cellularity in human obesity. Clin Endocrinol Metab 1976; 5: 299–311.
Salans LB, Knittle JL, Hirsch J . The role of adipose cell size and adipose tissue insulin sensitivity in the carbohydrate intolerance of human obesity. J Clin Invest 1968; 47: 153–165.
Stern JS, Batchelor BR, Hollander N, Cohn CK, Hirsch J . Adipose-cell size and immunoreactive insulin levels in obese and normal-weight adults. Lancet 1972; 2: 948–951.
Krotkiewski M, Sjostrom L, Bjorntorp P, Smith U . Regional adipose tissue cellularity in relation to metabolism in young and middle-aged women. Metabolism 1975; 24: 703–710.
Lundgren M, Svensson M, Lindmark S, Renstrom F, Ruge T, Eriksson JW . Fat cell enlargement is an independent marker of insulin resistance and 'hyperleptinaemia'. Diabetologia 2007; 50: 625–633.
Hoffstedt J, Arner E, Wahrenberg H, Andersson DP, Qvisth V, Lofgren P et al. Regional impact of adipose tissue morphology on the metabolic profile in morbid obesity. Diabetologia 2010; 53: 2496–2503.
Veilleux A, Caron-Jobin M, Noel S, Laberge PY, Tchernof A . Visceral adipocyte hypertrophy is associated with dyslipidemia independent of body composition and fat distribution in women. Diabetes 2011; 60: 1504–1511.
Weyer C, Foley JE, Bogardus C, Tataranni PA, Pratley RE . Enlarged subcutaneous abdominal adipocyte size, but not obesity itself, predicts type II diabetes independent of insulin resistance. Diabetologia 2000; 43: 1498–1506.
Lonn M, Mehlig K, Bengtsson C, Lissner L . Adipocyte size predicts incidence of type 2 diabetes in women. Faseb J 2010; 24: 326–331.
Andersson DP, Eriksson Hogling D, Thorell A, Toft E, Qvisth V, Naslund E et al. Changes in subcutaneous fat cell volume and insulin sensitivity after weight loss. Diabetes Care 2014; 37: 1831–1836.
Arner E, Westermark PO, Spalding KL, Britton T, Ryden M, Frisen J et al. Adipocyte turnover: relevance to human adipose tissue morphology. Diabetes 2010; 59: 105–109.
Petersen M, Taylor MA, Saris WH, Verdich C, Toubro S, Macdonald I et al. Randomized, multi-center trial of two hypo-energetic diets in obese subjects: high- versus low-fat content. Int J Obes (Lond) 2006; 30: 552–560.
Wahrenberg H, Hertel K, Leijonhufvud BM, Persson LG, Toft E, Arner P . Use of waist circumference to predict insulin resistance: retrospective study. BMJ 2005; 330: 1363–1364.
Wallace TM, Levy JC, Matthews DR . Use and abuse of HOMA modeling. Diabetes Care 2004; 27: 1487–1495.
Katsuki A, Sumida Y, Gabazza EC, Murashima S, Furuta M, Araki-Sasaki R et al. Homeostasis model assessment is a reliable indicator of insulin resistance during follow-up of patients with type 2 diabetes. Diabetes Care 2001; 24: 362–365.
Arner P, Andersson DP, Thorne A, Wiren M, Hoffstedt J, Naslund E et al. Variations in the size of the major omentum are primarily determined by fat cell number. J Clin Endocrinol Metab 2013; 98: E897–E901.
Hirsch J, Gallian E . Methods for the determination of adipose cell size in man and animals. J Lipid Res 1968; 9: 110–119.
Smith U, Sjostrom L, Bjornstorp P . Comparison of two methods for determining human adipose cell size. J Lipid Res 1972; 13: 822–824.
Tchoukalova YD, Harteneck DA, Karwoski RA, Tarara J, Jensen MD . A quick, reliable, and automated method for fat cell sizing. J Lipid Res 2003; 44: 1795–1801.
Goldrick RB . Morphological changes in the adipocyte during fat deposition and mobilization. Am J Physiol 1967; 212: 777–782.
Neff KJ, Olbers T, le Roux CW . Bariatric surgery: the challenges with candidate selection, individualizing treatment and clinical outcomes. BMC Med 2013; 11: 8.
Hayes MT, Hunt LA, Foo J, Tychinskaya Y, Stubbs RS . A model for predicting the resolution of type 2 diabetes in severely obese subjects following Roux-en Y gastric bypass surgery. Obes Surg 2011; 21: 910–916.
Aarts EO, Janssen J, Janssen IM, Berends FJ, Telting D, de Boer H . Preoperative fasting plasma C-peptide level may help to predict diabetes outcome after gastric bypass surgery. Obes Surg 2013; 23: 867–873.
Dixon JB, Chuang LM, Chong K, Chen SC, Lambert GW, Straznicky NE et al. Predicting the glycemic response to gastric bypass surgery in patients with type 2 diabetes. Diabetes Care 2013; 36: 20–26.
Krotkiewski M, Sjostrom L, Bjorntorp P, Carlgren G, Garellick G, Smith U . Adipose tissue cellularity in relation to prognosis for weight reduction. Int J Obes 1977; 1: 395–416.
Cotillard A, Poitou C, Torcivia A, Bouillot JL, Dietrich A, Kloting N et al. Adipocyte size threshold matters: link with risk of type 2 diabetes and improved insulin-resistance after gastric bypass. J Clin Endocrinol Metab 2014; 99: E1466–E1470.
Mutch DM, Temanni MR, Henegar C, Combes F, Pelloux V, Holst C et al. Adipose gene expression prior to weight loss can differentiate and weakly predict dietary responders. PLoS One 2007; 2: e1344.
Handjieva-Darlenska T, Holst C, Grau K, Blaak E, Martinez JA, Oppert JM et al. Clinical correlates of weight loss and attrition during a 10-week dietary intervention study: results from the NUGENOB project. Obes Facts 2012; 5: 928–936.
Santos JL, De la Cruz R, Holst C, Grau K, Naranjo C, Maiz A et al. Allelic variants of melanocortin 3 receptor gene (MC3R) and weight loss in obesity: a randomised trial of hypo-energetic high- versus low-fat diets. PLoS One 2011; 6: e19934.
Polak J, Kovacova Z, Holst C, Verdich C, Astrup A, Blaak E et al. Total adiponectin and adiponectin multimeric complexes in relation to weight loss-induced improvements in insulin sensitivity in obese women: the NUGENOB study. Eur J Endocrinol 2008; 158: 533–541.
Andersson DP, Wahrenberg H, Toft E, Qvisth V, Lofgren P, Hertel K et al. Waist circumference to assess reversal of insulin resistance following weight reduction after bariatric surgery: cohort and cross-sectional studies. Int J Obes (Lond) 2014; 38: 438–443.
Ahima RS, Lazar MA . Physiology. The health risk of obesity—better metrics imperative. Science 2013; 341: 856–858.
Sharma AM, Kushner RF . A proposed clinical staging system for obesity. Int J Obes (Lond) 2009; 33: 289–295.
Arner E, Arner P . Health and obesity: not just skin deep. Science 2013; 342: 558–559.
Gallagher D, Visser M, Sepulveda D, Pierson RN, Harris T, Heymsfield SB . How useful is body mass index for comparison of body fatness across age, sex, and ethnic groups? Am J Epidemiol 1996; 143: 228–239.
Sun K, Kusminski CM, Scherer PE . Adipose tissue remodeling and obesity. J Clin Invest 2011; 121: 2094–2101.
Gao H, Mejhert N, Fretz JA, Arner E, Lorente-Cebrian S, Ehrlund A et al. Early B Cell Factor 1 regulates adipocyte morphology and lipolysis in white adipose tissue. Cell Metab 2014; 19: 981–992.
Virtue S, Vidal-Puig A . Adipose tissue expandability, lipotoxicity and the Metabolic Syndrome—an allostatic perspective. Biochim Biophys Acta 2010; 1801: 338–349.
Acknowledgements
We are grateful for the excellent technical assistance of research nurses Britt-Marie Leijonhufvud, Katarina Hertel and Yvonne Widlund as well as laboratory technicians Eva Sjölin, Gaby Åström, Elisabeth Dungner and Kerstin Wåhlén. MR and PA conceived the study and wrote the first version of the manuscript, which was then read and approved by all co-authors. All authors contributed to subject recruitment and analyses. Mikael Rydén and Peter Arner are the guarantors of this work. This study was supported by grants from Swedish Research Council, Swedish Diabetes Association, Novo Nordisk Foundation, EASD/Eli-Lilly, The Erling-Persson Family Foundation, the EU/EFPIA Innovative Medicines Initiative Joint Undertaking (EMIF grant n° 115372) and the Diabetes Program at Karolinska Institutet.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Eriksson-Hogling, D., Andersson, D., Bäckdahl, J. et al. Adipose tissue morphology predicts improved insulin sensitivity following moderate or pronounced weight loss. Int J Obes 39, 893–898 (2015). https://doi.org/10.1038/ijo.2015.18
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2015.18
This article is cited by
-
Associations between subcutaneous adipocyte hypertrophy and nonalcoholic fatty liver disease
Scientific Reports (2022)
-
Using metabolic markers to identify insulin resistance in premenopausal women with and without polycystic ovary syndrome
Journal of Endocrinological Investigation (2021)
-
Elucidating nanoscale mechanical properties of diabetic human adipose tissue using atomic force microscopy
Scientific Reports (2020)
-
Consumption of out-of-season orange modulates fat accumulation, morphology and gene expression in the adipose tissue of Fischer 344 rats
European Journal of Nutrition (2020)
-
Adipose Morphology: a Critical Factor in Regulation of Human Metabolic Diseases and Adipose Tissue Dysfunction
Obesity Surgery (2020)