Abstract
The blood pressure goals set for the treatment of hypertensive patients have been lowered in recent guidelines. To reduce blood pressure levels sufficiently, combination therapies are often needed, but there is little evidence about which combination should be chosen. The present study was carried out to compare the effects of combination therapies, including the angiotensin receptor blocker olmesartan and either a calcium channel blocker (CCB) or a thiazide diuretic, in elderly patients with hypertension. A total of 65 patients aged 65–85 years, with blood pressures of 140/90 mm Hg or higher for those taking antihypertensive medication or 160/100 mm Hg or higher for those not on medication, were randomly assigned to either the group treated with olmesartan plus a dihydropyridine CCB or the group treated with olmesartan plus a thiazide diuretic; 58 patients completed the treatment for 6 months. Systolic and diastolic blood pressures (SBP and DBP) were reduced during the treatment period in both the groups. The reductions in SBP at 1 and 6 months were significantly (P<0.05) greater in the CCB combination group than in the diuretic group (−29 vs. −18 mm Hg, respectively, at 1 month; −32 vs. −23 mm Hg, respectively, at 6 months). Despite greater reduction in SBP in the CCB group, the serum creatinine level and the estimated glomerular filtration rate (eGFR) remained unchanged, whereas in the diuretic group, creatinine was elevated (+0.06 mg per 100 ml, P<0.05) and eGFR was reduced (−4.5 ml min−1 per 1.73 m2). In addition, high-density lipoprotein cholesterol levels were reduced in the diuretic group (−5.0 mg per 100 ml, P<0.01). These results suggest that olmesartan plus a CCB is the preferable combination therapy in comparison with olmesartan plus a thiazide diuretic for elderly patients with hypertension.
Similar content being viewed by others
Introduction
The healthcare costs related to cardiovascular diseases are becoming a substantial economic burden in developed countries, including Japan, where the populations are aging. It has been shown by a number of large-scale clinical trials that blood pressure reductions resulting from the use of antihypertensive treatment lowers the incidence of cardiovascular diseases.1, 2 According to recent clinical studies, the lower the blood pressure is, the better is the prognosis of hypertensive patients, with diabetes or impaired renal function.3, 4 In elderly patients with hypertension, the target blood pressure level has been lowered on the basis of clinical trials showing the benefits of antihypertensive treatment in this population.1, 5, 6 To achieve these blood pressure goals, combination therapy involving different classes of drug is often necessary. For example, according to Bakris et al.,3 an average of 3.2 antihypertensive medications are administered to sufficiently control blood pressure in patients with diabetes or impaired renal function. There is a significant amount of evidence that treating hypertensive patients with renin–angiotensin system (RAS) inhibitors, calcium channel blockers (CCB) or thiazide diuretics improves their prognoses.1, 2 In recently issued guidelines, antihypertensive treatment with an RAS inhibitor and either a CCB or a diuretic is recommended as a combination therapy.1, 2 Indeed, these drug combinations have been used in clinical studies that have demonstrated the benefits of RAS inhibitors;7, 8, 9, 10 however, there is little information available about which combination therapy is most effective for elderly patients with hypertension. We carried out an open-label, randomized two-arm study to compare two combination therapies, including the angiotensin receptor blocker (ARB) olmesartan and either a CCB or a thiazide diuretic in elderly Japanese patients with hypertension.
Methods
Study subjects
We recruited elderly patients (aged 65–85 years) with essential hypertension with blood pressure levels of 140/90 mm Hg or higher for those on antihypertensive medication or 160/100 mm Hg or higher if they were not on medication between October 2007 and March 2009. Patients were considered to be eligible for this study if they had any of the following cardiovascular risk factors or diseases: impaired glucose tolerance, diabetes, dyslipidemia, proteinuria, left-ventricular hypertrophy, ischemic heart disease and stroke. The exclusion criteria were secondary or malignant hypertension, cardiovascular events within the last 6 months, heart failure of the New York Heart Association functional class III or VI, atrial fibrillation or flutter, severe arrhythmia, liver dysfunction, renal dysfunction with a serum creatinine level of 2.0 mg per 100 ml or higher and life-threatening conditions such as malignant tumors. Patients were also excluded if it was inappropriate to change their treatment to the study drugs or if they had a history of serious side-effects related to the study drugs.
Study protocols
This was an open-label, randomized, two-arm clinical study and it was carried out at 10 clinics affiliated with the University of Miyazaki Faculty of Medicine (the MOTHER study group). This study was conducted in accordance with the principles of the Declaration of Helsinki under the approval of the ethics committee of the University of Miyazaki Faculty of Medicine. All subjects gave their written informed consent before participating in this study. The data center of the Department of Medicine of the University of Miyazaki randomly assigned eligible patients to either the ARB/CCB group or the ARB/diuretic group at a 1:1 ratio using gender as a grouping factor. In addition to the ARB olmesartan, a dihydropyridine CCB, such as azelnidipine or amlodipine, or a thiazide diuretic, such as indapamide or trichlormethiazide, was used, and the initial doses of the drugs administered were determined on a case-by-case basis. If the eligible patients were taking ARBs or angiotensin-converting enzyme inhibitors, those drugs were replaced with the ARB olmesartan. There were no restrictions on the concomitant use of drugs other than antihypertensive agents. The target blood pressure during the treatment period was <140/90 mm Hg in both treatment arms, whereas further targets were set by the investigators when necessary. If the target blood pressure level was not achieved, the dose of olmesartan, the CCB or the diuretic was increased, and antihypertensive treatment was continued for 6 months.
Measurements
Blood pressure levels and pulse rates were measured at 1, 3 and 6 months after randomization, and the laboratory tests were conducted before and after the 6-month treatment. The glomerular filtration rate was estimated (eGFR) from the serum creatinine concentration using the following formula: 194 × serum creatinine−1.094 × age−0.287 (further multiplied by 0.739 for women), according to the guidelines of the Japanese Society of Nephrology.11 Plasma renin activity and the aldosterone concentration were measured by radioimmunoassays as previously described, and plasma brain natriuretic peptide was assessed using an immunoradiometric assay.12, 13
Statistical analysis
All data were analyzed with SPSS software version 18.0 (SPSS, Chicago, IL, USA). Comparisons between the two groups were assessed using the unpaired t-test or the χ2-test, and comparisons between values before and after treatment were made using the paired t-test. Analysis of variance followed by Scheffe's test was used for multiple comparisons. All data are expressed as means±s.d. unless otherwise indicated, and P-values less than 0.05 were considered to be significant.
Results
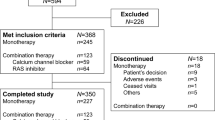
The 65 hypertensive patients, who were eligible for this study, were randomly assigned to combination therapy, including olmesartan and either a CCB (n=33) or a thiazide diuretic (n=32). The antihypertensive medicines prescribed before this study (n) were ARBs (30), angiotensin-converting enzyme inhibitors (1), CCBs (19), diuretic (1) and β-adrenergic blockers (3). Adverse events were reported for three and five patients of the CCB and diuretic combination groups, respectively. These events were sepsis, gout attack and cervical spondylosis in the CCB group, and inappropriate reduction or elevation of blood pressure, new onset of diabetes, bone fracture and positioning vertigo in the diuretic group. Among the patients experiencing adverse effects, three patients with sepsis and inappropriate reduction or elevation of blood pressure dropped out, but the remaining five completed the treatment protocol. Three patients stopped visiting the doctors’ offices because of transferring residences and one patient stopped visiting because of personal reasons. Therefore, 58 patients completed the treatment protocol, and their basal profiles are listed in Table 1; these profiles indicate that no significant differences between the CCB and diuretic combination groups were noted with respect to any of the parameters, including systolic and diastolic blood pressures (SBP and DBP). At the end of the treatment period, no difference was found with respect to the dose of olmesartan used between the two groups: CCB: 22.1±7.7 mg per day; and diuretic: 21.2±8.6 mg per day. The mean doses (mg per day) of CCB and diuretics used were as follows (n): azelnidipine: 13.3±3.9 (15); amlodipine: 3.5±1.3 (13); benidipine: 4 (1); trichlormethiazide: 1.47±0.51 (17); and indapamide: 0.96±0.40 (12).
Figure 1 shows the time course of blood pressure and pulse, and the reductions in blood pressure at 1, 3 and 6 months during the treatment period. Both SBP and DBP were significantly reduced at 1 month, and blood pressure-lowering effects were seen throughout the study period; no significant changes were noted for the pulse rate (Figure 1a). There were no differences in SBP or DBP between the CCB and diuretic combination groups, but as shown in Figure 1b, the SBP reduction (ΔSBP) relative to the control level observed in the CCB group was significantly greater than that in the diuretic group after 1 and 6 months of treatment.
We compared the clinical and humoral parameters of the patients before and after treatment for 6 months (Table 2). The total cholesterol levels were reduced in both groups, whereas the high-density lipoprotein cholesterol level was lowered by treatment with combination therapy, including a diuretic but not including a CCB. Neither the serum potassium nor the uric acid level changed in either group, but the serum creatinine level increased and eGFR decreased following combination treatment with a diuretic. The urinary albumin/creatinine ratios were substantially reduced in both groups; this reduction was statistically significant in the diuretic group but not in the CCB group. We found no significant difference in the Δurinary albumin/creatinine ratio between the two groups (39.7±40.2 vs. 24.7±7.9 mg g−1; means±s.e.). Plasma renin activity rose, and the aldosterone level was slightly reduced, but no differences were noted between the two treatment groups. The plasma brain natriuretic peptide levels remained unchanged.
Discussion
CCBs and thiazide diuretics are recommended as suitable drugs for combination with RAS inhibitors in recent guidelines for the treatment of hypertension, including those of the European Society of Hypertension and European Society of Cardiology (ESH-ESC 2007) and the Japanese Society of Hypertension (JSH 2009).1, 2 A number of clinical studies have demonstrated the ability of CCBs to reduce the incidence of cardiovascular events, although evidence for the efficacy of combination therapy, including an RAS inhibitor and a CCB, is limited.10 Meanwhile, many large-scale trials have been conducted to examine the effects of ARB or angiotensin-converting enzyme inhibitors in which a substantial number of patients were given diuretics as part of a combination treatment.7, 8, 9 Theoretically, the combination of an RAS inhibitor and a diuretic effectively lowers blood pressure because the former suppresses the activation of the RAS caused by the fluid volume reduction induced by the latter. For example, in the PROGRESS trial, the combination of perindopril and indapamide effectively reduced the recurrence of stroke, but monotherapy with perindopril did not.7 The antihypertensive effects of combination therapies, including diuretics are expected to be stronger in hypertensive patients in Japan, where the average salt intake is higher than in Western countries.14 However, in the present study, the standard clinical doses of the CCB and diuretics were used, and the combination therapy, including a CCB was found to be better than that including a diuretic in terms of blood pressure reduction.
RAS inhibitors have been shown to improve insulin resistance, whereas diuretics are known to have unfavorable effects on the glucose and lipid metabolism.1, 2, 15 In the present study, the total cholesterol levels were reduced in both combination groups, but this result was attributed to the new prescription of statins because the differences disappeared in the analysis with patients to whom no statins were given or whose prescription remained unchanged during the study period (data not shown). Meanwhile, irrespective of the prescription of statins, the high-density lipoprotein cholesterol level was found to be reduced by treatment with combination therapy that included a diuretic, but not in therapy that included a CCB. The J-LIT study, a large-scale cohort study of Japanese patients with hypercholesterolemia, showed a clear inverse relationship between high-density lipoprotein cholesterol levels and the risks of coronary events or cerebrovascular diseases.16, 17 Therefore, it is assumed that a diuretic-induced reduction in high-density lipoprotein cholesterol impairs the beneficial effects of diuretics, which are exerted through lowering the blood pressure, in Japanese patients with hypertension, but this important issue needs to be addressed by future clinical studies.
Antihypertensive treatment with diuretics is often associated with an increase in the serum creatinine level or a decrease in glomerular filtration rate, probably because the reduced fluid volume results in reduced renal blood flow.18 Similar patterns were detected in the present study, although we were unable to specify the mechanism of these changes, and the increase in the creatinine level and the decrease in glomerular filtration rate occurred despite the smaller reduction in blood pressure in the diuretic group. We need to compare the present results with those of the GUARD and ACCOMPLISH trials.19, 20, 21 Angiotensin-converting enzyme inhibitors were used in both of these studies, but the present findings are consistent with those of the GUARD study in which the treatment of hypertensive patients with type 2 diabetes was followed for 1 year. This study demonstrated that the reduction in the eGFRs of patients given benazepril plus hydrochlorothiazide (HCTZ) was greater than that observed in patients treated with benazepril plus amlodipine.19 In the GUARD study, urinary albumin secretion was reduced to a larger degree in the HCTZ combination group than in the amlodipine group,19 whereas in the present study, we observed decreased urinary albumin in both the groups without a difference in the Δurinary albumin. According to the recently published ACCOMPLISH trial results, which examined renal outcomes, doubling of the serum creatinine level and end-stage renal disease were more frequent in hypertensive patients treated with benazepril plus HCTZ than in those treated with benazepril plus amlodipine.21 We may, therefore need to raise the possibility that the effects of thiazide diuretics on renal function that were observed in the present study and in the GUARD trial can partly be translated into the hard end point of the progression of chronic kidney disease, including end-stage renal disease.
As discussed above, diuretics are expected to be beneficial for treating hypertensive patients in Japan. Recently, we reported the results of a single-arm clinical study examining the effects of a fixed-dose combination of 50 mg losartan and 12.5 mg HCTZ in Japanese hypertensive patients, for whom RAS inhibitors had not sufficiently lowered the blood pressure to achieve the goals recommended in the JSH guidelines.1, 5, 22 In that study, the combination of losartan and HCTZ was found to be effective for reducing blood pressure, and the blood pressure goals were achieved in a substantial number of patients.22 According to the CASE-J study, renal events are less common in patients treated with candesartan than in those treated with amlodipine, and a large number of the patients were given diuretics in that study.23 Thus, the long-term outcome of renal function following combination treatment, including an ARB and either a CCB or a thiazide diuretic, needs to be evaluated by further clinical trials, such as the COLM study, which examine the hard end points of renal events.24
In conclusion, antihypertensive treatment with olmesartan plus a dihydropyridine CCB was found to be a more favorable combination in terms of blood pressure reduction and its effects on renal function and lipid metabolism than a combination of olmesartan plus a thiazide diuretic in elderly patients with hypertension.
References
Ogihara T, Kikuchi K, Matsuoka H, Fujita T, Higaki J, Horiuchi M, Imai Y, Imaizumi T, Ito S, Iwao H, Kario K, Kawano Y, Kim-Mitsuyama S, Kimura G, Matsubara H, Matsuura H, Naruse M, Saito I, Shimada K, Shimamoto K, Suzuki H, Takishita S, Tanahashi N, Tsuchihashi T, Uchiyama M, Ueda S, Ueshima H, Umemura S, Ishimitsu T, Rakugi H, on behalf of The Japanese Society of Hypertension Committee for Guidelines for the Management of Hypertension. The Japanese Society of Hypertension Guidelines for the management of hypertension (JSH 2009). Hypertens Res 2009; 32: 3–107.
Mancia G, De Backer G, Dominiczak A, Cifkova R, Fagard R, Germano G, Grassi G, Heagerty AM, Kjeldsen SE, Laurent S, Narkiewicz K, Ruilope L, Rynkiewicz A, Schmieder RE, Boudier HA, Zanchetti A . 2007 guidelines for the management of arterial hypertension: the task force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens 2007; 25: 1105–1187.
Bakris GL, Williams M, Dworkin L, Elliott WJ, Epstein M, Toto R, Tuttle K, Douglas J, Hsueh W, Sowers J, for National Kidney Foundation Hypertension and Diabetes Executive Committees Working Group. Preserving renal function in adults with hypertension and diabetes: a consensus approach. Am J Kidney Dis 2000; 36: 646–661.
Hansson L, Zanchetti A, Carruthers SG, Dahlöf B, Elmfeldt D, Julius S, Ménard J, Rahn KH, Wedel H, Westerling S, for the HOT Study Group. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the hypertension optimal treatment (HOT) randomised trial. Lancet 1998; 351: 1755–1762.
Japanese Society of Hypertension. Japanese Society of Hypertension guidelines for the management of hypertension (JSH 2004). Hypertens Res 2006; 29 (Suppl): S1–S105.
Beckett NS, Peters R, Fletcher AE, Staessen JA, Liu L, Dumitrascu D, Stoyanovsky V, Antikainen RL, Nikitin Y, Anderson C, Belhani A, Forette F, Rajkumar C, Thijs L, Banya W, Bulpitt CJ, for the HYVET Study Group. Treatment of hypertension in patients 80 years of age or older. N Engl J Med 2008; 358: 1887–1898.
PROGRESS Collaborative Group. Randomised trial of a perindopril-based blood-pressure lowering regimen among 6,105 individuals with previous stroke or transient ischaemic attack. Lancet 2001; 358: 1033–1041.
Dahlöf B, Devereux RB, Kjeldsen SE, Julius S, Beevers G, de Faire U, Fyhrquist F, Ibsen H, Kristiansson K, Lederballe-Pedersen O, Lindholm LH, Nieminen MS, Omvik P, Oparil S, Wedel H, for the LIFE study group. Cardiovascular morbidity and mortality in the Losartan Intervention For End point reduction in hypertension study (LIFE): a randomised trial against atenolol. Lancet 2002; 359: 995–1003.
Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, Remuzzi G, Snapinn SM, Zhang Z, Shahinfar S, for the RENAAL Study Investigators. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 2001; 345: 861–869.
Dahlöf B, Sever PS, Poulter NR, Wedel H, Beevers DG, Caulfield M, Collins R, Kjeldsen SE, Kristinsson A, McInnes GT, Mehlsen J, Nieminen M, O’Brien E, Ostergren J, for the ASCOT investigators. Prevention of cardiovascular events with an antihypertensive regimen of amlodipine adding perindopril as required versus atenolol adding bendroflumethiazide as required, in the Anglo-Scandinavian cardiac outcomes trial-blood pressure lowering arm (ASCOT-BPLA): a multicentre randomised controlled trial. Lancet 2005; 366: 895–906.
Japanese Society of Nephrology. Evidence-based practice guideline for the treatment of CKD. Clin Exp Nephrol 2009; 13: 537–566.
Kato J, Kobayashi K, Etoh T, Tanaka M, Kitamura K, Imamura T, Koiwaya Y, Kangawa K, Eto T . Plasma adrenomedullin concentration in patients with heart failure. J Clin Endocrinol Metab 1996; 81: 180–183.
Kato J, Kitamura K, Uemura T, Kuwasako K, Kita T, Kangawa K, Eto T . Plasma levels of adrenomedullin and atrial and brain natriuretic peptides in the general population: their relations to age and pulse pressure. Hypertens Res 2002; 25: 887–892.
Zhou BF, Stamler J, Dennis B, Moag-Stahlberg A, Okuda N, Robertson C, Zhao L, Chan Q, Elliott P, for the INTERMAP Research Group. Nutrient intakes of middle-aged men and women in China, Japan, United Kingdom, and United States in the late 1990s: the INTERMAP study. J Hum Hypertens 2003; 17: 623–630.
ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: the antihypertensive and lipid-lowering treatment to prevent heart attack trial (ALLHAT). JAMA 2002; 288: 2981–2997.
Matsuzaki M, Kita T, Mabuchi H, Matsuzawa Y, Nakaya N, Oikawa S, Saito Y, Sasaki J, Shimamoto K, Itakura H, the J-LIT Study Group. Large scale cohort study of the relationship between serum cholesterol concentration and coronary events with low-dose simvastatin therapy in Japanese patients with hypercholesterolemia. Circ J 2002; 66: 1087–1095.
Nakaya N, Kita T, Mabuchi H, Matsuzaki M, Matsuzawa Y, Oikawa S, Saito Y, Sasaki J, Shimamoto K, Itakura H, the J-LIT Study Group. Large-scale cohort study on the relationship between serum lipid concentrations and risk of cerebrovascular disease under low-dose simvastatin in Japanese patients with hypercholesterolemia: sub-analysis of the Japan Lipid Intervention Trial (J-LIT). Circ J 2005; 69: 1016–1021.
Kaplan NM, Victor RG . Kaplan’s Clinical Hypertension, 10th edn. Lippincott Williams & Wilkins: Philadelphia, 2010, pp 192–273.
Bakris GL, Toto RD, McCullough PA, Rocha R, Purkayastha D, Davis P, On behalf of the GUARD (Gauging Albuminuria Reduction With Lotrel in Diabetic Patients With Hypertension) Study Investigators. Effects of different ACE inhibitor combinations on albuminuria: results of the GUARD study. Kidney Int 2008; 73: 1303–1309.
Jamerson K, Weber MA, Bakris GL, Dahlöf B, Pitt B, Shi V, Hester A, Gupte J, Gatlin M, Velazquez EJ, for the ACCOMPLISH trial investigators. Benazepril plus amlodipine or hydrochlorothiazide for hypertension in high-risk patients. N Engl J Med 2008; 359: 2417–2428.
Bakris GL, Sarafidis PA, Weir MR, Dahlöf B, Pitt B, Jamerson K, Velazquez EJ, Staikos-Byrne L, Kelly RY, Shi V, Chiang YT, Weber MA, for the ACCOMPLISH Trial investigators. Renal outcomes with different fixed-dose combination therapies in patients with hypertension at high risk for cardiovascular events (ACCOMPLISH): a prespecified secondary analysis of a randomised controlled trial. Lancet 2010; 375: 1173–1181.
Kita T, Yokota N, Ichiki Y, Ayabe T, Etoh T, Tamaki N, Kato J, Eto T, Kitamura K, for the PALM-1 study investigators. One-year effectiveness and safety of open-label losartan/hydrochlorothiazide combination therapy in Japanese patients with hypertension uncontrolled with ARBs or ACE inhibitors. Hypertens Res 2010; 33: 320–325.
Ogihara T, Nakao K, Fukui T, Fukiyama K, Ueshima K, Oba K, Sato T, Saruta T, for the Candesartan Antihypertensive Survival Evaluation in Japan Trial Group. Effects of candesartan compared with amlodipine in hypertensive patients with high cardiovascular risks: candesartan antihypertensive survival evaluation in Japan trial. Hypertension 2008; 51: 393–398.
Ogihara T, Saruta T, Rakugi H, Shimamoto K, Ito S, Matsuoka H, Horiuchi M, Imaizumi T, Takishita S, Higaki J, Katayama S, Saito I, Shimada K, on behalf of the COLM study investigators. Rationale, study design and implementation of the COLM study: the combination of olmesartan and calcium channel blocker or diuretic in high-risk elderly hypertensive patients. Hypertens Res 2009; 32: 163–167.
Acknowledgements
We are grateful for the participation of all the investigators involved in this study. The Miyazaki Olmesartan Therapy for Hypertension in the Elderly (MOTHER) study group included the following investigators: Takuma Etoh (Etoh Clinic), Toshinobu Higa (Miyazaki Higashi Hospital), Yuichiro Ishiyama (Miyazaki Higashi Hospital), Sumito Kariya (Kariya Iin), Hideyuki Komidori (Komidori Naika Clinic), Masatoshi Nishizono (Nishizono Naika), Masachika Kuwabara (Kuwabara Iin), Hiroshi Murata (Murata Nakika Ichoka), Sadahiko Sonoda (Nojiri Central Hospital), Noboru Tamaki (Tamaki Clinic) and Naoto Yokota (Yokota Naika).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kato, J., Yokota, N., Tamaki, N. et al. Comparison of combination therapies, including the angiotensin receptor blocker olmesartan and either a calcium channel blocker or a thiazide diuretic, in elderly patients with hypertension. Hypertens Res 34, 331–335 (2011). https://doi.org/10.1038/hr.2010.233
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2010.233
Keywords
This article is cited by
-
Thiazide Diuretic–Induced Change in Fasting Plasma Glucose: a Meta-analysis of Randomized Clinical Trials
Journal of General Internal Medicine (2020)
-
The benefits of angiotensin-converting enzyme inhibitors/angiotensin II receptor blockers combined with calcium channel blockers on metabolic, renal, and cardiovascular outcomes in hypertensive patients: a meta-analysis
International Urology and Nephrology (2018)
-
Antihypertensive and metabolic effects of hydrochlorothiazide versus amlodipine when added to losartan in patients with type 2 diabetes
Diabetology International (2016)
-
Olmesartan in the Treatment of Hypertension in Elderly Patients: a Review of the Primary Evidence
Drugs & Aging (2013)
-
Hydrochlorothiazide Versus Calcium Channel Blockers: What is the Best Add-on to a Renin-Angiotensin System Blocker for Treating Hypertension in Patients with Renal Disease?
Current Hypertension Reports (2011)