Abstract
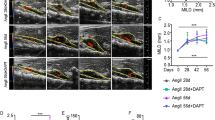
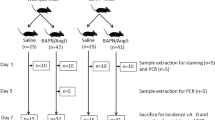
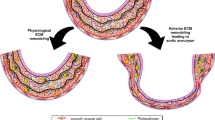
In this study, we investigated the effect of a specific chymase inhibitor, NK3201, in the progression of abdominal aortic aneurysm in a dog experimental model. Abdominal aortic aneurysms were induced in dogs by injecting elastase into the abdominal aorta. NK3201 (1 mg/kg per day, p.o.) or a placebo was started 3 days before elastase injection and continued for 8 weeks after the injection. On abdominal ultrasound, the aortic diameter was seen to gradually expand in the placebo-treated group, but not in the NK3201-treated group. Eight weeks after elastase injection, the ratio of the medial area to the total area in the placebo-treated group was significantly smaller than that in the normal group, but it was significantly larger than that in the NK3201-treated group. In addition to chymase activity, angiotensin II–forming and matrix metalloproteinase-9 activities were significantly higher in the placebo-treated group than in the normal group; in the NK3201-treated group, all of these activities were significantly decreased. On immunohistochemical analyses, there was a significantly greater number of chymase-positive cells in the placebo-treated group than in the normal group, but the number was significantly smaller in the NK3201-treated group than in the placebo-treated group. Thus, chymase inhibition may become a useful strategy for preventing abdominal aortic aneurysms.
Similar content being viewed by others
Article PDF
References
Thompson RW, Holmes DR, Mertens RA, et al: Production and localization of 92-kilodalton gelatinase in abdominal aortic aneurysms. An elastolytic metalloproteinase expressed by aneurysm-infiltrating macrophages. J Clin Invest 1995; 96: 318–326.
Freestone T, Turner RJ, Coady A, Higman DJ, Greenhalgh RM, Powell JT : Inflammation and matrix metalloproteinases in the enlarging abdominal aortic aneurysm. Arterioscler Thromb Vasc Biol 1995; 15: 1145–1151.
McMillan WD, Tamarina NA, Cipollone M, Johnson DA, Parker MA, Pearce WH : Size matters: the relationship between MMP-9 expression and aortic diameter. Circulation 1997; 96: 2228–2232.
Fang KC, Raymond WW, Blount JL, Caughey GH : Dog mast cell α-chymase activates progelatinase B by cleaving the Phe88-Gln89 and Phe91-Glu92 bonds of the catalytic domain. J Biol Chem 1997; 272: 25628–25635.
Pyo R, Lee JK, Shipley JM, et al: Targeted gene disruption of matrix metalloproteinase-9 (gelatinase B) suppresses development of experimental abdominal aortic aneurysms. J Clin Invest 2000; 105: 1641–1649.
Longo GM, Xiong W, Greiner TC, Zhao Y, Fiotti N, Baxter BT : Matrix metalloproteinases 2 and 9 work in concert to produce aortic aneurysms. J Clin Invest 2002; 110: 625–632.
Urata H, Kinoshita A, Misono K, Bumpus FM, Husain A : Identification of a highly specific chymase as the major angiotensin II–forming enzyme in the human heart. J Biol Chem 1990; 265: 22348–22357.
Takai S, Shiota N, Sakaguchi M, Muraguchi H, Matsumura E, Miyazaki M : Characterization of chymase from human vascular tissues. Clin Chim Acta 1997; 265: 13–20.
Caughey GH, Raymond WW, Wolters PJ : Angiotensin II generation by mast cell α- and β-chymases. Biochim Biophys Acta 2000; 1480: 245–257.
Nishimoto M, Takai S, Fukumoto H, et al: Increased local angiotensin II formation in aneurysmal aorta. Life Sci 2002; 71: 2195–2205.
Tsunemi K, Takai S, Nishimoto M, et al: Possible roles of angiotensin II–forming enzymes, angiotensin converting enzyme and chymase-like enzyme, in the human aneurysmal aorta. Hypertens Res 2002; 25: 817–822.
Daugherty A, Manning MW, Cassis LA : Angiotensin II promotes atherosclerotic lesions and aneurysms in apolipoprotein E–deficient mice. J Clin Invest 2000; 105: 1605–1612.
Ejiri J, Inoue N, Tsukube T, et al: Oxidative stress in the pathogenesis of thoracic aortic aneurysm: protective role of statin and angiotensin II type 1 receptor blocker. Cardiovasc Res 2003; 59: 988–996.
Tsunemi K, Takai S, Nishimoto M, et al: A specific chymase inhibitor, 2-(5-formylamino-6-oxo-2-phenyl-1,6-dihydropyrimidine-1-yl)-N-[[3,4-dioxo-1-phenyl-7-(2-pyridyloxy)]-2-heptyl]acetamide (NK3201), suppresses development of abdominal aortic aneurysm in hamsters. J Pharmacol Exp Ther 2004; 309: 879–883.
Takai S, Miyazaki M : Application of a chymase inhibitor, NK3201, for prevention of vascular proliferation. Cardiovasc Drug Rev 2003; 21: 185–198.
Cecconi M, Manfrin M, Moraca A, et al: Aortic dimensions in patients with bicuspid aortic valve without significant valve dysfunction. Am J Cardiol 2005; 95: 292–294.
Nishimoto M, Takai S, Kim S, et al: Significance of chymase-dependent angiotensin II–forming pathway in the development of vascular proliferation. Circulation 2001; 104: 1274–1279.
Jin D, Ueda H, Takai S, et al: Effect of chymase inhibition on the arteriovenous fistula stenosis in dogs. J Am Soc Nephrol 2005; 16: 1024–1034.
Todd MB, Waldron JA, Jennings TA, et al: Loss of myeloid differentiation antigens precedes blastic transformation in chronic myelogenous leukemia. Blood 1987; 70: 122–131.
Kirimura K, Takai S, Jin D, et al: Role of chymase-dependent angiotensin II formation in regulating blood pressure in spontaneously hypertensive rats. Hypertens Res 2005; 28: 457–464.
Jin D, Takai S, Sakaguchi M, Okamoto Y, Muramatsu M, Miyazaki M : An antiarrhythmic effect of a chymase inhibitor after myocardial infarction. J Pharmacol Exp Ther 2004; 309: 490–497.
Kanemitsu H, Takai S, Tsuneyoshi H, et al: Chymase inhibition prevents cardiac fibrosis and dysfunction after myocardial infarction in rats. Hypertens Res 2006; 29: 57–64.
Tchougounova E, Lundequist A, Fajardo I, Winberg JO, Abrink M, Pejler G : A key role for mast cell chymase in the activation of pro-matrix metalloprotease-9 and pro-matrix metalloprotease-2. J Biol Chem 2005; 280: 9291–9296.
Vine N, Powell JT : Metalloproteinases in degenerative aortic disease. Clin Sci 1991; 81: 233–239.
Knox JB, Sukhova GK, Whittemore AD, Libby P : Evidence for altered balance between matrix metalloproteinases and their inhibitors in human aortic diseases. Circulation 1997; 95: 205–212.
Longley BJ, Tyrrell L, Ma Y, et al: Chymase cleavage of stem cell factor yields a bioactive, soluble product. Proc Natl Acad Sci U S A 1997; 94: 9017–9021.
Takai S, Jin D, Sakaguchi M, et al: A novel chymase inhibitor, 4-[1-([bis-(4-methyl-phenyl)-methyl]-carbamoyl)3-(2-ethoxy-benzyl)-4-oxo-azetidine-2-yloxy]-benzoic acid (BCEAB), suppressed cardiac fibrosis in cardiomyopathic hamsters. J Pharmacol Exp Ther 2003; 305: 17–23.
Collins T, Read MA, Neish AS, Whitley MZ, Thanos D, Maniatis T : Transcriptional regulation of endothelial cell adhesion molecules: NF-κB and cytokine-inducible enhancers. FASEB J 1995; 9: 899–909.
Hernandez-Presa M, Bustos C, Ortego M, et al: Angiotensin-converting enzyme inhibition prevents arterial nuclear factor-κB activation, monocyte chemoattractant protein-1 expression, and macrophage infiltration in a rabbit model of early accelerated atherosclerosis. Circulation 1997; 95: 1532–1541.
Chen XL, Tummala PE, Olbrych MT, Alexander RW, Medford RM : Angiotensin II induces monocyte chemoattractant protein-1 gene expression in rat vascular smooth muscle cells. Circ Res 1998; 83: 952–959.
Mabuchi T, Kitagawa K, Ohtsuki T, et al: Contribution of microglia/macrophages to expansion of infarction and response of oligodendrocytes after focal cerebral ischemia in rats. Stroke 2000; 31: 1735–1743.
Hilgers KF, Hartner A, Porst M, et al: Monocyte chemoattractant protein-1 and macrophage infiltration in hypertensive kidney injury. Kidney Int 2000; 58: 2408–2419.
Fontaine V, Jacob MP, Houard X, et al: Involvement of the mural thrombus as a site of protease release and activation in human aortic aneurysms. Am J Pathol 2002; 161: 1701–1710.
Senior RM, Griffin GL, Mecham RP : Chemotactic activity of elastin-derived peptides. J Clin Invest 1980; 66: 859–862.
Hance KA, Tataria M, Ziporin SJ, Lee JK, Thompson RW : Monocyte chemotactic activity in human abdominal aortic aneurysms: role of elastin degradation peptides and the 67-kD cell surface elastin receptor. J Vasc Surg 2002; 35: 254–261.
Anidjar S, Dobrin PB, Eichorst M, Graham GP, Chejfec G : Correlation of inflammatory infiltrate with the enlargement of experimental aortic aneurysms. J Vasc Surg 1992; 16: 139–147.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Furubayashi, K., Takai, S., Jin, D. et al. The Significance of Chymase in the Progression of Abdominal Aortic Aneurysms in Dogs. Hypertens Res 30, 349–357 (2007). https://doi.org/10.1291/hypres.30.349
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1291/hypres.30.349
Keywords
This article is cited by
-
Histone deacetylase inhibition disturbs the balance between ACE and chymase expression in endothelial cells: a potential mechanism of chymase activation in preeclampsia
Hypertension Research (2019)
-
TElmisartan in the management of abDominal aortic aneurYsm (TEDY): The study protocol for a randomized controlled trial
Trials (2015)
-
Mast cells in human and experimental cardiometabolic diseases
Nature Reviews Cardiology (2015)
-
Possible roles of mast cell-derived chymase for skin rejuvenation
Lasers in Medical Science (2009)
-
The role of the renin-angiotensin system in aortic aneurysmal diseases
Current Hypertension Reports (2008)