Abstract
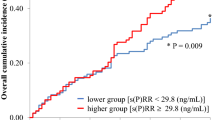
A high plasma aldosterone concentration (PAC) is known to be associated with poor outcome in patients with cardiac disease. However, the prognostic value of PAC in chronic hemodialysis (HD) patients is unknown. In 1996 we examined 128 hypertensive patients treated with antihypertensive drugs, excluding angiotensin-converting enzyme inhibitors, who were undergoing chronic HD (ages 61.8±13.8 years, 62% male), and for whom PAC (ng/dl) data were obtained. We followed up these patients until November 2003. During the follow-up period, 30 patients died. About half of all patients (48%) had PAC values above the normal range. We assigned the 128 patients to a lower (<22.9) or higher (≥22.9) PAC group according to the median baseline PAC. The survival rate as calculated by the Kaplan-Meier method was 90.6% in the higher PAC group and 62.5% in the lower PAC group (p=0.003). In multivariate analysis, serum potassium and plasma renin activity were independent determinants of PAC. Cox proportional hazards analysis, with adjustment for other variables including diabetes, showed that lower PAC was independently predictive of death. The adjusted hazard ratio (95% confidence interval) of the lower PAC group was 2.905 (1.187–7.112, p=0.020). The significance of PAC became marginal by adjustment with albumin or potassium. These results indicate that higher PAC is common, but not associated with an increase in total and cardiovascular deaths among hypertensive patients undergoing chronic HD. The association between lower PAC and poor survival may be driven by volume retention and/or lower potassium.
Similar content being viewed by others
Article PDF
References
Lonn EM, Yusuf S, Jha P, et al: Emerging role of angiotensin-converting enzyme inhibitors in cardiac and vascular protection. Circulation 1994; 90: 2056–2069.
Givertz MM : Manipulation of the renin-angiotensin system. Circulation 2001; 104: E14–E18.
Weber KT, Brilla CG : Pathological hypertrophy and cardiac interstitium. Fibrosis and renin-angiotensin-aldosterone system. Circulation 1991; 83: 1849–1865.
Pitt B, Reichek N, Willenbrock R, et al: Effects of eplerenone, enalapril, and eplerenone/enalapril in patients with essential hypertension and left ventricular hypertrophy: the 4E-left ventricular hypertrophy study. Circulation 2003; 108: 1831–1838.
Swedberg K, Eneroth P, Kjekshus J, Wilhelmsen L, CONSENSUS Trial Study Group: Hormones regulating cardiovascular function in patients with severe congestive heart failure and their relation to mortality. Circulation 1990; 82: 1730–1736.
Rouleau JL, Packer M, Moye L : Prognostic value of neurohumoral activation in patients with an acute myocardial infarction: effect of captopril. J Am Coll Cardiol 1994; 24: 583–591.
Sato A, Hayashi K, Narse M, Saruta T : Effectiveness aldosterone blockade in patients with diabetic nephropathy. Hypertension 2003; 41: 64–68.
Tozawa M, Iseki K, Iseki C, Takishita S : Pulse pressure and risk of total mortality and cardiovascular events in patients on chronic hemodialysis. Kidney Int 2002; 61: 717–726.
Schlaich MP, Schobel HP, Hilgers K, Schmieder RE : Impact of aldosterone on left ventricular structure and function in young normotensive and mildly hypertensive subjects. Am J Cardiol 2000; 85: 1199–1206.
Rocha R, Chander PN, Khanna K, Zuckerman A, Stier CT Jr : Mineralocorticoid blockade reduces vascular injury in stroke-prone hypertensive rats. Hypertension 1998; 31: 451–458.
Park JB, Schiffrin EL : Cardiac and vascular fibrosis and hypertrophy in aldosterone-infused rats: role of endothelin-1. Am J Hypertens 2002; 15: 164–169.
Lacolley P, Labat C, Pujol A, Delcayre C, Benetos A, Safar M : Increased carotid wall elastic modulus and fibronectin in aldosterone-salt–treated rats: effects of eplerenone. Circulation 2002; 106: 2848–2853.
Sato A, Funder JW, Saruta T : Involvement of aldosterone in left ventricular hypertrophy of patients with end-stage renal failure treated with hemodialysis. Am J Hypertens 1999; 12: 867–873.
Endemann DH, Touyz RM, Iglarz M, Savoia C, Schiffrin EL : Eplerenone prevents salt-induced vascular remodeling and cardiac fibrosis in stroke-prone spontaneously hypertensive rats. Hypertension 2004; 43: 1252–1257.
Sato A, Saruta T : Aldosterone-induced organ damage: plasma aldosterone level and inappropriate salt status. Hypertens Res 2004; 27: 303–310.
Hene RJ, Boer P, Koomans HA, Mees EJ : Plasma aldosterone concentrations in chronic renal disease. Kidney Int 1982; 21: 98–101.
Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY : Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med 2004; 351: 1296–1305.
Weber KT, Janicki JS, Pick R, Capasso J, Anversa P : Myocardial fibrosis and pathologic hypertrophy in the rat with renovascular hypertension. Am J Cardiol 1990; 65: 1G–7G.
Campbell SE, Janicki JS, Matsubara BB, Weber KT : Myocardial fibrosis in the rat with mineralocorticoid excess. Prevention of scarring by amiloride. Am J Hypertens 1993; 6: 487–495.
Franse LV, Pahor M, Di Bari M, Somes GW, Cushman WC, Applegate WB : Hypokalemia associated with diuretic use and cardiovascular events in the Systolic Hypertension in the Elderly Program. Hypertension 2000; 35: 1025–1030.
Wilson DR, Ing TS, Metcalfe-Gibson A, Wrong OM : The chemical composition of faeces in uraemia, as revealed by in-vivo faecal dialysis. Clin Sci 1968; 35: 197–209.
Oliver WJ, Cohen EL, Neel JV : Blood pressure, sodium intake, and sodium related hormones in the Yanomamo Indians, a “no-salt” culture. Circulation 1975; 52: 146–151.
Fernandez RG, Leehan JA, Pastrana RF, Muniz RO : Effect of malnutrition on K+ current in T lymphocytes. Clin Diagn Lab Immunol 2005; 12: 808–813.
Hatakeyama H, Miyamori I, Fujita T, Takeda Y, Takeda R, Yamamoto H : Vascular aldosterone. Biosynthesis and a link to angiotensin II–induced hypertrophy of vascular smooth muscle cells. J Biol Chem 1994; 269: 24316–24320.
Silvestre JS, Robert V, Heymes C, et al: Myocardial production of aldosterone and corticosterone in the rat. Physiological regulation. J Biol Chem 1998; 273: 4883–4891.
Takeda Y, Yoneda T, Demura M, Miyamori I, Mabuchi H : Cardiac aldosterone production in genetically hypertensive rats. Hypertension 2000; 36: 495–500.
Kigoshi T, Uehara K, Furuya K, et al: Relationship between impaired aldosterone response to adrenocorticotropic hormone and prevalence of hemodialysis in type 2 diabetic patients without azotemia. Hypertens Res 2005; 28: 21–26.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kohagura, K., Higashiuesato, Y., Ishiki, T. et al. Plasma Aldosterone in Hypertensive Patients on Chronic Hemodialysis: Distribution, Determinants and Impact on Survival. Hypertens Res 29, 597–604 (2006). https://doi.org/10.1291/hypres.29.597
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1291/hypres.29.597
Keywords
This article is cited by
-
A case of primary aldosteronism with excessive secretion of renin that was unmasked by kidney transplantation
CEN Case Reports (2023)
-
High cortisol levels are associated with oxidative stress and mortality in maintenance hemodialysis patients
BMC Nephrology (2022)
-
Relationship between parathyroid hormone and renin–angiotensin–aldosterone system in hemodialysis patients with secondary hyperparathyroidism
Journal of Bone and Mineral Metabolism (2021)
-
Impact of serum potassium on therapeutic prognosis of maintenance hemodialysis patients on angiotensin receptor antagonists
Renal Replacement Therapy (2016)