Abstract
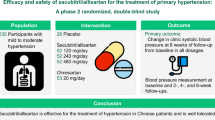
Aliskiren is a novel orally active renin inhibitor for the treatment of hypertension. This study evaluated the antihypertensive efficacy, safety and tolerability of aliskiren in Japanese patients with hypertension. Forty hundred and fifty-five Japanese men and women with a mean sitting diastolic blood pressure of 95–110 mmHg were randomized to receive once-daily double-blind treatment for 8 weeks with aliskiren 75, 150 or 300 mg or placebo. Aliskiren produced significant, dose-dependent reductions in mean sitting diastolic blood pressure (p<0.0005 vs. placebo for each dose) and mean sitting systolic blood pressure (p<0.001 vs. placebo for each dose). The placebo-corrected reductions in mean sitting systolic/diastolic blood pressure were 5.7/4.0, 5.9/4.5 and 11.2/7.5 mmHg in the aliskiren 75, 150 and 300 mg groups, respectively. After 8 weeks' treatment, 27.8%, 47.8%, 48.2% and 63.7% of patients in the placebo and aliskiren 75, 150 and 300 mg groups, respectively, achieved a successful treatment response (diastolic blood pressure <90 mmHg and/or reduced by ≥10 mmHg from baseline; p<0.005 vs. placebo for each dose). Aliskiren treatment was well tolerated, with the incidence of adverse events reported in the active treatment groups (53–55%) being similar to that in the placebo group (50%). This study, which is the first to assess the antihypertensive efficacy and safety of aliskiren in Japanese patients with hypertension, demonstrates that the once-daily oral renin inhibitor aliskiren provides significant, dose-dependent reductions in blood pressure with placebo-like tolerability.
Similar content being viewed by others
Article PDF
References
Integrated Management of Cardiovascular Risk, Report of a WHO Meeting. Geneva, 9–12 July 2002. Geneva, WHO, 2002.
The World Health Report 2002—Reducing Risks, Promoting Healthy Life. Geneva, WHO, 2002.
Chobanian AV, Bakris GL, Black HR, et al: Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension 2003; 42: 1206–1252.
Hozawa A, Ohkubo T, Kikuya M, et al: Blood pressure control assessed by home, ambulatory and conventional blood pressure measurements in the Japanese general population: the Ohasama study. Hypertens Res 2002; 25: 57–63.
Morgan T : Renin angiotensin, sodium and organ damage. Hypertens Res 2003; 26: 349–354.
Mori H, Ukai H, Yamamoto H, et al: Current status of antihypertensive prescription and associated blood pressure control in Japan. Hypertens Res 2006; 29: 143–151.
Dzau VJ : Theodore Cooper Lecture: tissue angiotensin and pathobiology of vascular disease: a unifying hypothesis. Hypertension 2001; 37: 1047–1052.
Weir MR, Dzau VJ : The renin-angiotensin-aldosterone system: a specific target for hypertension management. Am J Hypertens 1999; 12: 205S–213S.
Dahlof B, Devereux RB, Kjeldsen SE, et al: Cardiovascular morbidity and mortality in the Losartan Intervention For Endpoint reduction in hypertension study (LIFE): a randomised trial against atenolol. Lancet 2002; 359: 995–1003.
Dahlof B, Sever PS, Poulter NR, et al: Prevention of cardiovascular events with an antihypertensive regimen of amlodipine adding perindopril as required versus atenolol adding bendroflumethiazide as required, in the Anglo-Scandinavian Cardiac Outcomes Trial–Blood Pressure Lowering Arm (ASCOT-BPLA): a multicentre randomised controlled trial. Lancet 2005; 366: 895–906.
Yusuf S, Sleight P, Pogue J, Bosch J, Davies R, Dagenais G, The Heart Outcomes Prevention Evaluation Study Investigators : Effects of an angiotensin-converting–enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. N Engl J Med 2000; 342: 145–153.
Tomiyama H, Kushiro T, Abeta H, et al: Kinins contribute to the improvement of insulin sensitivity during treatment with angiotensin converting enzyme inhibitor. Hypertension 1994; 23: 450–455.
Israili ZH, Hall WD : Cough and angioneurotic edema associated with angiotensin-converting enzyme inhibitor therapy. A review of the literature and pathophysiology. Ann Intern Med 1992; 117: 234–242.
Hollenberg NK, Fisher ND, Price DA : Pathways for angiotensin II generation in intact human tissue: evidence from comparative pharmacological interruption of the renin system. Hypertension 1998; 32: 387–392.
Wolny A, Clozel JP, Rein J, et al: Functional and biochemical analysis of angiotensin II–forming pathways in the human heart. Circ Res 1997; 80: 219–227.
Okazaki H, Minamino T, Tsukamoto O, et al: Angiotensin II type 1 receptor blocker prevents atrial structural remodeling in rats with hypertension induced by chronic nitric oxide inhibition. Hypertens Res 2006; 29: 277–284.
Williams B : Angiotensin II and the pathophysiology of cardiovascular remodeling. Am J Cardiol 2001; 87: 10C–17C.
Senbonmatsu T, Ichihara S, Price E Jr, Gaffney FA, Inagami T : Evidence for angiotensin II type 2 receptor–mediated cardiac myocyte enlargement during in vivo pressure overload. J Clin Invest 2000; 106: R25–R29.
Azizi M, Menard J, Bissery A, et al: Pharmacologic demonstration of the synergistic effects of a combination of the renin inhibitor aliskiren and the AT1 receptor antagonist valsartan on the angiotensin II-renin feedback interruption. J Am Soc Nephrol 2004; 15: 3126–3133.
Nussberger J, Wuerzner G, Jensen C, Brunner HR : Angiotensin II suppression in humans by the orally active renin inhibitor Aliskiren (SPP100): comparison with enalapril. Hypertension 2002; 39: E1–E8.
Skeggs LT Jr, Kahn JR, Lentz K, Shumway NP : The preparation, purification, and amino acid sequence of a polypeptide renin substrate. J Exp Med 1957; 106: 439–453.
Fisher ND, Hollenberg NK : Renin inhibition: what are the therapeutic opportunities? J Am Soc Nephrol 2005; 16: 592–599.
Kokubu T, Hiwada K, Murakami E, Muneta S, Kitami Y, Salmon PF : ES-8891, an orally active inhibitor of human renin. Hypertension 1990; 15: 909–913.
Weber MA, Neutel JM, Essinger I, Glassman HN, Boger RS, Luther R : Assessment of renin dependency of hypertension with a dipeptide renin inhibitor. Circulation 1990; 81: 1768–1774.
Neutel JM, Luther RR, Boger RS, Weber MA : Immediate blood pressure effects of the renin inhibitor enalkiren and the angiotensin-converting enzyme inhibitor enalaprilat. Am Heart J 1991; 122: 1094–1100.
van den Meiracker AH, Admiraal PJ, Derkx FH, et al: Comparison of blood pressure and angiotensin responses to the renin inhibitor Ro 42–5892 and the angiotensin converting enzyme inhibitor enalapril in essential hypertension. J Hypertens 1993; 11: 831–838.
Kobrin I, Viskoper RJ, Laszt A, Bock J, Weber C, Charlon V : Effects of an orally active renin inhibitor, Ro 42–5892, in patients with essential hypertension. Am J Hypertens 1993; 6: 349–356.
Fisher ND, Hollenberg NK : Is there a future for renin inhibitors? Expert Opin Investig Drugs 2001; 10: 417–426.
Wood JM, Maibaum J, Rahuel J : Structure-based design of aliskiren, a novel orally effective renin inhibitor. Biochem Biophys Res Commun 2003; 308: 698–705.
Rahuel J, Rasetti V, Maibaum J, et al: Structure-based drug design: the discovery of novel nonpeptide orally active inhibitors of human renin. Chem Biol 2000; 7: 493–504.
Vaidyanathan S, Limoges D, Yeh C-M, Dieterich H-A : Aliskiren, an orally effective renin inhibitor, shows dose linear pharmacokinetics in healthy volunteers. Clin Pharmacol Ther 2006; 79: P64.
Gradman AH, Schmieder RE, Lins RL, et al: Aliskiren, a novel orally effective renin inhibitor, provides dose-dependent antihypertensive efficacy and placebo-like tolerability in hypertensive patients. Circulation 2005; 111: 1012–1018.
Stanton A, Jensen C, Nussberger J, O'Brien E : Blood pressure lowering in essential hypertension with an oral renin inhibitor, aliskiren. Hypertension 2003; 42: 1137–1143.
Williams B, Poulter NR, Brown MJ, et al: Guidelines for management of hypertension: report of the fourth working party of the British Hypertension Society, 2004-BHS IV. J Hum Hypertens 2004; 18: 139–185.
Frattola A, Parati G, Cuspidi C, Albini F, Mancia G : Prognostic value of 24-hour blood pressure variability. J Hypertens 1993; 11: 1133–1137.
Neutel JM, Smith DH, Weber MA : Low-dose combination therapy: an important first-line treatment in the management of hypertension. Am J Hypertens 2001; 14: 286–292.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kushiro, T., Itakura, H., Abo, Y. et al. Aliskiren, a Novel Oral Renin Inhibitor, Provides Dose-Dependent Efficacy and Placebo-Like Tolerability in Japanese Patients with Hypertension. Hypertens Res 29, 997–1005 (2006). https://doi.org/10.1291/hypres.29.997
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1291/hypres.29.997
Keywords
This article is cited by
-
The role of aliskiren in the management of hypertension and major cardiovascular outcomes: a systematic review and meta-analysis
Journal of Human Hypertension (2019)
-
Chapter 5. Treatment with antihypertensive drugs
Hypertension Research (2014)
-
Effect of aldosterone breakthrough on albuminuria during treatment with a direct renin inhibitor and combined effect with a mineralocorticoid receptor antagonist
Hypertension Research (2013)
-
Aliskiren vs. other antihypertensive drugs in the treatment of hypertension: a meta-analysis
Hypertension Research (2013)
-
Efficacy analysis of the renoprotective effects of aliskiren in hypertensive patients with chronic kidney disease
Heart and Vessels (2013)