Abstract
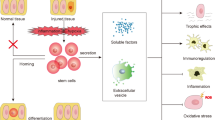
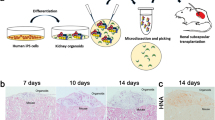
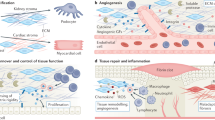
Functional recovery in acute renal failure is well known, and the adult kidney is generally recognized to have the capacity to regenerate and repair. Several groups have reported the contribution of bone marrow–derived cells in this process, and others have confirmed the existence of adult stem cells in the kidney, including slow-cycling cells, side population cells, CD133+ cells and rKS56 cells. However, recent data demonstrated that in vivo differentiation of bone marrow–derived cells into renal tubular cells may not occur at all, or is at most a minor component of the repair process. Moreover, it is now generally accepted that stem cells and multipotent cells contribute to the regenerative process by producing protective and regenerative factors rather than by directly differentiating to replace damaged cells. Therefore, for clinical regenerative medicine in kidney disease, the focus of stem cell biology will shift from multiple differentiation of cells or cell-therapy to multiple functions of the cells, such as the production of bone morphologic protein-7 and other regenerative factors.
Similar content being viewed by others
Article PDF
References
Rosenthal N : Prometheus's vulture and the stem-cell promise. N Engl J Med 2003; 349: 267–274.
Fioretto P, Steffes MW, Sutherland DE, Goetz FC, Mauer M : Reversal of lesions of diabetic nephropathy after pancreas transplantation. N Engl J Med 1998; 339: 69–75.
Zeisberg M, Hanai J, Sugimoto H, et al: BMP-7 counteracts TGF-beta1−induced epithelial-to-mesenchymal transition and reverses chronic renal injury. Nat Med 2003; 9: 964–968.
Dekel B, Burakova T, Arditti FD, et al: Human and porcine early kidney precursors as a new source for transplantation. Nat Med 2003; 9: 53–60.
Poulsom R, Forbes SJ, Hodivala-Dilke K, et al: Bone marrow contributes to renal parenchymal turnover and regeneration. J Pathol 2001; 195: 229–235.
Gupta S, Verfaillie C, Chmielewski D, Kim Y, Rosenberg ME : A role for extrarenal cells in the regeneration following acute renal failure. Kidney Int 2002; 62: 1285–1290.
Anjos-Afonso F, Siapati EK, Bonnet D : In vivo contribution of murine mesenchymal stem cells into multiple cell-types under minimal damage conditions. J Cell Sci 2004; 117: 5655–5664.
Szczypka MS, Westover AJ, Clouthier SG, Ferrara JL, Humes HD : Rare incorporation of bone marrow–derived cells into kidney after folic acid–induced injury. Stem Cells 2005; 23: 44–54.
Duffield JS, Park KM, Hsiao LL, et al: Restoration of tubular epithelial cells during repair of the postischemic kidney occurs independently of bone marrow–derived stem cells. J Clin Invest 2005; 115: 1743–1755.
Krause D, Cantley LG : Bone marrow plasticity revisited: protection or differentiation in the kidney tubule? J Clin Invest 2005; 115: 1705–1708.
Cotsarelis G, Cheng SZ, Dong G, Sun TT, Lavker RM : Existence of slow-cycling limbal epithelial basal cells that can be preferentially stimulated to proliferate: implications on epithelial stem cells. Cell 1989; 57: 201–209.
Johansson CB, Momma S, Clarke DL, Risling M, Lendahl U, Frisen J : Identification of a neural stem cell in the adult mammalian central nervous system. Cell 1999; 96: 25–34.
Lavker RM, Sun TT : Epidermal stem cells: properties markers and location. Proc Natl Acad Sci U S A 2000; 97: 13473–13475.
Oliver JA, Maarouf O, Cheema FH, Martens TP, Al-Awqati Q : The renal papilla is a niche for adult kidney stem cells. J Clin Invest 2004; 114: 795–804.
Goodell MA, Brose K, Paradis G, Conner AS, Mulligan RC : Isolation and functional properties of murine hematopoietic stem cells that are replicating in vivo. J Exp Med 1996; 183: 1797–1806.
Zhou S, Schuetz JD, Bunting KD, et al: The ABC transporter Bcrp1/ABCG2 is expressed in a wide variety of stem cells and is a molecular determinant of the side-population phenotype. Nat Med 2001; 7: 1028–1034.
Ivanova NB, Dimos JT, Schaniel C, Hackney JA, Moore KA, Lemischka IR : A stem cell molecular signature. Science 2002; 298: 601–604.
Ramalho-Santos M, Yoon S, Matsuzaki Y, Mulligan RC, Melton DA : “Stemness”: transcriptional profiling of embryonic and adult stem cells. Science 2002; 298: 597–600.
Easterday MC, Dougherty JD, Jackson RL, et al: Neural progenitor genes. Germinal zone expression and analysis of genetic overlap in stem cell populations. Dev Biol 2003; 264: 309–322.
Asakura A, Seale P, Girgis-Gabardo A, Rudnicki MA : Myogenic specification of side population cells in skeletal muscle. J Cell Biol 2002; 159: 123–134.
Majka SM, Jackson KA, Kienstra KA, Majesky MW, Goodell MA, Hirschi KK : Distinct progenitor populations in skeletal muscle are bone marrow derived and exhibit different cell fates during vascular regeneration. J Clin Invest 2003; 111: 71–79.
Jackson KA, Majka SM, Wang H, et al: Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells. J Clin Invest 2001; 107: 1395–1402.
Olmsted-Davis EA, Gugala Z, Camargo F, et al: Primitive adult hematopoietic stem cells can function as osteoblast precursors. Proc Natl Acad Sci U S A 2003; 100: 15877–15882.
Hishikawa K, Marumo T, Miura S, et al: Leukemia inhibitory factor induces multi-lineage differentiation of adult stem-like cells in kidney via kidney-specific cadherin 16. Biochem Biophys Res Commun 2005; 328: 288–291.
Hishikawa K, Marumo T, Miura S, et al: Musculin/MyoR is expressed in kidney side population cells and can regulate their function. J Cell Biol 2005; 169: 921–928.
Bussolati B, Bruno S, Grange C, et al: Isolation of renal progenitor cells from adult human kidney. Am J Pathol 2005; 166: 545–555.
Cha JH, Kim YH, Jung JY, Han KH, Madsen KM, Kim J : Cell proliferation in the loop of Henle in the developing rat kidney. J Am Soc Nephrol 2001; 12: 1410–1421.
Kitamura S, Yamasaki Y, Kinomura M, et al: Establishment and characterization of renal progenitor like cells from S3 segment of nephron in rat adult kidney. FASEB J 2005; 19: 1789–1797.
Kale S, Karihaloo A, Clark PR, Kashgarian M, Krause DS, Cantley LG : Bone marrow stem cells contribute to repair of the ischemically injured renal tubule. J Clin Invest 2003; 112: 42–49.
Morigi M, Imberti B, Zoja C, et al: Mesenchymal stem cells are renotropic helping to repair the kidney and improve function in acute renal failure. J Am Soc Nephrol 2004; 15: 1794–1804.
Togel F, Isaac J, Westenfelder C : Hematopoietic stem cell mobilization-associated granulocytosis severely worsens acute renal failure. J Am Soc Nephrol 2004; 15: 1261–1267.
Stokman G, Leemans JC, Claessen N, Weening JJ, Florquin S : Hematopoietic stem cell mobilization therapy accelerates recovery of renal function independent of stem cell contribution. J Am Soc Nephrol 2005; 16: 1684–1692.
Iwasaki M, Adachi Y, Minamino K, et al: Mobilization of bone marrow cells by G-CSF rescues mice from cisplatin-induced renal failure and M-CSF enhances the effects of G-CSF. J Am Soc Nephrol 2005; 16: 658–666.
Imanishi T, Kobayashi K, Hano T, Nishio I : Effect of estrogen on differentiation and senescence in endothelial progenitor cells derived from bone marrow in spontaneously hypertensive rats. Hypertens Res 2005; 28: 763–772.
Rogers SA, Lowell JA, Hammerman NA, Hammerman MR : Transplantation of developing metanephroi into adult rats. Kidney Int 1998; 54: 27–37.
Rogers SA, Liapis H, Hammerman MR : Transplantation of metanephroi across the major histocompatibility complex in rats. Am J Physiol Regul Integr Comp Physiol 2001: 280: R132–R136.
Rogers SA, Hammerman MR : Transplantation of rat metanephroi into mice. Am J Physiol Regul Integr Comp Physiol 2001; 280: R1865–R1869.
Rogers SA, Hammerman MR Transplantation of metanephroi after preservation in vitro. Am J Physiol Regul Integr Comp Physiol 2001; 281: R661–R665.
Yamamoto Y, Kohara K, Tabara Y, Igase M, Nakura J, Miki T : Plasma hepatocyte growth factor and the relationship between risk factors and carotid atherosclerosis. Hypertens Res 2002; 25: 661–667.
Schena FP : Role of growth factors in acute renal failure. Kidney Int Suppl 1998; 66: S11–S15.
Hishikawa K : Do we really need “stem” cell for renal regeneration? Kidney Int 2005; 68: 1966–1967.
Murry CE, Soonpaa MH, Reinecke H, et al: Haematopoietic stem cells do not transdifferentiate into cardiac myocytes in myocardial infarcts. Nature 2004; 428: 664–668.
Balsam LB, Wagers AJ, Christensen JL, Kofidis T, Weissman IL, Robbins RC : Haematopoietic stem cells adopt mature haematopoietic fates in ischaemic myocardium. Nature 2004; 428: 668–673.
Uchimura H, Marumo T, Takase O, et al: Intrarenal injection of bone marrow–derived angiogenic cells reduces endothelial injury and mesangial cell activation in experimental glomerulonephritis. J Am Soc Nephrol 2005; 16: 997–1004.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hishikawa, K., Fujita, T. Stem Cells and Kidney Disease. Hypertens Res 29, 745–749 (2006). https://doi.org/10.1291/hypres.29.745
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1291/hypres.29.745
Keywords
This article is cited by
-
Comparative effects of mesenchymal stem cell therapy in distinct stages of chronic renal failure
Clinical and Experimental Nephrology (2015)
-
Novel pharmacological approaches to the treatment of renal ischemia-reperfusion injury: a comprehensive review
Naunyn-Schmiedeberg's Archives of Pharmacology (2007)