Abstract
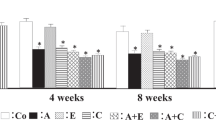
To confirm that α1, β adrenoceptor antagonists and angiotensin II type 1 receptor blockers (ARBs) have different abilities to attenuate progressive cardiac hypertrophy despite their comparable lowering of blood pressure, we compared the effect of these agents alone or in combination on hypertensive cardiac hypertrophy. Eight-week-old spontaneously hypertensive rats (SHR) were divided into 7 groups. Single administration of doxazosin, atenolol, or losartan, or half-dose combinations of these drugs were given orally for 6 weeks. The control group did not receive any drugs. The heart weight-to-body weight ratio (HW/BW), left ventricular mass index (LVMI), plasma brain natriuretic peptide (BNP) and left ventricular BNP mRNA expression were measured after 6-week administration. Blood pressure did not differ among the drug-treated groups, all of which showed lower blood pressure than the control group. The HW/BW and LVMI of the drug-treated groups, except the doxazosin group, were lower than in the control group. Moreover, the LVMI values of the groups receiving losartan were significantly lower than those in the groups without losartan (p<0.05). Plasma BNP of the drug-treated groups was lower than that in the control group (p<0.05). The left ventricular BNP mRNA expression of the drug-treated groups, except the doxazosin group, was lower than that in the control group. The atenolol group showed a higher level of BNP mRNA than the groups receiving losartan monotherapy or combination therapies (p<0.05). In conclusion, the ARB had the strongest attenuating effect on the development of hypertensive cardiac hypertrophy, and the α1 and β adrenergic receptor blockers were more effective in combination than as monotherapies in SHR.
Similar content being viewed by others
Article PDF
References
Levy D, Garrison RJ, Savage DD, Kannel WB, Castelli WP : Prognostic implications of echocardiographically determined left ventricular mass in Framingham heart study. N Engl J Med 1990; 322: 1561– 1566.
Komuro I, Yazaki Y : Control of cardiac gene expression by mechanical stress. Annu Rev Physiol 1993; 55: 55– 57.
Morgan HE, Baker KM : Cardiac hypertrophy. Mechanical, neural, and endocrine dependence. Circulation 1991; 83: 13– 25.
Yamazaki T, Komuro I, Zou Y, et al : Norepinephrine induced the raf-1 kinase/mitogen-activated protein kinase cascade through both α1- and β-adrenoceptors. Circulation 1997; 95: 1260– 1268.
Goldstein DS : Plasma catecholamines and essential hypertension. An analytical review. Hypertension 1983; 5: 86– 99.
Beker KM, Chernin MI, Wixson SK, Aceto JF : Renin-angiotensin system involvement in pressure-overload cardiac hypertrophy in rats. Am J Physiol Heart Circ Physiol 1990; 259: H324– H332.
Schunkert H, Dzau VJ, Tang SS, Hirsch AT, Apstein CS, Lorell BH : Increased rat cardiac angiotensin converting enzyme activity and mRNA expression in pressure overload left ventricular hypertrophy. Effects on coronary resistance, contractility, and relaxation. J Clin Invest 1990; 86: 1913– 1920.
Dang A, Zheng D, Wang B, et al : The role of renin-angiotensin and cardiac sympathetic nervous systems in the development of hypertension and left ventricular hypertrophy in spontaneously hypertensive rats. Hypertens Res 1999; 22: 217– 221.
Simpson P : Norepinephrine-stimulated hypertrophy of cultured rat myocardial cells is an alpha 1 adrenergic response. J Clin Invest 1983; 72: 732– 738.
Huang MH, Friend DS, Sunday ME, et al : An intrinsic adrenergic system in mammalian heart. J Clin Invest 1996; 98: 1298– 1303.
Crabos M, Roth M, Hahn AW, Erne P : Characterization of angiotensin II receptors in cultured adult rat cardiac fibroblasts. Coupling to signaling systems and gene expression. J Clin Invest 1994; 93: 2372– 2378.
Sano H, Okamoto H, Kitabatake A, Iizuka K, Murakami T, Kawaguchi H : Increased mRNA expression of cardiac renin-angiotensin system and collagen synthesis in spontaneously hypertensive rats. Mol Cell Biochem 1998; 178: 51– 58.
Griendling KK, Murphy TJ, Alexander RW : Molecular biology of the renin-angiotensin system. Circulation 1993; 87: 1816– 1828.
Arita M, Horinaka S, Komatsu K, Frohlich ED : Reversal of left ventricular hypertrophy with different classes of drugs causes differing ventricular biochemical changes. J Hypertens 1993; 11 ( Suppl 5): S354– S355.
Brilla CG : Regression of myocardial fibrosis in hypertensive heart disease: diverse effects of various antihypertensive drugs. Cardiovasc Res 2000; 46: 324– 331.
Varagic J, Susic D, Frohlich ED : Low-dose ACE with alpha- or beta-adrenergic receptor inhibitors have beneficial SHR cardiovascular effects. J Cardiovasc Pharmachol Ther 2001; 6: 57– 63.
Ohta K, Kim S, Iwao H : Role of angiotensin-converting enzyme, adrenergic receptors, and blood pressure in cardiac gene expression of spontaneously hypertensive rats during development. Hypertension 1996; 28: 627– 634.
Takeda T, Kohno M : Brain natriuretic peptide in hypertension. Hypertens Res 1995; 18: 259– 266.
Nakagawa O, Ogawa Y, Itoh H, et al : Rapid transcriptional activation and early mRNA turnover of brain natriuretic peptide in cardiocyte hypertrophy. Evidence for brain natriuretic peptide as an “emergency” cardiac hormone against ventricular overload. J Clin Invest 1995; 96: 1280– 1287.
Suzuki M, Hamada M, Yamamoto K, Kazatani Y, Hiwada K : Brain natriuretic peptide as a risk marker for incident hypertensive cardiovascular events. Hypertens Res 2002; 25: 669– 676.
Maeso R, Navarro-Cid J, Rodrigo E, Ruilope LM, Lahera V, Cachofeiro V : Differential effects of losartan and doxazosin on vascular function in senescent spontaneously hypertensive rats. Am J Hypertens 1999; 12: 1105– 1108.
Haneda T, Ogawa Y, Kato J, et al : Effect of celiprolol on cardiac hypertrophy in hypertension. Hypertens Res 2000; 23: 467– 474.
Devereux RB, Alonso DR, Lutas EM, et al : Echocardiographic assessment of left ventricular hypertrophy: comparison to necropsy findings. Am J Cardiol 1986; 57: 450– 458.
Brown LB, Nunez DJR, Wilkins MR : Differential regulation of natriuretic peptide receptor messenger RNAs during development of cardiac hypertrophy in the rat. J Clin Invest 1993; 92: 2702– 2712.
Terada Y, Tomita K, Nonoguchi H, Marumo F : Polymerase chain reaction localization of constitutive nitric oxide synthase and soluble guanylate cyclase messenger RNAs in microdissected rat nephron segments. J Clin Invest 1992; 90: 659– 665.
Ogawa Y, Nakao K, Mukoyama M, et al : Natriuretic peptides as cardiac hormones in normotensive and spontaneously hypertensive rats. The ventricle is a major site of synthesis and secretion of brain natriuretic peptide. Circ Res 1991; 69: 491– 500.
Mukoyama M, Nakao K, Hosoda K, et al : Brain natriuretic peptide as a novel cardiac hormone in humans. Evidence for an exquisite dual natriuretic peptide system, atrial natriuretic peptide and brain natriuretic peptide. J Clin Invest 1991; 87: 1402– 1412.
Magga J, Marttila M, Mantymaa P, Vuolteenaho O, Ruskoaho H : Brain natriuretic peptide in plasma, atria, and ventricles of vasopressin- and phenylephrine-infused conscious rats. Endocrinology 1994; 134: 2505– 2515.
Magga J, Kalliovalkama J, Romppanen H, et al : Differential regulation of cardiac adrenomedullin and natriuretic peptide gene expression by AT1 receptor antagonism and ACE inhibition in normotensive and hypertensive rats. J Hypertens 1999; 17: 1543– 1552.
Yokota N, Aburaya M, Yamamoto Y, et al : Increased plasma brain natriuretic peptide levels in DOCA-salt hypertensive rats: relation to blood pressure and cardiac concentration. Biochem Biophys Res Commun 1990; 173: 632– 638.
ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group : Diuretic versus α-blocker as first-step antihypertensive therapy: final results from the antihypertensive and lipid-lowering treatment to prevent heart attack trial (ALLHAT). Hypertension 2003; 42: 239– 246.
Dahlof B, Devereux RB, Kjeldsen SE, et al : Cardiovascular morbidity and mortality in the losartan intervention for endpoint reduction in hypertension study (LIFE): a randomised trial against atenolol. Lancet 2002; 359: 995– 1003.
Ohta K, Kim S, Iwao H : Role of angiotensin-converting enzyme, adrenergic receptors, and blood pressure in cardiac gene expression of spontaneously hypertensive rats during development. Hypertension 1996; 28: 627– 634.
Yamori Y, Tarazi RC, Ooshima A : Effect of beta-receptor-blocking agents on cardiovascular structural changes in spontaneous and noradrenaline-induced hypertension in rats. Clin Sci (Lond) 1980; 59 ( Suppl 6): 457s– 460s.
Ohlstein EH, Vickery L, Arleth A, et al : Carvedilol, a novel cardiovascular agent, inhibits development of vascular and ventricular hypertrophy in spontaneously hypertensive rats. Clin Exp Hypertens 1994; 16: 163– 177.
Lithell H, Aberg H, Selinus I : Metabolic effects of a change in antihypertensive treatment. Am J Med 1986; 80: 114– 119.
Bezerra SM, dos Santos CM, Moreira ED, Krieger EM, Michelini LC : Chronic AT1 receptor blockade alters autonomic balance and sympathetic responses in hypertension. Hypertension 2001; 38: 569– 575.
Pinto YM, Pinto-Sietsma SJ, Philipp T, et al : Reduction in left ventricular messenger RNA for transforming growth factor β1 attenuates left ventricular fibrosis and improves survival without lowering blood pressure in the hypertensive TGR(mRen2)27 rat. Hypertension 2000; 36: 747– 754.
Linz W, Schaper J, Wiemer G, Albus U, Scholkens BA : Ramipril prevents left ventricular hypertrophy with myocardial fibrosis without blood pressure reduction: a one year study in rats. Br J Pharmacol 1992; 107: 970– 975.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Asai, T., Kushiro, T., Fujita, H. et al. Different Effects on Inhibition of Cardiac Hypertrophy in Spontaneously Hypertensive Rats by Monotherapy and Combination Therapy of Adrenergic Receptor Antagonists and/or the Angiotensin II Type 1 Receptor Blocker under Comparable Blood Pressure Reduction. Hypertens Res 28, 79–87 (2005). https://doi.org/10.1291/hypres.28.79
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1291/hypres.28.79
Keywords
This article is cited by
-
Early regression of left ventricular hypertrophy after treatment with esmolol in an experimental rat model of primary hypertension
Hypertension Research (2013)
-
Is overall blockade superior to selective blockade of adrenergic receptor subtypes in suppressing left ventricular remodeling in spontaneously hypertensive rats?
Hypertension Research (2010)
-
Carvedilol inhibits right ventricular hypertrophy induced by chronic hypobaric hypoxia
Pflügers Archiv - European Journal of Physiology (2006)
-
Metabolic effects of antihypertensive agents: role of sympathoadrenal and renin-angiotensin systems
Naunyn-Schmiedeberg's Archives of Pharmacology (2006)