Abstract
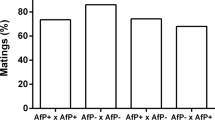
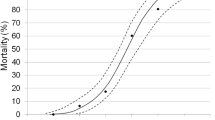
We investigated whether asexual (thelytokous) Apoanagyrus diversicornis and sexual (arrhenotokous) A. diversicornis (Hymenoptera: Encyrtidae) belong to the same biological species. The strains are allopatric and there are no morphological differences. The sexual A. diversicornis strain originates from south-western Brazil. It was introduced into Africa for the biological control of the cassava mealybug Phenacoccus manihoti. The asexual strain parasitizes P. herreni in Colombia. Feeding antibiotics to asexual A. diversicornis or rearing parasitized hosts at 33 instead of 25°C led to male offspring production. These males could be crossed with virgin females of the sexual strain, leading to fertile female (i.e. hybrid) offspring. There were no indications of hybrid breakdown. Thus, asexuality in A. diversicornis is caused by microorganisms, can be cured, and the two strains belong to the same biological species. The microorganisms associated with thelytoky in A. diversicornis belong to the genus Wolbachia. There were differences between A. diversicornis strains and between host species in survival probability from egg to adult. In addition, some mortality was associated with the feeding of tetracycline and with the production of males, but not with rifampicin, sulphadiazine (which was not effective), or a temperature increase from 25 to 33°C. Survival of hybrids was intermediate between the survival probabilities of the parental strains. Male behaviour and function were not different between the strains, but female behaviour clearly was. Females of the thelytokous strain were less often inseminated by males of either strain than were arrhenotokous females. This suggests a difference in the rate of decay between expressed traits (female sexual behaviour, which is possibly selected against) and traits that are not expressed (male sexual behaviour and function).
Similar content being viewed by others
Article PDF
References
Breeuwer, J A J, Stouthamer, R, Barns, S M, Pelletier, D A, Weisburg, W G, and Werren, J H. 1992. Phylogeny of cytoplasmic incompatibility micro-organisms in the parasitoid wasp genus Nasonia (Hymenoptera: Pteromalidae) based on 16S ribosomal DNA sequences. Insect Mol Biol, 1, 25–36.
Carson, H L, Chang, L S, and Lyttle, T R. 1982. Decay of female sexual behavior under parthenogenesis. Science, 218, 68–70.
Gordh, G, and Lacey, L. 1976. Biological studies of Plagiomerus diaspidis Crawford, a primary internal parasite of diaspidid scale insects (Hymenoptera: Encyrtidae; Homoptera: Diaspididae). Proc Entomol Soc Washington, 78, 132–144.
Haccou, P, and Meelis, E. 1992. Statistical Analysis of Behavioural Data. Oxford University Press, Oxford.
Herren, H R, and Neuenschwander, P. 1991. Biological control of cassava pests in Africa. Ann Rev Ent, 36, 257–283.
Hochberg, Y. 1988. A sharper Bonferroni procedure for multiple tests of significance. Biometrika, 75, 800–802.
Laraichi, M. 1978. L'effet de hautes temperatures sur le taux sexuel de Ooencyrtus fecundus Hymenoptera: Encyrtidae. Entomologia exp Appl, 23, 237–242.
Löhr, B, Varela, A M, and Santos, B. 1990. Exploration for natural enemies of the cassava mealybug, Phenacoccus manihoti (Homoptera: Pseudococcidae), in South America for the biological control of this introduced pest in Africa. Bull ent Res, 80, 417–425.
Noyes, J S, and Hayat, M. 1994. Oriental Mealybug Parasitoids of the Anagyrini (Hymenoptera: Encyrtidae). CAB International, Wallingford, UK.
O'Neill, S L, Giordano, R, Colbert, A M E, Karr, T L, and Robertson, H M. 1992. 16S rRNA phylogenetic analysis of the bacterial endosymbionts associated with cytoplasmic incompatibility in insects. Proc Natl Acad Sci USA, 89, 2699–2702.
Pijls, J W A M, and Van Alphen, J J M. 1996. On the coexistence of the cassava mealybug parasitoids Apoanagyrus diversicornis and A. lopezi (Hymenoptera: Encyrtidae) in their native South America. Bull ent Res, 86, (in press).
Rössler, Y, and Debach, P. 1972. The biosystematic relations between a thelytokous and an arrhenotokous form of Aphytis mytilaspidis (Le Baron) (Hymenoptera: Aphelinidae). I. The reproductive relations. Entomophaga, 17, 391–423.
Rössler, Y, and Debach, P. 1973. Genetic variability in a thelytokous form of Aphytis mytilaspidis (Le Baron) (Hymenoptera: Aphelinidae). Hilgardia, 42, 149–175.
Rousset, F, Bouchon, D, Pintureau, B, Juchault, P, and Solignac, M. 1992. Wolbachia endosymbionts responsible for various alterations of sexuality in arthropods. Proc R Soc B, 250, 91–98.
Simes, R J. 1986. An improved Bonferroni procedure for multiple tests of significance. Biometrika, 73, 751–754.
Stouthamer, R. 1990. Effectiveness of several antibiotics in reverting thelytoky to arrhenotoky in Trichogramma spp. Les Colloques de l'INRA, 56, 119–122.
Stouthamer, R, and Kazmer, D J. 1994. Cytogenetics of microbe-associated parthenogenesis and its consequences for gene flow in Trichogramma wasps. Heredity, 73, 317–327.
Stouthamer, R, Luck, R F, and Hamilton, W D. 1990a. Antibiotics cause parthenogenetic Trichogramma (Hymenoptera / Trichogrammatidae) to revert to sex. Proc Natl Acad Sci USA, 87, 2424–2427.
Stouthamer, R, Pinto, J D, Platner, G R, and Luck, R F. 1990b. Taxonomic status of thelytokous forms of Trichogramma (Hymenoptera: Trichogrammatidae). Ann Entomol Soc Am, 83, 475–481.
Stouthamer, R, Breeuwer, J A J, Luck, R F, and Werren, J H. 1993. Molecular identification of microorganisms associated with parthenogenesis. Nature, 361, 66–68.
Van Driesche, R G, Bellotti, A, Herrera, C J, and Castillo, J A. 1987. Host preferences of two encyrtid parasitoids for the Columbian Phenacoccus spp. of cassava mealybugs. Entomologia exp Appl, 43, 261–266.
Werren, J H, Zhang, W, and Guo, L R. 1995. Evolution and phylogeny of Wolbachia: reproductive parasites of arthropods. Proc R Soc B, 261, 55–71.
Yaseen, M. 1981. Report on surveys for cassava mealybugs (Phenacoccus spp.) and their natural enemies in South America (January-March 1981). Unpublished Report of the Commonwealth Institute of Biological Control, 7 pp.
Zchori-Fein, E, Roush, R T, and Hunter, M S. 1992. Male production induced by antibiotic treatment in Encarsia formosa (Hymenoptera: Aphelinidae), an asexual species. Experientia, 48, 102–105.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pijls, J., van Steenbergen, H. & van Alphen, J. Asexuality cured: the relations and differences between sexual and asexual Apoanagyrus diversicornis. Heredity 76, 506–513 (1996). https://doi.org/10.1038/hdy.1996.73
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1996.73
Keywords
This article is cited by
-
Co-occurrence of thelytokous and bisexual Trichogramma dendrolimi Matsumura (Hymenoptera: Trichogrammatidae) in a natural population
Scientific Reports (2019)
-
Genetics of decayed sexual traits in a parasitoid wasp with endosymbiont-induced asexuality
Heredity (2014)
-
Review of treatment methods to remove Wolbachia bacteria from arthropods
Symbiosis (2014)
-
Large variation in mitochondrial DNA of sexual and parthenogenetic Dahlica triquetrella (Lepidoptera: Psychidae) shows multiple origins of parthenogenesis
BMC Evolutionary Biology (2013)
-
Occasional males in parthenogenetic populations of Asobara japonica (Hymenoptera: Braconidae): low Wolbachia titer or incomplete coadaptation?
Heredity (2012)