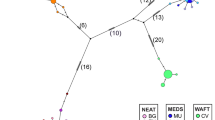
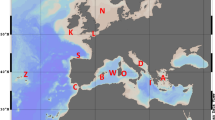
Abstract
The genetic relationships between the only natural population of Atlantic salmon (Salmo salar L.) in Denmark and seven other European salmon populations were studied using RFLP analysis of PCR amplified mitochondrial DNA segments. Six different haplotypes were detected by restriction enzyme analyses of the NADH dehydrogenase 1 segment, employing four endonucleases. Significant genetic differentiation was observed among populations. A hierarchical analysis of the distribution of the mtDNA variability revealed that only a small part was distributed among geographical groups within the study area. No correlation was found between genetic and geographic distance among populations. The effective migration of females (Nm )F among rivers was estimated to be approximately one per generation.
Similar content being viewed by others
Article PDF
References
Bermingham, E, Forbes, S H, Friedland, K, and Pla, C. 1991. Discrimination between Atlantic salmon (Salmo salar) of North American and European origin using restriction analysis of mitochondrial DNA. Can J Fish Aquat Sci, 48, 884–893.
Birky, C W, Jr., Maruyama, T, and Fuerst, P. 1983. An approach to population and evolutionary genetic theory for genes in mitochondra and chloroplasts, and some results. Genetics, 103, 513–527.
Billington, N, and Hebert, P D N. 1991. Mitochondrial DNA diversity in fishes and its implications for introductions. Can J Fish Aquat Sci, 48 (Suppl. 1), 80–93.
Blanco, G, Sánchez, J A, Vásquez, E, Rubio, J, and Utter, F M. 1992. Genetic differentiation among natural European populations of Atlantic salmon, Salmo salar L., from drainages of the Atlantic Ocean. Anim Genet, 23, 11–18.
Cronin, M A, Spearman, W J, Wilmot, R L, Patton, J C, and Bickham, J W. 1993. Mitochondrial DNA variation in Chinook (Oncorhynchus tshawytcha) and chum salmon (O. keta) detected by restriction enzyme analysis of polymerase chain reaction (PCR) products. Can J Fish Aquat Sci, 50, 708–715.
Crozier, W W. 1993. Evidence of genetic interaction between escaped farmed salmon and wild Atlantic salmon (Salmo salar L.) in a Northern Irish River. Aquaculture, 113, 19–29.
Davidson, W S, Birt, T P, and Green, J M. 1989. A review of genetic variation in Atlantic salmon, Salmo salar L., and its importance for stock identification, enhancement programmes and aquaculture. J Fish Biol, 34, 547–560.
Degnan, S M. 1993. The perils of single gene trees -mitochondrial vs. single copy nuclear DNA variation in white-eyes (Aves: Zosteropidae). Mol Ecol, 2, 219–225.
Elo, K. 1993. Gene flow and conservation of genetic variation in anadromous Atlantic salmon (Salmo salar). Hereditas, 119, 149–159.
Evans, D O, and Campbell, C W. 1991. Loss of exploited, indigenous populations of lake trout, Salvelinus namay-cush, by stocking of non-native stocks. Can J Fish Aquat Sci, 48 (Suppl. 1), 134–147.
Excoffier, L, Smouse, P E, and Quattro, J M. 1992. Analyses of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics, 131, 479–491.
Felsenstein, J. 1993. PHYLIP (Phylogeny Inference Package, Version 35c). Department of Genetics, SK, University of Washington, Seattle, WA.
Ferguson, M M, Danzmann, R G, and Hutchings, J A. 1991. Incongruent estimates of population differentiation among brook charr, Salvelinus fontinalis, from Cape Race, Newfoundland, Canada, based upon allozyme and mitochondrial DNA variation. J Fish Biol, 39 (Suppl. A), 79–85.
Fitch, W M, and Margoliash, E. 1967. Construction of phylogenetic trees. Science, 155, 279–284.
Flemming, I A, and Gross, M R. 1993. Breeding sucess of hatchery and wild coho salmon (Oncorhynchus kisutch) in competition. Ecol Appl, 3, 230–345.
Hansen, M M, and Loeschcke, V. 1996. Genetic differentiation among Danish brown trout (Salmo trutta) populations, as detected by RFLP analysis of PCR amplified mitochondrial DNA segments. J Fish Biol, 48, 422–436.
Hansen, M M, Hynes, R A, Loeschcke, V, and Rasmus-Sen, G. 1995. Assessment of the stocked or wild origin of anadromous brown trout (Salmo trutta L.) in a Danish river system, using mitochondrial DNA RFLP analysis. Mol Ecol, 4, 189–198.
Hindar, K, Ryman, N, and Utter, F. 1991. Genetic effects of cultured fish on natural fish populations. Can J Fish Aquat Sci, 48, 945–957.
Hovey, S J, King, D P F, Thompson, D, and Scott, A. 1989. Mitochondrial DNA and allozyme analysis of Atlantic salmon, Salmo salar., in England and Wales. J Fish Biol, 35 (Suppl. A), 253–260.
Hutchings, J A. 1991. The threat of extinction to native populations experiencing spawning intrusions by cultured Atlantic salmon. Aquaculture, 98, 119–132.
Hudson, R R, Slatkin, M, and Maddison, W P. 1992. Estimation of gene flow from DNA sequence data. Genetics, 132, 583–589.
Jordan, W C, Youngson, A F, Hay, D W, and Ferguson, A. 1992. Genetic protein variation in natural populations of Atlantic salmon (Salmo salar) in Scotland: temporal and spatial variation. Can J Fish Aquat Sci, 49, 1863–1872.
King, D P F, Hovey, S J, Thompson, D, and Scott, A. 1993. Mitochondrial DNA variation in Atlantic salmon, Salmo salar L., populations. J Fish Biol, 42, 25–33.
Knox, D, and Verspoor, E. 1991. A mitochondrial DNA restriction fragment length polymorphism of potential use for discrimination of farmed Norwegian and wild Atlantic salmon populations in Scotland. Aquaculture, 98, 249–257.
Kouonen, M L. 1989. Electrophoretically detectable genetic variation in natural and hatchery stocks of Atlantic salmon in Finland. Hereditas, 110, 23–35
McElroy, D, Moran, P, Bermingham, E, and Kornfield, I. 1991. REAP The Restriction Enzyme Analyses Package. Version 4.0. Department of Zoology, Migratory Fish Research Institute and Center for Marine Studies, University of Maine, Orono, USA.
Nei, M, and Li, W H. 1979. Mathematical models for studying genetic variation in terms of restriction endo-nucleases. Proc Natl Acad Sci USA, 76, 5269–5273.
Pallson, S, and Arnason, E. 1994. Sequence variation for cytochrome b genes of three salmonid species from Iceland. Aquaculture, 128, 29–39.
Palva, T K, Lehväslaiho, H, and Palva, E T. 1989. Identification of anadromous and non-anadromous salmon stocks in Finland by mitochondrial DNA analysis. Aquaculture, 81, 237–244.
Pamilo, P, and Nei, M. 1988. Relationship between gene trees and species trees. Mol Biol Evol, 5, 568–583.
Rice, W R. 1989. Analyzing tables of statistical tests. Evolution, 43, 223–225.
Roff, D A, and Bentzen, P. 1989. The statistical analyses of mitochondrial DNA polymorphisms: χ2 and the problem of small samples. Mol Biol Evol, 6, 539–545.
Ryman, N. 1983. Patterns of distribution of biochemical genetic variation in salmonids: differences between species. Aquaculture, 33, 1–21.
Ryman, N. 1991. Conservation genetics considerations in fishery management. J Fish Biol, 39 (Suppl. A), 211–224.
Schaffer, H E. 1983. Determination of DNA fragment size from gel electrophoresis mobility. In: Weir, B. S. (ed.) Statistical Analyses of DNA Sequence Data, pp. 1–14. Dekker, New York.
Siegismund, H R. 1993. G-Stat, ver 3 Genetical Statistical Programs for the Analysis of Population Data. The Arboretum, Royal Veterinary and Agricultural University, Kierkegaardsvej 3A, DK2970 Hoersholm, Denmark.
Slatkin, M. 1993. Isolation by distance in equilibrium and non-equilibrium populations. Evolution, 47, 264–279.
Ståhl, G. 1981. Genetic differentiation among natural populations of Atlantic salmon (Salmo salar) in Northern Sweden. In: Ryman, N. (ed.) Fish Gene Pools Ecol Bull, 34, 95–105.
Ståhl, G. 1987. Genetic population structure of Atlantic salmon. In: Ryman, N. and Utter, F. (eds) Population Genetics and Fishery Management, pp. 121–140. University of Washington Press, Seattle.
Taggart, J B, Hynes, R A, Prodöhl, P A, and Ferguson, A. 1993. A simplified protocol for routine total DNA isolation from salmonid fishes. J Fish Biol, 40, 963–965.
Thompson, D, and Russell, I C. 1991. Allele frequency variation at the s AAT-4 locus as a potential measure of the relative performance of native and introduced Atlantic salmon in the river Test. Aquaculture, 98, 243–247.
Vasquez, E, Presa, P, Sánchez, J A, Blanco, G, and Utter, F. 1993. Genetic characterization of introduced populations of Atlantic salmon, Salmo salar, in Asturias (Northern Spain). Hereditas, 119, 47–51.
Wright, S. 1931. Evolution in Mendelian populations. Genetics, 16, 97–159.
Acknowledgements
Acknowledgements We are grateful to Bob Krebs and Philip W. Hedrick for helpful comments on the manuscript and to all those who provided us with salmon samples: S0ren Larsen and J0rgen J0rgensen helped us to obtain samples from the Skjern river. Gert Holdensgaard, John Taggart, Kjetil Hindar, Sna3bj0rn Pallson and Einar Arnasson kindly provided us with Atlantic salmon samples. We would like to thank the Danish Inland Fisheries Laboratory and the Danish National Research Council (grant no. 94-0163-1) for financial support.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nielsen, E., Hansen, M. & Loeschcke, V. Genetic structure of European populations of Salmo salar L. (Atlantic salmon) inferred from mitochondrial DNA. Heredity 77, 351–358 (1996). https://doi.org/10.1038/hdy.1996.153
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1996.153
Keywords
This article is cited by
-
Use of multiple markers demonstrates a cryptic western refugium and postglacial colonisation routes of Atlantic salmon (Salmo salar L.) in northwest Europe
Heredity (2013)
-
Introgression and genetic structure in northern Spanish Atlantic salmon (Salmo salar L.) populations according to mtDNA data
Conservation Genetics (2008)
-
Stocking may increase mitochondrial DNA diversity but fails to halt the decline of endangered Atlantic salmon populations
Conservation Genetics (2007)
-
Extensive immigration from compensatory hatchery releases into wild Atlantic salmon population in the Baltic sea: spatio-temporal analysis over 18 years
Heredity (2005)