Abstract
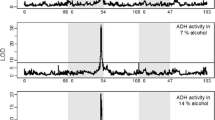
Biochemical properties and expression patterns of alcohol dehydrogenase were compared among members of the virilis phylad of Drosophila. Quantitative differences in stage- and tissue-specific expression of ADH of D. virilis, D. novamexicana, D. texana and D. americana were observed. These differences seem to correlate with the temporal differences in ethanol tolerance among these species. Quantitative differences in ADH protein levels account for most, if not all, of the activity differences between species. Analysis of the interspecific hybrids revealed that these differences arise by a mechanism affecting monomer synthesis. A comparison with earlier studies, where independently evolved regulatory differences were related to adaptations, revealed contrasting underlying mechanisms.
Similar content being viewed by others
Article PDF
References
Choo, J K, and Oshima, C. 1974. Phototactic selection and its effect on some quantitative characters of Drosophila virilis. Environ Control Biol, 12, 41–51.
Crawford, D L, and Powers, D A. 1989. Molecular basis of evolutionary adaptation at the lactic dehydrogenase-B locus in the fish Fundulus heteroclitus. Proc Natl Acad Sci USA, 86, 9365–9369.
David. J R. 1988. Ethanol adaptation and alcohol dehydrogenase polymorphism in Drosophila: from phenotypic functions to genetic structures. In: de Jong, G. (ed.) Population Genetics and Evolution, pp. 163–172. Springer-Verlag, Berlin.
David, J R, and Bocquet, C. 1977. Genetic tolerance to ethanol in Drosophila melanogaster, increase by selection and analysis of correlated responses. Genetica, 47, 43–48.
David, J R, and Van Herrewege, J. 1983. Adaptation to alcoholic fermentation in Drosophila species: relationship between alcohol tolerance and larval habitat. Comp Biochem Physiol, 74A, 283–288.
David, J R, and Kitagawa, O. 1982. Possible similarities in ethanol tolerance and latitudinal variations between Drosophila virilis and Drosophila melanogaster. Jpn J Genet, 57, 89–95.
Dickinson, W J. 1980. Complex cis-acting regulatory genes demonstrated in Drosophila hybrids. Dev Genet, 1, 229–240.
Dickinson, W J. 1991. The evolution of regulatory genes and patterns in Drosophila. Evol Biol, 25, 127–173.
Dickinson, W J, Rowan, R G, and Brennan, M D. 1984. Regulatory gene evolution: adaptive differences in expression of alcohol dehydrogenase in Drosophila melanogaster and Drosophila simulans. Heredity, 52, 215–225.
Dobson, D E, Prager, E M, and Wilson, A C. 1984. Stomach lysozymes of ruminants. I. J Biol Chem, 259, 11607–11616.
Fahey, J, and McKelvey, E. 1965. Quantitative determination of serum immunoglobulins in antibody agar plates. J Immunol, 94, 84–90.
Ganschow, R, and Schimke, R T. 1969. Independent genetic control of the catalytic activity and the rate of degradation of catalase in mice. J Biol Chem, 244, 4649–4658.
Irwin, D M, Prager, E M, and Wilson, A C. 1992. Evolutionary genetics of ruminant lysozymes. An Genet, 23, 193–202.
Irwin, D M, Sidow, A, White, R T, and Wilson, A C. 1989. Multiple genes for ruminant lysozymes. In: Smith-Gill, S. J. and Sercarz, E. E. (eds) The Immune Response to Structurally Defined Proteins: The Lysozyme Model, pp. 73–85. Adenine Press, New York.
Jessen, T, Weber, R E, Fermi, G, Tame, J, and Braunitzer, G. 1991. Adaptation of bird haemoglobins to high altitudes: demonstration of molecular mechanism by protein engineering. Proc Natl Acad Sci USA, 88, 6519–6522.
Juan, E, and Gonzalez-Duarte, R. 1981. Determination of some biochemical and structural features of alcohol dehydrogenase from D. melanogaster, D. simulans and D. virilis. Biochem J, 195, 61–69.
Kerver, J W M, and Rotman, G. 1987. Development of ethanol tolerance in relation to the alcohol dehydrogenase locus in Drosophila melanogaster. II. The influence of phenotypic adaptation and maternal effect on survival on alcohol supplemented media. Heredity, 58, 239–248.
Kerver, J W M, and Van Delden, W. 1985. Development of tolerance to ethanol in relation to the alcohol dehydrogenase locus in Drosophila melanogaster. I. Adult and egg-to-adult survival in relation to ADH activity. Heredity, 55, 355–367.
King, J J, and McDonald, J F. 1983. Genetic localization and biochemical characterization of a irans-acting regulatory effect in Drosophila. Genetics, 105, 55–69.
Laemmli, U K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 277, 680–685.
Lai, Y K, and Scandalios, J G. 1980. Genetic determination of the developmental program for maize scutellar alcohol dehydrogenase: involvement of a recessive trans-acting temporal regulatory gene. Dev Genet, 1, 311–324.
Laurie, C C, Heath, E M, Jacobson, J W, and Thomson, M S. 1990. Genetic basis of the difference in alcohol dehydrogenase expression between Drosophila melanogaster and Drosophila simulans. Proc Natl Acad Sci USA, 87, 9674–9678.
Laurie-Ahlberg, C C, Maroni, G, Bewley, G G, Lucchesi, J G, and Weir, B S. 1980. Quantitative genetic variation of enzyme activities in natural populations of Drosophila melanogaster. Proc Natl Acad Sci USA, 77, 1073–1077.
Laurie-Ahlberg, G G, Wilton, A N, Curtsinger, J W, and Emigh, T H. 1982. Naturally occuring enzyme activity variation in Drosophila melanogaster I. Sources of variation for 23 enzymes. Genetics, 102, 191–206.
Lineweaver, H, and Burk, D. 1934. The determination of enzyme dissociation constants. J Am Chem Soc, 56, 658–666.
McDonald, J F. 1983. The molecular basis of adaptation: a critical review of relevant ideas and observations. Ann Rev Ecol Syst, 14, 77–102.
McDonald, J F, and Avise, J C. 1976. Evidence for the adaptive significance of enzyme activity levels. Interspecific activity levels of GPDH and ADH in Drosophila. Biochem Genet, 14, 347–355.
McKenzie, J A, and Parsons, P A. 1972. Alcohol tolerance: an ecological parameter in the reletive success of Drosophila melanogaster and Drosophila simulans. Oecologia, 10, 373–388.
Mancini, G, Carbonara, A O, and Heremans, J F. 1965. Immunochemical quantification of antigens by single radial immunodiffusion. Immunochemistry, 2, 235–254.
Ranganayakulu, G. 1990. Regulation of Tissue Specific Expression of Alcohol Dehydrogenase in the virilis Group of Drosophila. Ph.D Thesis, University of Hyderabad, India.
Ranganayakulu, G, Kirkpatrick, R B, Martin, P F, and Reddy, A R. 1991. Species specific differences in tissue specific expression of alcohol dehydrogenase are under the control of complex cis-acting loci: evidence from Drosophila hybrids. Biochem Genet, 29, 577–592.
Schwartz, M, Gerace, L, O'Donnell, J M, and Sofer, W. 1975. Drosophila alcohol dehydrogenase: origin of the multiple forms. In: Market, C. L. (ed.) Isozymes, 1, pp. 725–751. Academic Press, New York.
Sofer, W, and Ursprung, H. 1968. Drosophila alcohol dehydrogenase: purification and partial characterization. J Biol Chem, 243, 3110–3115.
Spieth, H T. 1979. The virilis group of Drosophila and the beaver castor. Am Nat, 114, 312–316.
Stewart, C B, Schilling, J W, and Wilson, A C. 1987. Adaptive evolution in the stomach lysozymes of foregut fermenters. Nature, 330, 401–404.
Throckmorton, L H. 1975. The phylogeny, ecology, and geography of Drosophila. In: King, R. C. (ed.) Handbook of Genetics, 3, pp. 421–469. Plenum Press, New York.
Weber, K, Pringle, J R, and Osborne, M. 1972. Measurement of molecular weights by electrophoresis on SDS-acrylamide gels. Methods EnzymoL, 26, 3–27.
Wilson, A C, Carlson, S S, and White, T J. 1977. Biochemical evolution. Ann Rev Biochem, 46, 473–639.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ranganayakulu, G., Reddy, A. Regulatory differences in developmental expression of alcohol dehydrogenase are related to interspecies differences in ethanol tolerance of Drosophila. Heredity 72, 374–383 (1994). https://doi.org/10.1038/hdy.1994.54
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1994.54