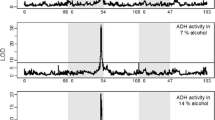
Abstract
Allele specific oligonucleotide probes have been used to show that DNA, amplified by the polymerase chain reaction, from eleven thermostable Adh alleles extracted from populations on different continents contains the triplet TCC, which distinguishes AdhFChD (Fast Chateau Douglas) from AdhF (Fast). The molecular similarity of Adh thermostable alleles suggests that they had a common origin, and it is argued that the mutation probably occurred in China where high frequencies of AdhFChD are found.
Similar content being viewed by others
Article PDF
References
Ashburner, M, Bodmer, M, and Lemeunier, F. 1984. On the evolutionary relationships of Drosophila melanogaster. Develop Genet, 4, 295–312.
Bodmer, M, and Ashburner, M. 1984. Conservation and change in the DNA sequences coding for alcohol de-hydrogenase in sibling species of Drosophila. Nature, 309, 425–430.
Chambers, G K, Laver, W G, Campbell, S, and Gibson, J B. 1981a. Structural analysis of an electrophoretically cryptic alcohol dehydrogenase variant from an Australian population of Drosophila melanogaster. Proc Natl Acad Sci USA, 78, 3103–3107.
Chambers, G K, Wilks, A V, and Gibson, J B. 1981b. An electrophoretically cryptic alcohol dehydrogenase variant in Drosophila melanogaster. III. Biochemical properties and comparison to the common enzyme forms. Aust J Biol Sci, 34, 625–637.
Chia, W, Savakis, C, Karp, R, Pelham, H, and Ashburner, M. 1985. Mutation of the Adh gene of Drosophila melanogaster containing an internal tandem duplication. J Mol Biol, 186, 679–688.
Collet, C. 1988. Recent origin for a thermostable alcohol dehydrogenase allele of Drosophila melanogaster. J Mol Evol, 27, 142–146.
Conner, B J, Reyes, A A, Morin, C, Itakura, K, Teplitz, R L, and Bruce Wallace, R. 1983. Detection of sickle cell βs-globin allele by hybridization with synthetic oligonucleotides. Proc Natl Acad Sci USA, 80 278–282.
Gibson, J B, Anderson, D G, and Wilks, A V. 1982a. Electrophoretic variability at the alcohol dehydrogenase locus in Drosophila melanogaster. Genetics, 101, 331–333.
Gibson, J B, Chambers, G K, Wilks, A V, and Oake-Shott, J G. 1980. An electrophoretically cryptic alcohol dehydrogenase variant in Drosophila melanogaster. I. Activity ratio, genetic localization and comparison with two other thermostable variants. Aust J Biol Sci, 33, 479–489.
Gibson, J B, Wilks, A V, and Chambers, G K. 1982b. Genetic variation at the alcohol dehydrogenase locus in Drosophila melanogaster: A third ubiquitous allele. Experientia. 38, 653–654.
Hernández, M, PadrÖn, G, and Cabrera, V M. 1986. Persistence of an alcohol dehydrogenase thermostable variant in a natural population of Drosophila melanogaster. Genet Res Camb, 47, 143–146.
Jiang, C, Gibson, J B, and Chen, H. 1989. Genetic differentiation in populations of Drosophila melanogaster from the Peoples' Republic of China: comparison with patterns on other continents. Heredity, 62 193–198.
Kidd, V J, Bruce Wallace, R, Itakura, K, and Woo, S L C. 1983. α1 Antitrypsin deficiency detection by direct analysis of the mutation in the gene. Nature, 304, 230–234.
Kreitman, M. 1983. Nucleotide polymorphism at the alcohol dehydrogenase locus of Drosophila melanogaster. Nature, 304, 412–417.
Lachaise, D, Cariou, M, David, J R, Lemeunier, F, Tsacas, L, and Ashburner, M. 1988. Historical bio-geography of the Drosophila melanogaster species subgroup. Evol Biol, 22 159–225.
Lemeunier, F, David, J R, Tsacas, L, and Ashburner, M. 1985. The Drosophila melanogaster species group. In Ashburner, M., Carson, H. L. and Thompson, J. M. (eds) The Genetics and Biology of Drosophila, Academic Press, London.
Lewis, N, and Gibson, J B. 1978. Enzyme protein amount variation in natural populations. Biochem Genet, 16, 159–170.
Milkman, R. 1976. Further evidence of thermostability variation within electrophoretic mobility classes of enzymes. Biochem Genet, 14, 383–387.
Oakeshott, J G, Gibson, J B, Anderson, P R, Knibb, W R, Anderson, D G, and Chambers, G K. 1982. Alcohol dehydrogenase and glycerol-3-phosphate de-hydrogenase clines in Drosophila melanogaster on different continents. Evolution, 36, 86–96.
Saiki, R K, Bugawan, T L, Horn, G T, Mullis, K B, and Erlich, H A. 1986. Analyses of amplified β-globin and HLA-DQα DNA with allele-specific oligonucleotide probes. Nature, 324, 163–166.
Saiki, R K, Gelfand, D H, Stoffel, S, Scharf, S J, Higuchi, R, Horn, G T, Mullis, K B, and Erlich, H A. 1988. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science, 239, 487–491.
Sampsell, B. 1977. Isolation and genetic characterization of alcohol dehydrogenase thermostability variants occurring in natural populations of Drosophila melanogaster. Biochem Genet, 15 971–988.
Sampsell, B, and Steward, E. 1983. Alcohol dehydrogenase thermostability variants in Drosophila melanogaster: Comparison of activity ratios and enzyme levels. Biochem Genet, 21, 1071–1087.
Singh, R S, Hickey, D A, and David, J R. 1982. Genetic differentiation between geographically distant populations of Drosophila melanogaster. Genetics, 101, 235–256.
Stephens, J C, and Nei, M. 1985. Phylogenetic analysis of polymorphic DNA sequences at the Adh locus of Drosophila melanogaster and its sibling species. J Mol Evol, 22, 289–300.
Sturtevant, A H. 1920. Genetic studies on Drosophila simulans. I. Introduction. Hybrids with D. melanogaster. Genetics, 5, 488–500.
Thatcher, D R. 1980. The complete amino acid sequence of three alcohol dehydrogenase alleloenzymes (Adhn11, Adhs and AdhUF) from the fruitfly Drosophila melanogaster. Biochem J, 187, 875–883.
Thörig, G E W, Schoone, A A, and Scharloo, W. 1975. Variation between electrophoretically identical alleles at the alcohol dehydrogenase locus in Drosophila melanogaster. Biochem Genet, 13, 721–731.
Vigue, C L, and Johnson, F M. 1973. Isozyme variability in species of the genus Drosophila. VI. Frequency-property-environment relationships of allelic alcohol dehydrogenase in D melanogaster. Biochem Genet, 9, 213–227.
Wallace, R B, Schöld, M, Johnson, M J, Dembek, P, and Itakura, K. 1981. Oligonucleotide directed mutagenesis of the human βx-globin gene: a general method for producing specific point mutations in cloned DNA. Nuc Acid Res, 9, 3647–3656.
Wilks, A V, Gibson, J B, Oakeshott, J G, and Chambers, G K. 1980. An electrophoretically cryptic alcohol de-hydrogenase variant in Drosophila melanogaster. II. Post-electrophoresis heat treatment screening of natural populations. Aust J Biol Sci, 33, 575–585.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gibson, J., Reed, D., Wilks, A. et al. Molecular similarity of Drosophila melanogaster alcohol dehydrogenase thermostable alleles from populations on different continents. Heredity 65, 1–6 (1990). https://doi.org/10.1038/hdy.1990.62
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1990.62
This article is cited by
-
Substrate and inhibitor specificities of the thermostable alcohol dehydrogenase allozymes ADH-71k and ADH-FCh.D. ofDrosophila melanogaster
Biochemical Genetics (1994)
-
The alcohol dehydrogenase polymorphism in natural populations of Drosophila melanogaster: ADH activity variation restriction site polymorphism and the Adh cline
Heredity (1992)