Abstract
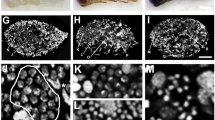
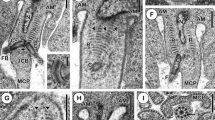
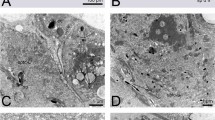
Frequency and position of chiasmata at metaphase I were studied in spermatocytes and oocytes from six species of gomphocerine grasshoppers (2n♂ = 16 + XO; 2n♀ = 16 + XX), namely: Omocestus panteli, Euchorthippus pulvinatus, Euchorthippus chopardi, Chorthippus vagans, Chorthippus parallelus and Chorthippus jucundus. In the latter species mean cell chiasma frequency was similar in both sexes. However in the remainder chiasma frequencies were higher in males than in females. Within each species different bivalents are responsible for the sex chiasma differences that can be interpreted as a relatively simple bivalent based system of chiasma control. Chiasmata were non-localised in both sexes of all species analysed but females showed altered chiasma distributions compared with males, with fewer proximal chiasmata and more interstitial and distal chiasmata.
Similar content being viewed by others
Article PDF
References
Bennett, M D, Finch, R A, Smith, J B, and Rao, M K. (1973). The time and duration of female meiosis in rye, wheat and barley. Proceedings of the Royal Society, London B 183, 301–319.
Bennett, J J H, Hayman, D L, and Hope, R M. (1986). Novel sex differences in linkage values and meiotic chromosome behaviour in a marsupial. Nature, 323, 59–60.
Callan, H G, and Perry, P E. (1977). Recombination in male and female meiocytes contrasted. Phil Trans Roy Soc London Series B, 277, 227–233.
Cano, M I, Jones, G H, and Santos, J L. (1987). Se-x differen-ces in chiasma frequency and distribution in natural popu-lations of Eyprepocnemis plorans containing B chromo-somes. Herediy, 59, 237–243.
Cano, M I, and Santos, J L. (1988). B chromosomes of the grasshopper Heteracris littoralis: meiotic behaviour and endophenotypic effects in both sexes. Genome, 30, 797–801.
Church, H K. (1974). Meiosis in the grasshopper. III. Chiasma frequency in females after elevated temperature. Heredity, 32, 159–164.
Davies, E D G, and Jones, G H. (1974). Chiasma variation and control in pollen mother cells and embryo-sac mother cells of rye. Genet Res, 23, 185–190.
Dünn, L C, and Bennett, D. (1967). Sex differences in re-combination of linked genes in animals. Genet Res, 9, 211–220.
Fletcher, H L, and Hewitt, G M. (1980). A comparison of chiasma frequency and distribution between sexes in three species of grasshoppers. Chromosoma, 77, 129–144.
Fogwill, M. (1958). Differences in crossing-over and chromo-some size in the sex cells of Lilium and Fritillaria. Chromosoma, 2, 493–504.
Gohil, R N, and Kaul, R. (1980). Studies on males and females meiosis in Indian Allium I Four diploid species. Chromosoma, 77, 123–127.
Haldane, J B S. (1922). Sex ratio and unisexual sterility in hybrid animal. J Genet, 12, 101–109.
Henriques-Gil, N, Jones, G H, Cano, M I, Arana, P, and Santos, J L. (1986). Female meiosis during oocyte matur-ation in Eyprepocnemis plorans (Othoptera, Acrididae). Can J Genet Cytol, 28, 84–87.
Hewitt, G M. (1976). Meiotic drive for B-chromosomes in the primary oocytes of Myrmeleotettix maculatus (Orthoptera: Acrididae). Chromosoma, 56, 381–391.
Hewitt, G M, and John, B. (1965). The influence of numerical and structural chromosome mutations on chiasma condi-tions. Heredity, 20, 123–135.
Jones, G H, Stamford, W K, and Perry, P E. (1975) Male and female meiosis in grasshoppers II Chorthippus brun- neus. Chromosoma, 51, 381–389.
Parker, J S. (1975). Chromosome-specific control of chiasma formation. Chromosoma, 49, 391–406.
Pastor, J B, Callan, H G.(1952). Chiasma formation in spermatocytes and oocytes of the turbellarian Den- drocoelum lacteum. J Genet, 50, 449–454.
Perry, P E, and Jones, G H. (1974). Male and female meiosis in grasshoppers I Stethophyma grossum. Chromosoma, 47, 227–236.
Rhoades, M M. (1978). Genetic effects of heterochromatin in maize. In Waiden, D. B. (ed.) Maize Breeding and Genetics , pp.641–671.
Santos, J L, Cipres, G, and Lacadena, J R. (1989). A quantitative study of chiasma terminalization in the grass-hopper Chorthippus jucundus, Heredity, 62, 51–57.
Southern, D I. (1967). Chiasma distribution in truxaline grasshoppers. Chromosoma, 22, 164–191.
Speed, R M. (1977). The effects of ageing on the meiotic chromosomes of male and female mice. Chromosoma, 64, 241–254.
Tease, C, and Jones, G H. (1976). Chromosome specific con-trol of chiasma formation in Crépis capillaris. Chromosoma, 57, 33–49.
Vosa, C G. (1972). Two track heredity: differentiation of male and female meiosis in Tulbaghia Caryologia, 25, 275–281.
Watson, I D, and Callan, H G. (1963). The form of bivalent chromosomes in newt oocytes at first metaphase of meiosis. Quart J Microsc Set, 104, 281–295.
White, M J D. (1973).Animal Cytology and Evolution, 3rd ed.Cambridge University Press, London.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cano, M., Santos, J. Chiasma frequencies and distributions in gomphocerine grasshoppers: a comparative study between sexes. Heredity 64, 17–23 (1990). https://doi.org/10.1038/hdy.1990.3
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1990.3
This article is cited by
-
Occurrence of differential meiotic associations and additional chromosomes in the embryo-sac mother cells of Allium roylei Stearn
Journal of Genetics (2011)
-
Sex differences in recombination and mapping adaptations
Genetica (2005)
-
Sex-dependent synaptic behaviour in triploid turbot, Scophthalmus maximus (Pisces, Scophthalmidae)
Heredity (2002)
-
Chiasma localization, heterochromatin and synaptonemal complexes in the grasshopperPyrgomorpha conica
Chromosome Research (1996)
-
Proximal chiasmata induce non-disjunctional orientation of Robertsonian trivalents in a grasshopper
Heredity (1992)