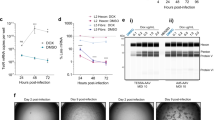
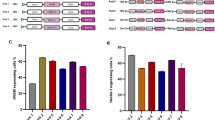
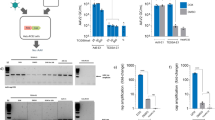
Abstract
Helper-dependent adenovirus vectors (HDVs) are safe and efficient tools for gene transfer with high cloning capacity. However, the multiple amplification steps needed to produce HDVs hamper a robust production process and in turn the availability of high-quality vectors. To understand the factors behind the low productivity, we analyzed the progression of HDV life cycle. Canine adenovirus (Ad) type 2 vectors, holding attractive features to overcome immunogenic concerns and treat neurobiological disorders, were the focus of this work. When compared with E1-deleted (ΔE1) vectors, we found a faster helper genome replication during HDV production. This was consistent with an upregulation of the Ad polymerase and pre-terminal protein and led to higher and earlier expression of structural proteins. Although genome packaging occurred similarly to ΔE1 vectors, more immature capsids were obtained during HDV production, which led to a ~4-fold increase in physical-to-infectious particles ratio. The higher viral protein content in HDV-producing cells was also consistent with an increased activation of autophagy and cell death, in which earlier cell death compromised volumetric productivity. The increased empty capsids and earlier cell death found in HDV production may partially contribute to the lower vector infectivity. However, an HDV-specific factor responsible for a defective maturation process should be also involved to fully explain the low infectious titers. This study showed how a deregulated Ad cycle progression affected cell line homeostasis and HDV propagation, highlighting the impact of vector genome design on virus–cell interaction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Lowenstein PR, Mandel RJ, Xiong WD, Kroeger K, Castro MG . Immune responses to adenovirus and adeno-associated vectors used for gene therapy of brain diseases: the role of immunological synapses in understanding the cell biology of neuroimmune interactions. Curr Gene Ther 2007; 7: 347–360.
Kremer EJ, Boutin S, Chillon M, Danos O . Canine adenovirus vectors: an alternative for adenovirus-mediated gene transfer. J Virol 2000; 74: 505–512.
Perreau M, Kremer EJ . Frequency, proliferation, and activation of human memory T cells induced by a nonhuman adenovirus. J Virol 2005; 79: 14595–14605.
Keriel A, Rene C, Galer C, Zabner J, Kremer EJ . Canine adenovirus vectors for lung-directed gene transfer: efficacy, immune response, and duration of transgene expression using helper-dependent vectors. J Virol 2006; 80: 1487–1496.
Perreau M, Mennechet F, Serratrice N, Glasgow JN, Curiel DT, Wodrich H et al. Contrasting effects of human, canine, and hybrid adenovirus vectors on the phenotypical and functional maturation of human dendritic cells: implications for clinical efficacy. J Virol 2007; 81: 3272–3284.
Soudais C, Laplace-Builhe C, Kissa K, Kremer EJ . Preferential transduction of neurons by canine adenovirus vectors and their efficient retrograde transport in vivo. FASEB J 2001; 15: 2283–2285.
Soudais C, Skander N, Kremer EJ . Long-term in vivo transduction of neurons throughout the rat CNS using novel helper-dependent CAV-2 vectors. FASEB J 2004; 18: 391–393.
Cubizolle A, Serratrice N, Skander N, Colle MA, Ibanes S, Gennetier A et al. Corrective GUSB transfer to the canine Mucopolysaccharidosis VII brain. Mol Ther 2014; 22: 762–773.
Ariza L, Gimenez-Llort L, Cubizolle A, Pages G, Garcia-Lareu B, Serratrice N et al. Central nervous system delivery of helper-dependent canine adenovirus corrects neuropathology and behavior in Mucopolysaccharidosis type VII mice. Hum Gene Ther 2014; 25: 199–211.
Dormond E, Perrier M, Kamen A . From the first to the third generation adenoviral vector: what parameters are governing the production yield? Biotechnol Adv 2009; 27: 133–144.
Ng P, Beauchamp C, Evelegh C, Parks R, Graham FL . Development of a FLP/frt system for generating helper-dependent adenoviral vectors. Mol Ther 2001; 3: 809–815.
Umana P, Gerdes CA, Stone D, Davis JR, Ward D, Castro MG et al. Efficient FLPe recombinase enables scalable production of helper-dependent adenoviral vectors with negligible helper-virus contamination. Nat Biotechnol 2001; 19: 582–585.
Parks RJ, Chen L, Anton M, Sankar U, Rudnicki MA, Graham FL . A helper-dependent adenovirus vector system: removal of helper virus by Cre-mediated excision of the viral packaging signal. Proc Natl Acad Sci USA 1996; 93: 13565–13570.
Gonzalez-Aparicio M, Mauleon I, Alzuguren P, Bunuales M, Gonzalez-Aseguinolaza G, San Martin C et al. Self-inactivating helper virus for the production of high-capacity adenoviral vectors. Gene Therapy 2011; 18: 1025–1033.
Alba R, Hearing P, Bosch A, Chillon M . Differential amplification of adenovirus vectors by flanking the packaging signal with attB/attP-PhiC31 sequences: implications for helper-dependent adenovirus production. Virology 2007; 367: 51–58.
Soudais C, Boutin S, Kremer EJ . Characterization of cis-acting sequences involved in canine adenovirus packaging. Mol Ther 2001; 3: 631–640.
Kanegae Y, Ishimura M, Kondo S, Saito I . Influence of loxP insertion upstream of the cis-acting packaging domain on adenovirus packaging efficiency. Microbiol Immunol 2012; 56: 447–455.
Hartigan-O'Connor D, Amalfitano A, Chamberlain JS . Improved production of gutted adenovirus in cells expressing adenovirus preterminal protein and DNA polymerase. J Virol 1999; 73: 7835–7841.
Dormond E, Meneses-Acosta A, Jacob D, Durocher Y, Gilbert R, Perrier M et al. An efficient and scalable process for helper-dependent adenoviral vector production using polyethylenimine-adenofection. Biotechnol Bioeng 2009; 102: 800–810.
Ibanes S, Kremer EJ . Canine adenovirus type 2 vector generation via I-Sce1-mediated intracellular genome release. PLoS One 2013; 8: e71032.
Palmer D, Ng P . Improved system for helper-dependent adenoviral vector production. Mol Ther 2003; 8: 846–852.
Suzuki M, Cela R, Clarke C, Bertin TK, Mourino S, Lee B . Large-scale production of high-quality helper-dependent adenoviral vectors using adherent cells in cell factories. Hum Gene Ther 2010; 21: 120–126.
Kreppel F, Biermann V, Kochanek S, Schiedner G . A DNA-based method to assay total and infectious particle contents and helper virus contamination in high-capacity adenoviral vector preparations. Hum Gene Ther 2002; 13: 1151–1156.
Flint SJ, Enquist LW, Racaniello VR, Skalka AM . Principles of Virology: Molecular Biology, Pathogenesis, and Control of Animal Viruses, 2nd edn. ASM Press: Washington, DC, 2004.
Greber UF, Suomalainen M, Stidwill RP, Boucke K, Ebersold MW, Helenius A . The role of the nuclear pore complex in adenovirus DNA entry. EMBO J 1997; 16: 5998–6007.
Perez-Berna AJ, Ortega-Esteban A, Menendez-Conejero R, Winkler DC, Menendez M, Steven AC et al. The role of capsid maturation on adenovirus priming for sequential uncoating. J Biol Chem 2012; 287: 31582–31595.
Graziano V, McGrath WJ, Suomalainen M, Greber UF, Freimuth P, Blainey PC et al. Regulation of a viral proteinase by a peptide and DNA in one-dimensional space: I. Binding to DNA AND to hexon of the precursor to protein VI, pVI, of human adenovirus. J Biol Chem 2013; 288: 2059–2067.
Blainey PC, Graziano V, Perez-Berna AJ, McGrath WJ, Flint SJ, San Martin C et al. Regulation of a viral proteinase by a peptide and DNA in one-dimensional space: IV. Viral proteinase slides along DNA to locate and process its substrates. J Biol Chem 2013; 288: 2092–2102.
Kamen A, Henry O . Development and optimization of an adenovirus production process. J Gene Med 2004; 6: 184–192.
Swaminathan S, Thimmapaya B . Regulation of adenovirus E2 transcription Uuit. In: Doerfler W, Böhm P (eds) The Molecular Repertoire of Adenoviruses III vol. 199/3. Springer: Berlin, Heidelberg, 1995, pp 177–194.
Weber J . Genetic analysis of adenovirus type 2 III. Temperature sensitivity of processing viral proteins. J Virol 1976; 17: 462–471.
Tribouley C, Lutz P, Staub A, Kedinger C . The product of the adenovirus intermediate gene IVa2 is a transcriptional activator of the major late promoter. J Virol 1994; 68: 4450–4457.
Zhang W, Low JA, Christensen JB, Imperiale MJ . Role for the adenovirus IVa2 protein in packaging of viral DNA. J Virol 2001; 75: 10446–10454.
Altaras NE, Aunins JG, Evans RK, Kamen A, Konz JO, Wolf JJ . Production and formulation of adenovirus vectors. Adv Biochem Eng Biotechnol 2005; 99: 193–260.
Jiang H, White EJ, Rios-Vicil CI, Xu J, Gomez-Manzano C, Fueyo J . Human adenovirus type 5 induces cell lysis through autophagy and autophagy-triggered caspase activity. J Virol 2011; 85: 4720–4729.
Rodriguez-Rocha H, Gomez-Gutierrez JG, Garcia-Garcia A, Rao XM, Chen L, McMasters KM et al. Adenoviruses induce autophagy to promote virus replication and oncolysis. Virology 2011; 416: 9–15.
Nevins JR . Regulation of early adenovirus gene expression. Microbiol Rev 1987; 51: 419–430.
Ross PJ, Kennedy MA, Parks RJ . Host cell detection of noncoding stuffer DNA contained in helper-dependent adenovirus vectors leads to epigenetic repression of transgene expression. J Virol 2009; 83: 8409–8417.
Parks RJ, Bramson JL, Wan Y, Addison CL, Graham FL . Effects of stuffer DNA on transgene expression from helper-dependent adenovirus vectors. J Virol 1999; 73: 8027–8034.
Ohsumi Y . Molecular dissection of autophagy: two ubiquitin-like systems. Nat Rev Mol Cell Biol 2001; 2: 211–216.
Levine B, Klionsky DJ . Development by self-digestion: molecular mechanisms and biological functions of autophagy. Developmental cell 2004; 6: 463–477.
Baehrecke EH . Autophagy: dual roles in life and death? Nat Rev Mol Cell Biol 2005; 6: 505–510.
Gozuacik D, Kimchi A . Autophagy and cell death. Curr Topics Dev Biol 2007; 78: 217–245.
Ostapchuk P, Hearing P . Control of adenovirus packaging. J Cell Biochem 2005; 96: 25–35.
Perez-Romero P, Gustin KE, Imperiale MJ . Dependence of the encapsidation function of the adenovirus L1 52/55-kilodalton protein on its ability to bind the packaging sequence. J Virol 2006; 80: 1965–1971.
Ostapchuk P, Anderson ME, Chandrasekhar S, Hearing P . The L4 22-kilodalton protein plays a role in packaging of the adenovirus genome. J Virol 2006; 80: 6973–6981.
Ma HC, Hearing P . Adenovirus structural protein IIIa is involved in the serotype specificity of viral DNA packaging. J Virol 2011; 85: 7849–7855.
Erturk E, Ostapchuk P, Wells SI, Yang J, Gregg K, Nepveu A et al. Binding of CCAAT displacement protein CDP to adenovirus packaging sequences. J Virol 2003; 77: 6255–6264.
Mangel WF, McGrath WJ, Toledo DL, Anderson CW . Viral DNA and a viral peptide can act as cofactors of adenovirus virion proteinase activity. Nature 1993; 361: 274–275.
Chen PH, Ornelles DA, Shenk T . The adenovirus L3 23-kilodalton proteinase cleaves the amino-terminal head domain from cytokeratin 18 and disrupts the cytokeratin network of HeLa cells. J Virol 1993; 67: 3507–3514.
Fernandes P, Santiago VM, Rodrigues AF, Tomas H, Kremer EJ, Alves PM et al. Impact of E1 and Cre on adenovirus vector amplification: developing MDCK CAV-2-E1 and E1-Cre transcomplementing cell lines. PLoS One 2013; 8: e60342.
Kramberger P, Ciringer M, Strancar A, Peterka M . Evaluation of nanoparticle tracking analysis for total virus particle determination. Virol J 2012; 9: 265.
Serra M, Leite SB, Brito C, Costa J, Carrondo MJ, Alves PM . Novel culture strategy for human stem cell proliferation and neuronal differentiation. J Neurosci Res 2007; 85: 3557–3566.
Salinas S, Bilsland LG, Henaff D, Weston AE, Keriel A, Schiavo G et al. CAR-associated vesicular transport of an adenovirus in motor neuron axons. PLoS Pathogens 2009; 5: e1000442.
Russell WC . Update on adenovirus and its vectors. J Gen Virol 2000; 81 (Pt 11): 2573–2604.
Weitzman MD, Ornelles DA . Inactivating intracellular antiviral responses during adenovirus infection. Oncogene 2005; 24: 7686–7696.
Acknowledgements
We thank Sandy Ibanes and Aurelie Gennetier for the initial preparations of CAV-2 vectors, Francisca Monteiro, Vanessa Bandeira and Rute Castro for technical support. This work was supported by Fundação para a Ciência e a Tecnologia (FCT)—Portugal, through the project PTDC/EBB-BIO/118615/2010, and European Commission through FP7 project BrainCAV (222992). Paulo Fernandes acknowledges FCT for his PhD grant (SFRH/BD/70810/2010).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Fernandes, P., Simão, D., Guerreiro, M. et al. Impact of adenovirus life cycle progression on the generation of canine helper-dependent vectors. Gene Ther 22, 40–49 (2015). https://doi.org/10.1038/gt.2014.92
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2014.92