Abstract
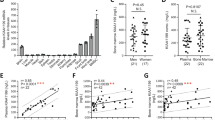
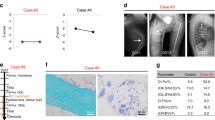
Fibrodysplasia ossificans progressiva (FOP) is a rare autosomal dominant disorder of progressive heterotopic ossification for which there is presently no cure. FOP is caused by a recurrent heterozygous activating mutation (c.617G>A; R206H) of Activin receptor type IA/Activin-like kinase-2 (ACVR1/ALK2), a bone morphogenetic protein (BMP) type I receptor that occurs in all classically affected individuals. The FOP mutation dysregulates BMP signaling and initiates the formation of a disabling second skeleton of heterotopic bone. We generated allele-specific siRNA (ASP-RNAi) duplexes capable of specifically suppressing the expression of the mutant c.617A allele in mesenchymal progenitor cells from FOP patients and showed that this ASP-RNAi approach decreased the elevated BMP signaling that is characteristic of patient cells to levels similar to control cells and restored enhanced osteogenic differentiation to control levels. Our results provide proof-of-principle that ASP-RNAi has potential therapeutic efficacy for the treatment of FOP.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Shore EM, Xu M, Feldman GJ, Fenstermacher DA, Cho TJ, Choi IH et al. A recurrent mutation in the BMP type I receptor ACVR1 causes inherited and sporadic fibrodysplasia ossificans progressiva. Nat Genet 2006; 38: 525–527.
Billings PC, Fiori JL, Bentwood JL, O’Connell MP, Jiao X, Nussbaum B et al. Dysregulated BMP signaling and enhanced osteogenic differentiation of connective tissue progenitor cells from patients with fibrodysplasia ossificans progressiva (FOP). J Bone Miner Res 2008; 23: 305–313.
Shen Q, Little SC, Xu M, Haupt J, Ast C, Katagiri T et al. The fibrodysplasia ossificans progressiva R206H ACVR1 mutation activates BMP-independent chondrogenesis and zebrafish embryo ventralization. J Clin Invest 2009; 119: 3462–3472.
Yu PB, Deng DY, Lai CS, Hong CC, Cuny GD, Bouxsein ML et al. BMP type I receptor inhibition reduces heterotopic ossification. Nat Med 2008; 14: 1363–1369.
Miller VM, Xia H, Marrs GL, Gouvion CM, Lee G, Davidson BL et al. Allele-specific silencing of dominant disease genes. Proc Natl Acad Sci USA 2003; 100: 7195–7200.
Rodriguez-Lebron E, Paulson HL . Allele-specific RNA interference for neurological disease. Gene Therapy 2006; 13: 576–581.
Ohnishi Y, Tamura Y, Yoshida M, Tokunaga K, Hohjoh H . Enhancement of allele discrimination by introduction of nucleotide mismatches into siRNA in allele-specific gene silencing by RNAi. PLoS One 2008; 3: e2248.
Pfister EL, Kennington L, Straubhaar J, Wagh S, Liu W, DiFiglia M . Five siRNAs Targeting Three SNPs May Provide Therapy for Three-Quarters of Huntington's Disease patients. Curr Biol 2009; 19: 774–778.
Harper SQ, Staber PD, He X, Eliason SL, Martins IH, Mao Q et al. RNA interference improves motor and neuropathological abnormalities in a Huntington's disease mouse model. Proc Natl Acad Sci USA 2005; 102: 5820–5825.
Miller VM, Gouvion CM, Davidson BL, Paulson HL . Targeting Alzheimer's disease genes with RNA interference: an efficient strategy for silencing mutant alleles. Nucleic Acids Res 2004; 32: 661–668.
Rodriguez-Lebron E, Gouvion CM, Moore SA, Davidson BL, Paulson HL . Allele-specific RNAi mitigates phenotypic progression in a transgenic model of Alzheimer's disease. Mol Ther 2009; 17: 1563–1573.
Xia X, Zhou H, Huang Y, Xu Z . Allele-specific RNAi selectively silences mutant SOD1 and achieves significant therapeutic benefit in vivo. Neurobiol Dis 2006; 23: 578–586.
Raoul C, Abbas-Terki T, Bensadoun JC, Guillot S, Haase G, Szulc et al. Lentiviral-mediated silencing of SOD1 through RNA interference retards disease onset and progression in a mouse model of ALS. Nat Med 2005; 11: 423–428.
Ding H, Schwarz DS, Keene A, Affar el B, Fenton L, Xia X et al. Selective silencing by RNAi of a dominant allele that causes amyotrophic lateral sclerosis. Aging Cell 2003; 2: 209–217.
Miura M, Gronthos S, Zhao M, Lu B, Fisher LW, Robey PG et al. SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci USA 2003; 100: 5807–5812.
Yamaza T, Kentaro A, Chen C, Liu Y, Gronthos S, Wang S et al. Immunomodulatory properties of stem cells from human exfoliated deciduous teeth. Stem Cell Res Ther 2010; 1: 5–15.
Chadipiralla K, Yochim JM, Bahuleyan B, Huang CY Garcia- Godoy F, Murray PE et al. Osteogenic differentiation of stem cells derived from human periodontal ligaments and pulp of human exfoliated deciduous teeth. Cell Tissue Res 2010; 340: 323–333.
Pei Y, Tuschl T . On the art of identifying effective and specific siRNAs. Nat Methods 2006; 14: 1557–1565.
Seyhan AA . RNAi: a potential new class of therapeutic for human genetic disease. Hum Genet 2011; 130: 583–605.
Kurreck J . RNA interference: From basic research to therapeutic applications. Angew Chem Int Ed Engl 2009; 48: 1378–1398.
Higuchi Y, Kawakami S, Hashida M . Strategies for in vivo delivery of siRNAs: recent progress. BioDrugs 2010; 24: 195–205.
Dykxhoorn DM . Advances in cell-type specific delivery of RNAi-based therapeutics. IDrugs 2010; 13: 325–331.
Gronthos S, Mankani M, Brahim J, Robey PG, Shi S . Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci USA 2000; 97: 13625–13630.
Acknowledgements
We thank the members of our research laboratory for their helpful discussions during the development of these studies. This work was supported in part by the Center for Research in FOP and Related Disorders, the International FOP Association (IFOPA), the Ian Cali Endowment, the Weldon Family Endowment, the Isaac and Rose Nassau Professorship of Orthopaedic Molecular Medicine, the Rita Allen Foundation, and the Penn Musculoskeletal Center, and by grants from the NIH (R01-AR41916 and R01-AR046831).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Kaplan, J., Kaplan, F. & Shore, E. Restoration of normal BMP signaling levels and osteogenic differentiation in FOP mesenchymal progenitor cells by mutant allele-specific targeting. Gene Ther 19, 786–790 (2012). https://doi.org/10.1038/gt.2011.152
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2011.152
Keywords
This article is cited by
-
Genetics and future therapy prospects of fibrodysplasia ossificans progressiva
Medizinische Genetik (2020)
-
The biological function of type I receptors of bone morphogenetic protein in bone
Bone Research (2016)
-
Identification and characterization of regulatory elements in the promoter of ACVR1, the gene mutated in Fibrodysplasia Ossificans Progressiva
Orphanet Journal of Rare Diseases (2013)
-
Repair of large segmental bone defects: BMP-2 gene activated muscle grafts vs. autologous bone grafting
BMC Biotechnology (2013)
-
Overactive bone morphogenetic protein signaling in heterotopic ossification and Duchenne muscular dystrophy
Cellular and Molecular Life Sciences (2013)