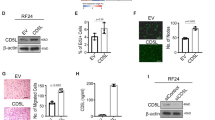
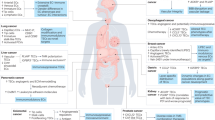
Abstract
Endothelial progenitor cells (EPCs) are promising for cancer therapy because they specifically target tumors. They have the capacity to home to, invade, migrate within and incorporate into tumor structures. They are easily expanded and can be armed with therapeutic payloads protected within the progenitor cells. Once in the tumor, armed EPCs can be triggered to induce cell death in surrounding tumor cells while being transiently protected from premature demise. In preclinical studies, therapeutic EPCs attenuated tumor growth and increased survival. Enhancing homing, self-protection and collateral tumor cell damage will increase the efficacy of EPCs for cancer gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Choi K, Kennedy M, Kazarov A, Papadimitriou JC, Keller G . A common precursor for hematopoietic and endothelial cells. Development 1998; 125: 725–732.
Shalaby F, Rossant J, Yamaguchi TP, Gertsenstein M, Wu XF, Breitman ML et al. Failure of blood-island formation and vasculogenesis in Flk-1-deficient mice. Nature 1995; 376: 62–66.
Ferrara N, Carver-Moore K, Chen H, Dowd M, Lu L, O’Shea KS et al. Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature 1996; 380: 439–442.
Asahara T, Murohara T, Sullivan A, Silver M, van der Zee R, Li T et al. Isolation of putative progenitor endothelial cells for angiogenesis. Science 1997; 275: 964–967.
Gunsilius E, Petzer AL, Duba HC, Kahler CM, Gastl G . Circulating endothelial cells after transplantation. Lancet 2001; 357: 1449–1450.
Rafii S, Lyden D . Therapeutic stem and progenitor cell transplantation for organ vascularization and regeneration. Nat Med 2003; 9: 702–712.
Peichev M, Naiyer AJ, Pereira D, Zhu Z, Lane WJ, Williams M et al. Expression of VEGFR-2 and AC133 by circulating human CD34(+) cells identifies a population of functional endothelial precursors. Blood 2000; 95: 952–958.
Gehling UM, Ergun S, Schumacher U, Wagener C, Pantel K, Otte M et al. In vitro differentiation of endothelial cells from AC133-positive progenitor cells. Blood 2000; 95: 3106–3112.
Case J, Mead LE, Bessler WK, Prater D, White HA, Saadatzadeh MR et al. Human CD34+AC133+VEGFR-2+ cells are not endothelial progenitor cells but distinct, primitive hematopoietic progenitors. Exp Hematol 2007; 35: 1109–1118.
Harraz M, Jiao C, Hanlon HD, Hartley RS, Schatteman GC . CD34− blood-derived human endothelial cell progenitors. Stem Cells 2001; 19: 304–312.
Shi Q, Rafii S, Wu MH, Wijelath ES, Yu C, Ishida A et al. Evidence for circulating bone marrow-derived endothelial cells. Blood 1998; 92: 362–367.
Bertolini F, Paul S, Mancuso P, Monestiroli S, Gobbi A, Shaked Y et al. Maximum tolerable dose and low-dose metronomic chemotherapy have opposite effects on the mobilization and viability of circulating endothelial progenitor cells. Cancer Res 2003; 63: 4342–4346.
Ho JW, Pang RW, Lau C, Sun CK, Yu WC, Fan ST et al. Significance of circulating endothelial progenitor cells in hepatocellular carcinoma. Hepatology 2006; 44: 836–843.
Dome B, Timar J, Dobos J, Meszaros L, Raso E, Paku S et al. Identification and clinical significance of circulating endothelial progenitor cells in human non-small cell lung cancer. Cancer Res 2006; 66: 7341–7347.
Asahara T, Masuda H, Takahashi T, Kalka C, Pastore C, Silver M et al. Bone marrow origin of endothelial progenitor cells responsible for postnatal vasculogenesis in physiological and pathological neovascularization. Circ Res 1999; 85: 221–228.
Davidoff AM, Ng CY, Brown P, Leary MA, Spurbeck WW, Zhou J et al. Bone marrow-derived cells contribute to tumor neovasculature and, when modified to express an angiogenesis inhibitor, can restrict tumor growth in mice. Clin Cancer Res 2001; 7: 2870–2879.
Garcia-Barros M, Paris F, Cordon-Cardo C, Lyden D, Rafii S, Haimovitz-Friedman A et al. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science 2003; 300: 1155–1159.
Lyden D, Hattori K, Dias S, Costa C, Blaikie P, Butros L et al. Impaired recruitment of bone-marrow-derived endothelial and hematopoietic precursor cells blocks tumor angiogenesis and growth. Nat Med 2001; 7: 1194–1201.
Spring H, Schuler T, Arnold B, Hammerling GJ, Ganss R . Chemokines direct endothelial progenitors into tumor neovessels. Proc Natl Acad Sci USA 2005; 102: 18111–18116.
Nolan DJ, Ciarrocchi A, Mellick AS, Jaggi JS, Bambino K, Gupta S et al. Bone marrow-derived endothelial progenitor cells are a major determinant of nascent tumor neovascularization. Genes Dev 2007; 21: 1546–1558.
Shaked Y, Ciarrocchi A, Franco M, Lee CR, Man S, Cheung AM et al. Therapy-induced acute recruitment of circulating endothelial progenitor cells to tumors. Science 2006; 313: 1785–1787.
Yu D, Sun X, Qiu Y, Zhou J, Wu Y, Zhuang L et al. Identification and clinical significance of mobilized endothelial progenitor cells in tumor vasculogenesis of hepatocellular carcinoma. Clin Cancer Res 2007; 13: 3814–3824.
Gothert JR, Gustin SE, van Eekelen JA, Schmidt U, Hall MA, Jane SM et al. Genetically tagging endothelial cells in vivo: bone marrow-derived cells do not contribute to tumor endothelium. Blood 2004; 104: 1769–1777.
Larrivee B, Niessen K, Pollet I, Corbel SY, Long M, Rossi FM et al. Minimal contribution of marrow-derived endothelial precursors to tumor vasculature. J Immunol 2005; 175: 2890–2899.
De Palma M, Venneri MA, Roca C, Naldini L . Targeting exogenous genes to tumor angiogenesis by transplantation of genetically modified hematopoietic stem cells. Nat Med 2003; 9: 789–795.
Rajantie I, Ilmonen M, Alminaite A, Ozerdem U, Alitalo K, Salven P . Adult bone marrow-derived cells recruited during angiogenesis comprise precursors for periendothelial vascular mural cells. Blood 2004; 104: 2084–2086.
Kopp HG, Ramos CA, Rafii S . Contribution of endothelial progenitors and proangiogenic hematopoietic cells to vascularization of tumor and ischemic tissue. Curr Opin Hematol 2006; 13: 175–181.
Heissig B, Hattori K, Dias S, Friedrich M, Ferris B, Hackett NR et al. Recruitment of stem and progenitor cells from the bone marrow niche requires MMP-9 mediated release of kit-ligand. Cell 2002; 109: 625–637.
Gill M, Dias S, Hattori K, Rivera ML, Hicklin D, Witte L et al. Vascular trauma induces rapid but transient mobilization of VEGFR2(+)AC133(+) endothelial precursor cells. Circ Res 2001; 88: 167–174.
Carmeliet P, Moons L, Luttun A, Vincenti V, Compernolle V, De Mol M et al. Synergism between vascular endothelial growth factor and placental growth factor contributes to angiogenesis and plasma extravasation in pathological conditions. Nat Med 2001; 7: 575–583.
Kim KJ, Li B, Winer J, Armanini M, Gillett N, Phillips HS et al. Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 1993; 362: 841–844.
Millauer B, Shawver LK, Plate KH, Risau W, Ullrich A . Glioblastoma growth inhibited in vivo by a dominant-negative Flk-1 mutant. Nature 1994; 367: 576–579.
Luttun A, Tjwa M, Moons L, Wu Y, Angelillo-Scherrer A, Liao F et al. Revascularization of ischemic tissues by PlGF treatment, and inhibition of tumor angiogenesis, arthritis and atherosclerosis by anti-Flt1. Nat Med 2002; 8: 831–840.
Aghi M, Cohen KS, Klein RJ, Scadden DT, Chiocca EA . Tumor stromal-derived factor-1 recruits vascular progenitors to mitotic neovasculature, where microenvironment influences their differentiated phenotypes. Cancer Res 2006; 66: 9054–9064.
Petit I, Jin D, Rafii S . The SDF-1-CXCR4 signaling pathway: a molecular hub modulating neo-angiogenesis. Trends Immunol 2007; 28: 299–307.
Carmeliet P, Ferreira V, Breier G, Pollefeyt S, Kieckens L, Gertsenstein M et al. Abnormal blood vessel development and lethality in embryos lacking a single VEGF allele. Nature 1996; 380: 435–439.
Shalaby F, Ho J, Stanford WL, Fischer KD, Schuh AC, Schwartz L et al. A requirement for Flk1 in primitive and definitive hematopoiesis and vasculogenesis. Cell 1997; 89: 981–990.
Gerber HP, McMurtrey A, Kowalski J, Yan M, Keyt BA, Dixit V et al. Vascular endothelial growth factor regulates endothelial cell survival through the phosphatidylinositol 3′-kinase/Akt signal transduction pathway. Requirement for Flk-1/KDR activation. J Biol Chem 1998; 273: 30336–30343.
Kim I, Kim HG, So JN, Kim JH, Kwak HJ, Koh GY . Angiopoietin-1 regulates endothelial cell survival through the phosphatidylinositol 3′-Kinase/Akt signal transduction pathway. Circ Res 2000; 86: 24–29.
Datta SR, Katsov A, Hu L, Petros A, Fesik SW, Yaffe MB et al. 14-3-3 proteins and survival kinases cooperate to inactivate BAD by BH3 domain phosphorylation. Mol Cell 2000; 6: 41–51.
Dimmeler S, Haendeler J, Nehls M, Zeiher AM . Suppression of apoptosis by nitric oxide via inhibition of interleukin-1beta-converting enzyme (ICE)-like and cysteine protease protein (CPP)-32-like proteases. J Exp Med 1997; 185: 601–607.
Butzal M, Loges S, Schweizer M, Fischer U, Gehling UM, Hossfeld DK et al. Rapamycin inhibits proliferation and differentiation of human endothelial progenitor cells in vitro. Exp Cell Res 2004; 300: 65–71.
Gratton JP, Morales-Ruiz M, Kureishi Y, Fulton D, Walsh K, Sessa WC . Akt down-regulation of p38 signaling provides a novel mechanism of vascular endothelial growth factor-mediated cytoprotection in endothelial cells. J Biol Chem 2001; 276: 30359–30365.
Gerber HP, Dixit V, Ferrara N . Vascular endothelial growth factor induces expression of the antiapoptotic proteins Bcl-2 and A1 in vascular endothelial cells. J Biol Chem 1998; 273: 13313–13316.
O’Connor DS, Schechner JS, Adida C, Mesri M, Rothermel AL, Li F et al. Control of apoptosis during angiogenesis by survivin expression in endothelial cells. Am J Pathol 2000; 156: 393–398.
Tran J, Rak J, Sheehan C, Saibil SD, LaCasse E, Korneluk RG et al. Marked induction of the IAP family antiapoptotic proteins survivin and XIAP by VEGF in vascular endothelial cells. Biochem Biophys Res Commun 1999; 264: 781–788.
Carmeliet P, Lampugnani MG, Moons L, Breviario F, Compernolle V, Bono F et al. Targeted deficiency or cytosolic truncation of the VE-cadherin gene in mice impairs VEGF-mediated endothelial survival and angiogenesis. Cell 1999; 98: 147–157.
Gao C, Sun W, Christofidou-Solomidou M, Sawada M, Newman DK, Bergom C et al. PECAM-1 functions as a specific and potent inhibitor of mitochondrial-dependent apoptosis. Blood 2003; 102: 169–179.
Malyankar UM, Scatena M, Suchland KL, Yun TJ, Clark EA, Giachelli CM . Osteoprotegerin is an alpha vbeta 3-induced, NF-kappa B-dependent survival factor for endothelial cells. J Biol Chem 2000; 275: 20959–20962.
Stupack DG, Cheresh DA . Apoptotic cues from the extracellular matrix: regulators of angiogenesis. Oncogene 2003; 22: 9022–9029.
Benjamin LE, Hemo I, Keshet E . A plasticity window for blood vessel remodelling is defined by pericyte coverage of the preformed endothelial network and is regulated by PDGF-B and VEGF. Development 1998; 125: 1591–1598.
Dernbach E, Urbich C, Brandes RP, Hofmann WK, Zeiher AM, Dimmeler S . Antioxidative stress-associated genes in circulating progenitor cells: evidence for enhanced resistance against oxidative stress. Blood 2004; 104: 3591–3597.
Levenberg S, Golub JS, Amit M, Itskovitz-Eldor J, Langer R . Endothelial cells derived from human embryonic stem cells. Proc Natl Acad Sci USA 2002; 99: 4391–4396.
Wei J, Blum S, Unger M, Jarmy G, Lamparter M, Geishauser A et al. Embryonic endothelial progenitor cells armed with a suicide gene target hypoxic lung metastases after intravenous delivery. Cancer Cell 2004; 5: 477–488.
Ingram DA, Mead LE, Moore DB, Woodard W, Fenoglio A, Yoder MC . Vessel wall derived endothelial cells rapidly proliferate because they contain a complete hierarchy of endothelial progenitor cells. Blood 2004; 104: 2752–2760.
Murohara T, Ikeda H, Duan J, Shintani S, Sasaki K, Eguchi H et al. Transplanted cord blood-derived endothelial precursor cells augment postnatal neovascularization. J Clin Invest 2000; 105: 1527–1536.
Yoder MC, Mead LE, Prater D, Krier TR, Mroueh KN, Li F et al. Redefining endothelial progenitor cells via clonal analysis and hematopoietic stem/progenitor cell principals. Blood 2007; 109: 1801–1809.
Lin Y, Weisdorf DJ, Solovey A, Hebbel RP . Origins of circulating endothelial cells and endothelial outgrowth from blood. J Clin Invest 2000; 105: 71–77.
Reyes M, Dudek A, Jahagirdar B, Koodie L, Marker PH, Verfaillie CM . Origin of endothelial progenitors in human postnatal bone marrow. J Clin Invest 2002; 109: 337–346.
Asahara T, Takahashi T, Masuda H, Kalka C, Chen D, Iwaguro H et al. VEGF contributes to postnatal neovascularization by mobilizing bone marrow-derived endothelial progenitor cells. EMBO J 1999; 18: 3964–3972.
Dimmeler S, Aicher A, Vasa M, Mildner-Rihm C, Adler K, Tiemann M et al. HMG-CoA reductase inhibitors (statins) increase endothelial progenitor cells via the PI 3-kinase/Akt pathway. J Clin Invest 2001; 108: 391–397.
Shaked Y, Kerbel RS . Antiangiogenic strategies on defense: on the possibility of blocking rebounds by the tumor vasculature after chemotherapy. Cancer Res 2007; 67: 7055–7058.
Ferrari N, Glod J, Lee J, Kobiler D, Fine HA . Bone marrow-derived, endothelial progenitor-like cells as angiogenesis-selective gene-targeting vectors. Gene Therapy 2003; 10: 647–656.
Moore XL, Lu J, Sun L, Zhu CJ, Tan P, Wong MC . Endothelial progenitor cells’ ‘homing’ specificity to brain tumors. Gene Therapy 2004; 11: 811–818.
Le Ricousse-Roussanne S, Barateau V, Contreres JO, Boval B, Kraus-Berthier L, Tobelem G . Ex vivo differentiated endothelial and smooth muscle cells from human cord blood progenitors home to the angiogenic tumor vasculature. Cardiovasc Res 2004; 62: 176–184.
Freeman SM, Abboud CN, Whartenby KA, Packman CH, Koeplin DS, Moolten FL et al. The ‘bystander effect’: tumor regression when a fraction of the tumor mass is genetically modified. Cancer Res 1993; 53: 5274–5283.
Rancourt C, Robertson III MW, Wang M, Goldman CK, Kelly JF, Alvarez RD et al. Endothelial cell vehicles for delivery of cytotoxic genes as a gene therapy approach for carcinoma of the ovary. Clin Cancer Res 1998; 4: 265–270.
Gomez-Navarro J, Contreras JL, Arafat W, Jiang XL, Krisky D, Oligino T et al. Genetically modified CD34+ cells as cellular vehicles for gene delivery into areas of angiogenesis in a rhesus model. Gene Therapy 2000; 7: 43–52.
Arafat WO, Casado E, Wang M, Alvarez RD, Siegal GP, Glorioso JC et al. Genetically modified CD34+ cells exert a cytotoxic bystander effect on human endothelial and cancer cells. Clin Cancer Res 2000; 6: 4442–4448.
Benouchan M, Do Nascimento F, Perret GY, Colombo BM . Delivery of the bacterial nitroreductase gene into endothelial cells prolongs the survival of tumour-bearing mice by bystander mechanisms. Int J Oncol 2006; 28: 457–462.
Wei J, Jarmy G, Genuneit J, Debatin KM, Beltinger C . Human blood late outgrowth endothelial cells for gene therapy of cancer: determinants of efficacy. Gene Therapy 2007; 14: 344–356.
Dudek AZ, Bodempudi V, Welsh BW, Jasinski P, Griffin RJ, Milbauer L et al. Systemic inhibition of tumour angiogenesis by endothelial cell-based gene therapy. Br J Cancer 2007; 97: 513–522.
Jevremovic D, Gulati R, Hennig I, Diaz RM, Cole C, Kleppe L et al. Use of blood outgrowth endothelial cells as virus-producing vectors for gene delivery to tumors. Am J Physiol Heart Circ Physiol 2004; 287: H494–H500.
Wei J, Zhou S, Bachem G, Debatin KM, Beltinger C . Infiltration of blood outgrowth endothelial cells into tumor spheroids: role of matrix metalloproteinases and irradiation. Anticancer Res 2007; 27: 1415–1421.
Ojeifo JO, Lee HR, Rezza P, Su N, Zwiebel JA . Endothelial cell-based systemic gene therapy of metastatic melanoma. Cancer Gene Ther 2001; 8: 636–648.
Wei J, Wahl J, Nakamura T, Stiller D, Mertens T, Debatin KM et al. Targeted release of oncolytic measles virus by blood outgrowth endothelial cells in situ inhibits orthotopic gliomas. Gene Therapy 2007; 14: 1573–1586.
Phuong LK, Allen C, Peng KW, Giannini C, Greiner S, TenEyck CJ et al. Use of a vaccine strain of measles virus genetically engineered to produce carcinoembryonic antigen as a novel therapeutic agent against glioblastoma multiforme. Cancer Res 2003; 63: 2462–2469.
Oh HK, Ha JM, O E, Lee BH, Lee SK, Shim BS et al. Tumor angiogenesis promoted by ex vivo differentiated endothelial progenitor cells is effectively inhibited by an angiogenesis inhibitor, TK1-2. Cancer Res 2007; 67: 4851–4859.
Acknowledgements
We thank Nora Hipp for clerical assistance and Helgard Knauß for artwork. This work was supported in part by a grant from the Deutsche Forschungsgemeinschaft (to CB).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Debatin, KM., Wei, J. & Beltinger, C. Endothelial progenitor cells for cancer gene therapy. Gene Ther 15, 780–786 (2008). https://doi.org/10.1038/gt.2008.36
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2008.36