Abstract
Purpose: To study the inheritance and characteristics of familial Meniere disease in Finland and genetic linkage to the previously proposed locus on chromosome 12p12.3.
Methods: Sixteen Meniere families recruited from Kainuu Central Hospital and Helsinki and Oulu University Hospitals in the period 2001–2004 were reevaluated in 2009 using hospital records and mailed questionnaire forms. Ten highly polymorphic microsatellite markers were selected from the area of chromosome 12p12.3 and studied for linkage using the GENEHUNTER protocol.
Results: The families showed autosomal dominant inheritance without cosegregation with migraine. Anticipation was seen only in one family, and in the rest of the families, the age of onset varied randomly among generations and individuals. The severity of the disease was not related to descending generations. None of the maximum logarithm of odds (LOD)/heterogeneity LOD scores in the analysis of chromosome 12p12.3 in Finnish Meniere families reached a significant value of 3.0 (maximum cumulative LOD score: −7.29, heterogeneity LOD: −0.95, α = 0.4).
Conclusions: Families affected by Meniere disease are highly heterogeneous. Migraine, age at onset, anticipation, or penetrance was not a shared feature. The findings support the multifactorial nature of the disease and indicate that genetic heterogeneity exists within familial Meniere disease.
Similar content being viewed by others
Main
Meniere disease (MD) is an inner-ear disorder characterized by tinnitus, vertigo, and sensorineural hearing loss. Although its etiology is still unknown, endolymphatic hydrops, fluid overload in the inner ear has been demonstrated in histological examinations.1 The prevalence of MD in Finland was estimated by Kotimäki et al.2 to be at least 43 of 100,000, with the highest prevalence in Northern Finland. There have been numerous reports on familial MD (FMD),3–6 the estimated proportion of which in Finland is approximately 15%.7
In addition to familial clustering, differences in race and population incidences8 and geographical distribution2 support the role of genetic factors in the development of MD. A link has been demonstrated between migraine and MD,9,10 and it has even been suggested that MD develops as a complication of migraine-associated symptoms.11 The overrepresentation of migraine among patients with MD might also be explained by migraine-associated vertigo, mimicking MD.12 Anticipation, a phenomenon in which the symptoms of an inheritable disorder become apparent at an earlier age as they are passed on to the next generation, has also been described in MD.13 Most reports concerned with FMD suggest dominant inheritance with incomplete penetrance, but families with recessive inheritance have been described.3
Several candidate genes involved in the regulation of inner-ear fluid homeostasis have been associated with MD, but to date, no positive replications have been reported.14–20 To the best of our knowledge, only one genome-wide linkage study of MD has been published previously, indicating a putative MD locus on chromosome 12p12.3 in three Swedish families.21 No mutations in the 463-kb candidate region have been reported so far.
In this study, we describe 16 Finnish MD families, their reevaluation 5 years after the initial recruitment, and a study of genetic linkage to the previously proposed locus on chromosome 12p12.3. This represents the first molecular genetic study of patients with MD in Finland.
MATERIALS AND METHODS
Subjects
The series comprised a total of 16 families, the probands of which were recruited from the ear, nose, and throat (ENT) clinics of Oulu University Hospital, Kainuu Central Hospital, and Helsinki University Hospital during the years 2001–2004. Probands were searched for by screening the hospital records for patients with a family history of MD. Sixteen families willing to participate in this study were found. Blood samples were received from 67 individuals for genomic DNA isolation after obtaining a signed consent form. The study was approved by the Ethics Committee of the Northern Ostrobothnia Hospital District.
Clinical assessment
Relatives were interviewed on the phone to determine their disease status (2001–2004). All the patients with any symptoms resembling MD were evaluated by an ENT specialist conversant with MD at either a university or central hospital. In addition to the clinical examinations and interviews, an audiogram was used to measure hearing loss, and either a head magnetic resonance imaging or auditory brainstem response examination was performed to exclude VIII cranial nerve tumors. Relatives who reported having no symptoms resembling MD were not subjected to clinical studies.
All the families were reevaluated in 2009 by means of telephone interviews, mailed questionnaire, and hospital records. A questionnaire concerning heredity, the prevalence of Meniere-like symptoms, the severity and frequency of the disease, and age at its onset was sent to all the subjects. Relatives who had manifested Meniere-like symptoms during the years 2004–2009 were referred to an ENT clinic if clinical examination had not been performed previously.
The patients were classified using the latest guidelines of the American Academy of Otolaryngology—Head and Neck Surgery.22 For definite MD, two or more vertiginous episodes lasting at least for 20 minutes, documented sensorineural hearing loss (0.5, 1.0, 2.0, and 3.0 kHz), and tinnitus or aural fullness were required with other causes excluded. For probable MD, the diagnostic criteria were the same as for definite disease, for the exception that only one vertiginous episode was required. Patients with tinnitus, hearing loss, or vertigo episodes that did not fulfill the previously described criteria were considered a separate group with symptoms possibly related to the MD inherited in these families (other causes excluded).
Genotyping
Genomic DNA was extracted from white blood cells according to standard protocols. Ten highly polymorphic microsatellite markers were analyzed from chromosomal 12p12.3 region, which had previously been linked to MD in Swedish families,21 and four additional markers were selected from the MAP-O-MAT database (http://compgen.rutgers.edu/mapomat/). The 5′-fluorescent-labeled markers were amplified by polymerase chain reaction (PCR) in a volume of 11 μL, which contained 20 ng of genomic DNA, 3 pmol of PCR primers, 1.5 mM of MgCl2, 0.2 mM dNTPs, and one unit of Supertherm Taq DNA polymerase (Medox Biotech India). The conditions after initial denaturation at 94.5°C were 30 cycles of 30 seconds at 94.5°C, 30 seconds at 56–62°C and 30 seconds at 72°C, and a final extension at 72°C for 7 minutes. The PCR products from the same individual were pooled and combined with formamide and an internal size standard (GeneScan-500LIZ, Applied Biosystems, Foster City, CA). The pooling was designed in such a way that markers could be identified based on their size and/or different fluorescent dyes. After denaturation, the pooled products were separated on an Applied Biosystems 3130XL Genetic analyzer. The genotyping results were analyzed using Genemapper Software, version 4.0 (Applied Biosystems).
Statistical analysis
Patients with definite or probable MD were classified as affected in the statistical analysis. Because the age at onset of FMD was below 62 years in all the families, patients older than 65 years with no symptoms resembling MD were classified as healthy. Patients with Meniere-like symptoms that did not fulfill the diagnostic criteria described previously, healthy relatives younger than 65 years, and those who did not answer the follow-up questionnaire were set with unknown disease status in the statistical analysis. A total of 42 affected individuals, 10 healthy relatives, and 15 relatives of unknown disease status were analyzed. Mendelian consistency was checked using PedCheck V.1.1.23 Allele frequencies were estimated from the data using observed and reconstructed genotypes for the founders within the pedigrees. Both parametric and nonparametric logarithm of the odds (LOD)/heterogeneity LOD (HLOD)/NPL scores were calculated using GENEHUNTER, version 2.1.R5,24 using two inheritance models: dominant with 95% and dominant with 60% penetrance. The disease allele frequency was estimated to be 0.0005 in the dominant model with 95% penetrance and 0.0007 in the dominant model with 60% penetrance according to the estimated prevalence of FMD in Finland.
RESULTS
Finnish FMD
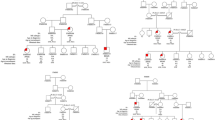
All the living patients with MD and 83% of the relatives replied to the mailed follow-up questionnaire sent out in 2009. Three patients with MD and three relatives had died during the years 2004–2009. Of the 30 individuals previously classified as healthy, seven (23%) had developed MD or symptoms resembling MD in the intervening years, and the telephone interviews revealed additional two cases of MD in relatives who had not originally been included in the series. Recessive inheritance had been suspected in Families 1 and 12 after the initial evaluation, but the more detailed data obtained in the questionnaire and interviews revealed a history of Meniere-like symptoms in deceased individuals in earlier generations. After the reevaluation in 2009, all families were consistent with autosomal dominant mode of inheritance. High variability of penetrance was seen among the families. The pedigrees of the families with updated information on clinical status are presented in Figure 1.
A slight female predominance was seen, with 65% of the patients with definite FMD and 62% of those with some symptoms resembling MD being female. Twenty-three percent of the affected individuals also suffered from migraine, but no obvious cosegregation of migraine and MD was seen. Only in Family 1 did all the affected individuals have migraine.
Anticipation was seen when the ages at the time of diagnosis retrieved from hospital records were compared between generations, but once the questionnaire data on the onset of each symptom had been taken into account, true anticipation was seen only in Family 2. In the rest of the families, the onset varied randomly between generations and individuals. The age at diagnosis varied from 21 to 62 years, but in most cases, patients reported having had their first symptoms 5–20 years before the actual diagnosis (Fig. 2). No tendency for successive generations to suffer from more severe clinical manifestations of MD was seen. Patients with a longer Meniere history had more severe hearing loss but less frequent vertigo attacks. The occurrence of unilateral (73%) and bilateral cases (27%) seemed to be random and not linked to descending generations.
Genotyping and statistical analysis
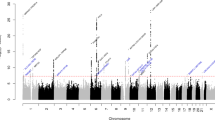
Ten polymorphic markers in the 2.5 cM MD candidate region on 12p12.3 were analyzed in 16 Finnish MD families, and the resulting LOD/HLOD scores in the dominant model with 95% and 60% penetrance are shown in Figure 3. The cumulative LOD/HLOD scores were negative in all models when using the GENEHUNTER protocol. At the level of individual families, a positive LOD score was obtained for three families (Families 4, 10, and 11), with a maximum value of 0.59 (Table 1). Haplotype analysis excluded a shared chromosomal region for the affected members of Families 10 and 11, but this was not possible for Family 4 due to the limited number of affected individuals. The result for Family 8 was nonexclusive. None of the NPL scores were statistically significant.
DISCUSSION
No evidence of anticipation or cosegregation with migraine was observed in these Finnish Meniere families, but variability in penetrance and a high tendency for relatives to manifest Meniere-like symptoms were seen. In addition, the results suggest heterogeneity in FMD, as linkage to chromosome 12p12.3, currently the only locus reported for MD, was excluded.
Late onset is considered typical of MD. According to the data from 1992 to 1996 collected from hospital records by Kotimäki et al.25 on 121 definite Finnish patients with MD, their age distribution was 33–89 years (mean, 59.5 years and median, 60 years). Although the data are not directly comparable, a much earlier average age at the onset of FMD is suggested by our family data, as has also been reported previously.6,7 In particular, Morrison et al.6 demonstrated the occurrence of the onset of FMD in the fourth decade, whereas the average onset seen in sporadic patients was in the fifth decade. This is consistent with the ages at the time of diagnosis recorded for our families. The peak for the onset of the first symptoms in our families nevertheless seemed to be even earlier, before the age of 30 years. It should be remembered, however, that MD is a well-known disorder among these families, and it is possible that greater knowledge of it may have led to an earlier diagnosis. The true differences between familial and sporadic MD can only be characterized by comparing these patient groups directly with one other.
Most reports suggest dominant inheritance for FMD,26 but families with possible recessive inheritance have been mentioned.3 Our results using a comprehensive heredity questionnaire and follow-up imply that the few reports of recessive inheritance may be the result of small family sizes or short follow-up times and that the most likely inheritance pattern for FMD is dominant with reduced penetrance. It is though possible, that the protocol used to recruit patients into this study could have led us toward finding dominant families.
Divergent results have been presented regarding the sex distribution of MD. Both male and female preponderances have been reported,27,28 but no clear difference between the sexes has been shown. The slight female preponderance in FMD seen in our results is supported by two earlier reports6,7 and may suggest that hormonal factors affect the pathology of this disease, although such an assumption would need to be studied in larger groups of patients.
Migraine has been associated to MD in numerous studies,9,10 and clustering of migraine and episodic vertigo has been reported in family members of patients with MD.29 It has not been established whether the two conditions share a pathophysiological link or whether migraine-associated symptoms are mimicking MD.12 In our study, only one family showed possible cosegregation of migraine and MD. The lack of association to migraine could be caused by not only the genetic heterogeneity but also the selection of the study material. Families with migraine-associated symptoms could have been left out from this study if only one family member fulfilled the strict diagnostic criteria for define MD. In Finland, migraine is generally treated in the basic health care system, and therefore, often no information about migraine is available from hospital records. Information about migraine was retrieved directly from patients, and this could also create a certain bias to this study. More studies are still needed to determine the heredity of migraine and MD-associated symptoms.
Strong genetic anticipation has been described in British MD families.6,13 Earlier diagnosis in subsequent generations may be a consequence of the more developed health care system relative to that available to affected relatives in preceding generations. This was seen in the Finnish families, where many of the deceased individuals in earlier generations had never been evaluated by a physician although their symptoms as described by family members were consistent with MD. Also, a greater knowledge of the disease may have led the offspring to seek medical attention earlier. The natural progression of the disease must also be considered before comparing the severity of symptoms between generations. Taking these factors into consideration, anticipation was not commonly seen in Finnish families, at least when information on two or more generations was available. The lack of anticipation seen in Finnish MD may be due to genetic heterogeneity in the disease itself, but more extensive evaluations of multigeneration families would be needed to determine whether anticipation really exists in FMD.
Klar et al.21 reported the first linkage of MD to chromosome 12p12.3 in three Swedish families with a significant LOD score of 3.46 using a dominant inheritance pattern and 100% penetrance. Further evidence for Swedish FMD susceptibility locus was obtained by Gabriková et al. using haplotype analysis in 15 Swedish families (including the families used in the work of Klar et al.), they identified a 1.48-Mb region on chromosome 12p12.3 that associated with FMD in 60% of the families.30 Our series nevertheless failed to provide any evidence of linkage to chromosome 12p12.3. Given that an obvious earlier onset in subsequent generations was seen in two of the three families studied by Klar et al., the lack of both anticipation and linkage to chromosome 12p12.3 in the Finnish families may suggest locus heterogeneity in FMD. The small number of families, however, causes limitations to both studies. Additional constraint may arise from the selection of markers in our study as only the peak region indicated by Klar et al. was studied. The negative LOD scores in 13 of the 16 families and the maximum cumulative LOD score of −7.29/HLOD −0.95 α = 0.4 (in the dominant model with 60% penetrance) indicate that genetic susceptibility to MD in these Finnish families must be located elsewhere.
In summary, we have shown that families affected by MD are highly heterogeneous and that migraine, age at onset, anticipation, or penetrance was not a shared feature. FMD has been studied relatively little, and the genetic basis of the disease is still unknown. This work represents the first replication study of the only locus suggested for MD, but it is clear that the genetic study of MD is greatly complicated by its multifactorial origin, genetic heterogeneity, and reduced penetrance. Greater numbers of larger families with detailed clinical information will be needed in the future to reveal the underlying genetic factors. As FMD is relatively rare, however, a joint effort will have to be made by researchers to reach a sufficient statistical power in genome-wide linkage analysis for adequate results to be achieved.
REFERENCES
Semaan MT, Alagramam KN, Megerian CA . The basic science of Meniere's disease and endolymphatic hydrops. Curr Opin Otolaryngol Head Neck Surg 2005; 13: 301–307.
Kotimäki J, Sorri M, Aantaa E, Nuutinen J . Prevalence of Meniere disease in Finland. Laryngoscope 1999; 109: 748–753.
Birgerson L, Gustavson KH, Stahle J . Familial Meniere's disease: a genetic investigation. Am J Otol 1987; 8: 323–326.
Oliveira CA, Braga AM . Meniere's syndrome inherited as an autosomal dominant trait. Ann Otol Rhinol Laryngol 1992; 101: 590–594.
Arweiler DJ, Jahnke K, Grosse-Wilde H . Meniere disease as an autosome dominant hereditary disease. Laryngorhinootologie 1995; 74: 512–515.
Morrison AW, Bailey ME, Morrison GA . Familial Meniere's disease: clinical and genetic aspects. J Laryngol Otol 2009; 123: 29–37.
Klockars T, Kentala E . Inheritance of Meniere's disease in the Finnish population. Arch Otolaryngol Head Neck Surg 2007; 133: 73–77.
Okafor BC . Incidence of Meniere's disease. J Laryngol Otol 1984; 98: 775–779.
Radtke A, Lempert T, Gresty MA, Brookes GB, Bronstein AM, Neuhauser H . Migraine and Meniere's disease: is there a link?. Neurology 2002; 59: 1700–1704.
Perez Lopez L, Belinchon de Diego A, Bermell Carrion A, Perez Garrigues H, Morera Perez C . Meniere's disease and migraine. Acta Otorrinolaringol Esp 2006; 57: 126–129.
Cha YH, Brodsky J, Ishiyama G, Sabatti C, Baloh RW . The relevance of migraine in patients with Meniere's disease. Acta Otolaryngol 2007; 127: 1241–1245.
Lempert T, Neuhauser H . Epidemiology of vertigo, migraine and vestibular migraine. J Neurol 2009; 256: 333–338.
Morrison AW . Anticipation in Meniere's disease. J Laryngol Otol 1995; 109: 499–502.
Fransen E, Van Camp G . The COCH gene: a frequent cause of hearing impairment and vestibular dysfunction?. Br J Audiol 1999; 33: 297–302.
Doi K, Sato T, Kuramasu T, et al. Meniere's disease is associated with single nucleotide polymorphisms in the human potassium channel genes, KCNE1 and KCNE3. ORL J Otorhinolaryngol Relat Spec 2005; 67: 289–293.
Kawaguchi S, Hagiwara A, Suzuki M . Polymorphic analysis of the heat-shock protein 70 gene (HSPA1A) in Meniere's disease. Acta Otolaryngol 2008; 128: 1173–1177.
Teggi R, Lanzani C, Zagato L, et al. Gly460Trp alpha-adducin mutation as a possible mechanism leading to endolymphatic hydrops in Meniere's syndrome. Otol Neurotol 2008; 29: 824–828.
Vrabec JT, Liu L, Li B, Leal SM . Sequence variants in host cell factor C1 are associated with Meniere's disease. Otol Neurotol 2008; 29: 561–566.
Lopez-Escamez JA, Moreno A, Bernal M, et al. Poly(ADP-ribose) polymerase-1 (PARP-1) longer alleles spanning the promoter region may confer protection to bilateral Meniere's disease. Acta Otolaryngol 2009; 129: 1222–1225.
Lopez-Escamez JA, Saenz-Lopez P, Acosta L, et al. Association of a functional polymorphism of PTPN22 encoding a lymphoid protein phosphatase in bilateral Meniere's disease. Laryngoscope 2010; 120: 103–107.
Klar J, Frykholm C, Friberg U, Dahl N . A Meniere's disease gene linked to chromosome 12p12.3. Am J Med Genet B Neuropsychiatr Genet 2006; 141B: 463–467.
Committee on Hearing and Equilibrium guidelines for the diagnosis and evaluation of therapy in Meniere's disease American Academy of Otolaryngology-Head and Neck Foundation, Inc. Otolaryngol Head Neck Surg 1995; 113: 181–185.
O'Connell JR, Weeks DE . PedCheck: a program for identification of genotype incompatibilities in linkage analysis. Am J Hum Genet 1998; 63: 259–266.
Kruglyak L, Daly MJ, Reeve-Daly MP, Lander ES . Parametric and nonparametric linkage analysis: a unified multipoint approach. Am J Hum Genet 1996; 58: 1347–1363.
Kotimäki J . Meniere's disease in Finland. An epidemiological and clinical study on occurrence, clinical picture and policy. Int J Circumpolar Health 2003; 62: 449–450.
Morrison AW, Johnson KJ . Genetics (molecular biology) and Meniere's disease. Otolaryngol Clin North Am 2002; 35: 497–516.
Stahle J . Advanced Meniere's disease. A study of 356 severely disabled patients. Acta Otolaryngol 1976; 81: 113–119.
Watanabe I . Meniere's disease in males and females. Acta Otolaryngol 1981; 91: 511–514.
Cha YH, Kane MJ, Baloh RW . Familial clustering of migraine, episodic vertigo, and Meniere's disease. Otol Neurotol 2008; 29: 93–96.
Gabriková D, Frykholm C, Friberg U, et al. Familial Meniere's disease restricted to 1.48 Mb on chromosome 12p. 12.3 by allelic and haplotype association. J Hum Genet 2010; 55: 834–837.
Acknowledgements
This work was supported by Grants from the Finnish Foundation for Ear Diseases and the Eemil Aaltonen Foundation. The authors thank the patients and their families for their participation and also Aira Erkkilä and Helena Satulehto for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclosure: The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Hietikko, E., Kotimäki, J., Kentala, E. et al. Finnish familial Meniere disease is not linked to chromosome 12p12.3, and anticipation and cosegregation with migraine are not common findings. Genet Med 13, 415–420 (2011). https://doi.org/10.1097/GIM.0b013e3182091a41
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1097/GIM.0b013e3182091a41
Keywords
This article is cited by
-
Genetic advances in Meniere Disease
Molecular Biology Reports (2023)
-
Molecular genetic studies of familial Meniere’s disease
Science China Life Sciences (2019)
-
M. Menière: Diagnostische Kriterien des Internationalen Klassifikationskomitees der Bárány-Gesellschaft
HNO (2017)