Abstract
Abstract. The osteochondrodysplasias, or skeletal dysplasias are a genetically heterogeneous group of over 350 distinct disorders, and many of them can present in the prenatal period as demonstrated by ultrasound. Differentiating these disorders in the prenatal period can be challenging because they are rare and many of the ultrasound findings are not necessarily pathognomic for a specific disorder. However, differentiating known lethal disorders from nonlethal disorders, providing differential diagnoses before delivery, determining postdelivery management plans and ultimately determining accurate recurrences risks to the at-risk couples improves patient care. These guidelines provide an approach to a fetus suspected of manifesting a skeletal dysplasia.
Similar content being viewed by others
Main
Diagnosis of prenatal-onset skeletal dysplasias can be accomplished by ultrasound evaluation and confirmed by both molecular testing using invasive procedures and postdelivery radiographs and autopsy, including histomorphic analysis of cartilage and bone. Obtaining a precise diagnosis by prenatal ultrasound diagnosis can be challenging. However, utilization of two and three-dimensional ultrasound can identify abnormal skeletal elements, and by analyzing the constellation of findings, a differential diagnosis can be achieved. These diagnoses based on the constellation of findings can be used for counseling regarding optimal patient management (both fetal and maternal) and calculation of recurrence risk. This policy statement discusses clinical guidelines for screening of fetuses suspected of manifesting a skeletal dysplasia or for pregnancies at-risk.
THE OSTEOCHONDRODYSPLASIAS (THE SKELETAL DYSPLASIAS)
The osteochondrodysplasias and dysostoses comprise a group of more than 350 disorders of the skeleton.1–3 By definition, the osteochondrodysplasias, or skeletal dysplasias, refer to disorders with generalized abnormalities of the skeleton, whereas the dysostoses are those disorders that have a single or group of abnormal bones. However, as more is known about all of these disorders, the distinction between osteochondrodysplasias and dysostoses has become blurred. In most osteochondrodysplasias, there is a generalized abnormality in linear skeletal growth and in some disorders there are concomitant abnormalities in organ systems other than the skeleton.4 The skeletal dysplasias can be inherited as autosomal dominant, autosomal recessive, or X-linked disorders, and some disorders that result from imprinting errors, somatic mosaicism, and teratogen exposure.3,5–8 There has been substantial progress in identification of the molecular defects responsible for the osteochondrodysplasias, and the genetic defects have been identified for approximately 160 of the 350 well-recognized disorders.3 Many of these discoveries have led to availability of DNA diagnostics for both molecular confirmation of ultrasound and postmortem findings, as well as invasive prenatal diagnosis for at-risk families.
Although the occurrence of each individual skeletal dysplasia may be rare,9 as a group they account for a significant number of newborns with congenital anomalies. Many of the prenatal onset skeletal dysplasias are associated with lethality because of pulmonary insufficiency or concomitant visceral abnormalities.10,4 Many of these disorders result from new dominant mutations and for the autosomal recessive disorders, many occur in families with no history of skeletal dysplasias.
The fetal skeleton develops relatively early in the fetal period and, thus, prenatal diagnosis of these disorders is possible. The appendicular and the axial skeleton undergo a programmed pattern of endochondral ossification, whereas the calvarium and portions of the clavicle and pubis ossify via membraneous ossification.11,12 Ossification occurs at relatively early human gestational ages: clavicle and mandible at 8 weeks; the appendicular skeleton, ileum, and scapula by 12 weeks; and the metacarpals and metatarsals are ossified by 12–16 weeks.13,14 Secondary (epiphyseal) ossification centers are seen by radiographs at approximately 20 weeks gestation and at a similar time period by ultrasound.
ULTRASOUND EVALUATION
Second trimester ultrasound evaluation of the fetus for detection of congenital anomalies has become standard of care in many communities.15,16 The fetal skeleton is readily visualized by two-dimensional ultrasound by 14 weeks, and measurements of the fetal femora and humeri are considered part of any basic midtrimester ultrasound evaluation. Any fetus showing femora or humeri length measurements less than 5th centile or −2 SD from the mean in the second trimester (<24 weeks) should be evaluated in a center that has expertise in evaluating the entire fetal skeleton and has the ability to provide genetic counseling. The following fetal ultrasound parameters must be visualized and plotted against normative values when a fetus manifesting a skeletal dysplasia is suspected (Table 1); fetal cranium (biparietal diameter, occipital-frontal diameter, and head circumference), abdominal circumference, mandible, clavicle, scapula, chest circumference, and all fetal long bones. Comparison of the relative length of all the long bones and against normative values will determine whether there is primarily rhizomelia, mesomelia, or that both segments are involved. One helpful ratio is the femur to foot ratio, which approaches 1.0 throughout gestation. Many skeletal dysplasias show disproportion based on those parameters.17 For example, those disorders that primarily present with rhizomelia in the prenatal period will show altered femur to foot ratio (<1). In addition to evaluation of the long bones, there are other ultrasound parameters that should be evaluated and can be helpful in these differentiating disorders (Table 1). These include the fetal facial profile (glabellar bossing, flattened nasal bridge, micrognathia; Fig. 1), presence and shape of the vertebral bodies (Table 2), and relative appearance of the hands and feet (extra, missing or malformed digits). There are many prenatal onset skeletal dysplasias that are associated with relative brachydactyly and equinovarus (Table 2). Fetuses with long bone measurements more than 3D below the mean should be strongly suspected of having a skeletal dysplasia, especially if the head circumference is greater than the 75th centile. Most prenatal-onset skeletal dysplasias present with relative disproportion of the skeletal measurements when compared with those of the cranium. In addition, close attention should be paid to the shape and mineralization pattern of the fetal calvarium and fetal skeleton (poor or ectopic mineralization). Determining the elements of the skeleton that are abnormal, coupled with the findings of mineralization and shape of the bones can aid in diagnosis (Table 2). Appropriate consultation with a geneticist or genetic counselor is recommended to assess the constellation of abnormalities and determine the most likely differential diagnoses. Prognosis and natural history can then be discussed using the most likely diagnoses as the basis for discussion.
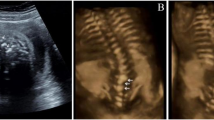
Three dimensional ultrasound images of fetuses demonstrating the abnormal facial images in thanatophoric dysplasia, achondrogenesis II and achondroplasia. A, Fetus with thanatophoric dysplasia showing severe nasal flattening and distorted facial shape because of craniosynostosis. B, Profile of fetus with thanatophoric dysplasia demonstrating severe nasal flattening and trident configuration of the hands (brachydactyly). C, Fetus with achondrogenesis II showing flattened nasal bridge but no distortion of facial shape. D, Fetus with achondroplasia showing mild flattening of the nasal bridge but not as severe as thanatophoric dysplasia.
One of the most important determinations that must be made by ultrasound is that of neonatal or infantile lethality. The definition of lethality can be a difficult one. Lethality occurs in most skeletal dysplasias as a result of a small chest circumference and resultant pulmonary hypoplasia. However, not all skeletal dysplasias associated with small thoracic circumferences are associated with immediate lethality. By using ultrasound criteria for lethality, chest-to-abdominal circumference ratio of <0.618 and femur length-to-abdominal circumference ratios of <0.16 are strongly suggestive of lethality.19 When concomitant abnormalities in other organ systems are visualized, there is increased morbidity and mortality in these disorders. It is important to note that the accuracy of prenatal diagnosis of the skeletal dysplasias using routine ultrasound approaches 40%20–26 and misdiagnosis can lead to inaccurate recurrence risk information and suboptimal management of the patients. Thus, all cases of prenatally diagnosed skeletal dysplasias should have a final diagnosis made by expert clinical and radiologic evaluation. When available, there is also a role for autopsy and histomorphic analysis of the cartilage growth plate, especially in cases of very rare skeletal dysplasias. It is critical to counsel families and their physicians to consider obtaining and storing tissue and/or DNA, especially because many skeletal dysplasias are associated with significant recurrence risk. Resources such as the International Skeletal Dysplasia Registry (http://www.csmc.edu/skeletaldysplasia) and the European Skeletal Dysplasia Network (http://www.esdn.org/) can be used to aid in final diagnosis.
IN-UTERO RADIOGRAPHY, FETAL MRI, AND THREE-DIMENSIONAL ULTRASONOGRAPHY
Historically, in-utero radiographs of a fetus suspected to have a skeletal dysplasia were used to confirm the ultrasound findings and help aid in counseling and patient management. It is our opinion that with marked improvements in ultrasound imaging in the last 10 years, the poor resolution of the radiographs obtained in utero, and since few centers have the expertise to analyze these radiographs, there is little value to this methodology. Fetal MRI can be useful for analysis of the fetal spine if there is a suggestion of severe scoliosis and there is concern for diastematomyelia or vertebral malformations. Three-dimensional ultrasound is helpful to distinguish facial abnormalities that may help differentiate these disorders (Fig. 1).27
MOLECULAR EVALUATION
The molecular defect has been identified in almost half of the well-recognized skeletal dysplasias. However, the application of these findings to direct patient care is not yet possible for many of these disorders. For families who have had a previously affected child with a molecularly confirmed diagnosis and are at-risk for recurrence, molecular analysis of DNA derived from either chorionic villus cells or amniocytes may be available, either by direct mutational analysis or by linkage analysis (GeneTests; www.genetests.org/). One controversial issue is whether patients who have had an affected child with a new dominant disorder should be offered invasive prenatal testing for a relatively low risk of germline mosaicism.
One direct application of molecular testing is in the case of pregnancies in which two affected parents have the same or different autosomal dominant skeletal disorders (nonassortative mating). This places the fetus at-risk for homozysosity or compound heterozygosity which is frequently associated with lethality, although there are exceptions.28 If both parents have achondroplasia, then testing the pregnancy for homozygosity or compound heterozygosity for mutations in FGFR3 is readily available, because 97% of patients with achondroplasia have a known identifiable mutation. If the parents have skeletal dysplasias associated with private mutations, they should be strongly encouraged to have molecular testing performed before pregnancy, since the time necessary for DNA diagnosis can be lengthy and not all mutations are identifiable, and consideration for linkage analysis may be necessary.
The role of molecular testing in a sporadically occurring skeletal dysplasia in an ongoing pregnancy is controversial. The time from invasive testing to diagnosis can be lengthy and may not change the outcome of the pregnancy, especially if the ultrasound appearance of the chest suggests lethality. A positive result will provide an answer and may affect care at time of delivery; however, failure to identify a mutation does not negate the ultrasound findings.
PREGNANCY AND MODE OF DELIVERY
With improvement in obstetrical care of both mother and infants, considerations need to be made for delivery of individuals affected by skeletal dysplasias. Advocacy groups for individuals with short stature (Little People of America and the Osteogenesis Imperfecta Foundation) have seen an increasing number of individuals affected by skeletal dysplasias undergo pregnancies with minimal complications, changing many professional opinions regarding outcome of pregnancies in short statured individuals. The issues surrounding pregnancy in short statured women include fetuses at-risk for homozygosity or compound heterozygosity, whether the mother is affected by a long-trunk versus short trunk skeletal dysplasia, need for operative delivery in most cases, regional versus general anesthesia, and optimal time for delivery. Although overall the data on these gestations is relatively limited,29 there are available resources (Little People of America; www.lpaonline.org/ Osteogenesis Imperfecta Foundation; www.oif.org/ and the International Skeletal Dysplasia Registry; www.csmc.edu/skeletaldysplasia), which can help provide some information and recommendations based on previous experiences.
In average stature women whose fetuses have a skeletal dysplasia there are multiple issues that need to be addressed before delivery. Many individuals whose fetuses have a genetic disorder or significant malformations choose not to continue the gestation to term. It is critical that fetuses presumed to have skeletal dysplasias that are delivered previably have appropriate postmortem evaluations, so subsequent counseling is based on accurate information. For those individuals who choose to continue the gestation to term, it is critical to assess the fetus for signs of possible lethality (diminished femur length to abdominal circumference ratio, presence of hydrops fetalis, severe polyhydramnios, visceral abnormalities) versus those fetuses that seem to have skeletal disorders not usually associated with lethality. Predelivery consultations and development of a delivery and resuscitation plan with clinical geneticists, neonatalogists, obstetricians, and anesthesiologists improve the postnatal management of these fetuses. Many fetuses with nonlethal skeletal dysplasias can have some respiratory compromise in the immediate newborn period. Further, obstetricians should be made aware that many fetuses with both lethal and nonlethal skeletal dysplasias delivered at or near term manifest relative macrocephaly and vaginal delivery may not be readily accomplished.
RECOMMENDATIONS
-
1
Fetuses with long bone measurements at or less than the 5th centile or >3 SD below the mean should be evaluated in a center with expertise in the recognition of skeletal dysplasias. If the patient cannot travel, arrangements may be able to be made for evaluation of ultrasound videotapes or hard copy images.
-
2
The following fetal ultrasound measurements should be visualized and plotted against normative values: fetal cranium (biparietal diameter and head circumference), facial profile, mandible, clavicle, scapula, chest circumference, vertebral bodies, all fetal long bones, and the hands and feet. Fetuses with long bone parameters >3 SD below the mean should be strongly suspected of having a skeletal dysplasia, especially if the head circumference is greater than the 75th centile (Table 1).
-
3
Lethality should be determined by chest circumference to abdominal circumference ratio and/or femur length to abdominal circumference measurement ratio. A chest- to-abdominal circumference ratio of <0.6 or femur length to abdominal circumference ratio of 0.16 strongly suggests a perinatal lethal disorder, although there are exceptions. The findings should be conveyed to the physicians caring for the patient and to the patient.
-
4
Molecular testing should be offered in those pregnancies at-risk for homozygosity or compound heterozygosity for skeletal dysplasias. Both parents' mutations should have been identified, ideally before pregnancy.
-
5
Individuals with skeletal dysplasias known to be due to a number of different mutations should be encouraged to obtain molecular analysis before pregnancy.
-
6
In cases where molecular testing is performed and ultrasound findings suggest a lethal prognosis, then counseling should be based on clinical findings and molecular testing should be considered to confirm the clinical findings.
-
7
All fetuses suspected of having a skeletal dysplasia should have the diagnosis confirmed by postdelivery clinical and radiologic evaluation. Postdelivery and/or postmortem evaluation includes anterior-posterior radiographs of the appendicular skeleton including hands and feet, and anterior-posterior and lateral radiographs of the cranium and spine (vertebral column). In all appropriate cases, photographs should be taken and autopsies should be offered and encouraged, because it provides the most useful information for accurate diagnosis. Pathologists should collect cartilage and bone, ideally femora and humeri for histomorphic analysis. Tissue (fibroblasts, cartilage and bone) and/or DNA should be saved for molecular analysis whenever possible, because many skeletal disorders are associated with a significant recurrence risk.
-
8
Mothers who themselves have skeletal dysplasias need consultation with obstetricians and anesthesiologists regarding optimal management, including mode of delivery.
-
9
For fetuses suspected as having a skeletal dysplasia delivered at a viable gestational age, predelivery consultations and management plan should be initiated between the obstetrical, neonatal, anesthesia, and genetics consultants.
-
10
Resources such as the International Skeletal Dysplasia Registry, the European Skeletal Dysplasia Network and other qualified genetic centers should be used whenever possible, including the proper collection of autopsy material, especially if the diagnosis and counseling are uncertain.
REFERENCES
Superti-Furga A, Bonafé L, Rimoin DL . Molecular-pathogenetic classification of genetic disorders of the skeleton. Am J Med Genet 2001; 106: 282–293.
Hall CM . International nosology and classification of constitutional disorders of bone (2001). Am J Med Genet 2002; 113: 65–77.
Superti-Furga A, Unger S . Nosology and classification of genetic skeletal disorders: 2006 revision. Am J Med Genet A 2007; 143: 1–18.
Lachman RS, editor. Taybi and Lachman's radiology of syndromes, metabolic disorders and skeletal dysplasias. Philadelphia: Mosby Elsevier 2007;
Sutton VR, McAlister WH, Bertin TK, et al. Skeletal defects in paternal uniparental disomy for chromosome 14 are re-capitulated in the mouse model (paternal uniparental disomy 12). Hum Genet 2003; 113: 447–451.
Walter CA, Shaffer LG, Kaye CI, et al. Short-limb dwarfism and hypertrophic cardiomyopathy in a patient with paternal isodisomy 14: 45, XY, idic(14)(p11). Am J Med Genet 1996; 65: 259–265.
Shanske AL, Bernstein L, Herzog R . Chondrodysplasia punctata and maternal autoimmune disease: a new case and review of the literature. Pediatrics 2007; 120: e436–e441.
Savarirayan R . Common phenotype and etiology in warfarin embryopathy and X-linked chondrodysplasia punctata (CDPX). Pediatr Radiol 1999; 29: 322.
Orioli IM, Castilla EE, Barbosa-Neto JG . The birth prevalence rates for the skeletal dysplasias. J Med Gene 1986; 23: 328–332.
Spranger JW, Brill PW, Poznaski A, editor. Bone dysplasias. New York: Oxford University Press, 2002.
Eames BF, de la Fuente L, Helms JA . Molecular ontogeny of the skeleton. Birth Defects Res C Embryo Today 2003; 69: 93–101.
Olsen BR, Reginato AM, Wang W . Bone development. Annu Rev Cell Dev Biol 2000; 6: 191–220.
Larson WJ, Sherman LS, Potter SS, Scott WJ . Human embryology. NewYork: Churchill Livingstone 1993; 281–310.
van Zalen-Sprock RM, Brons JT, van Vugt JM, van der Harten HJ, van Geijn HP . Ultrasonographic and radiologic visualization of the developing embryonic skeleton. Ultrasound Obstet Gynecol 1997; 9: 392–397.
Bahado-Singh RO, Raymond M . The 20-week scan: beyond biometry and anatomy. Clin Obstet Gynecol 2007; 50: 478–486.
Saltvedt S, Almström H, Kublickas M, Valentin L, Grunewald C . Detection of malformations in chromosomally normal fetuses by routine ultrasound at 12 or 18 weeks of gestation-a randomised controlled trial in 39,572 pregnancies. BJOG 2006; 113: 664–674.
Campbell J, Henderson A, Campbell S . The fetal femur/foot length ratio: a new parameter to assess dysplastic limb reduction. Obstet Gynecol 1988; 72: 181–184.
Yoshimura S, Masuzaki H, Gotoh H, Fukuda H, Ishimaru T . Ultrasonographic prediction of lethal pulmonary hypoplasia: comparison of eight different ultrasonographic parameters. Am J Obstet Gynecol 1996; 175: 477–483.
Rahemtullah A, McGillivray B, Wilson RD . Suspected skeletal dysplasias: femur length to abdominal circumference ratio can be used in ultrasonographic prediction of fetal outcome. Am J Obstet Gyneco 1997; 177: 864–869.
Parilla BV, Leeth EA, Kambich MP, Chilis P, MacGregor SN . Antenatal detection of skeletal dysplasias. J Ultrasound Med 2003; 22: 255–258.
Doray B, Favre R, Viville B, Langer B, Dreyfus M, Stoll C . Prenatal sonographic diagnosis of skeletal dysplasias. A report of 47 cases. Ann Genet 2000; 43: 163–169.
Gaffney G, Manning N, Boyd PA, Rai V, Gould S, Chamberlain P . Prenatal sonographic diagnosis of skeletal dysplasias–a report of the diagnostic and prognostic accuracy in 35 cases. Prenat Diagn 1998; 18: 357–362.
Tretter AE, Saunders RC, Meyers CM, et al. Antenatal diagnosis of lethal skeletal dysplasias. Am J Med Gene 1998; 75: 518–522.
Rasmussen SA, Bieber FR, Benacerraf BR, Lachman RS, Rimoin DL, Holmes LB . Epidemiology of osteochondrodysplasias: changing trends due to advances in prenatal diagnosis. Am J Med Genet 1996; 61: 49–58.
Sharony R, Browne C, Lachman RS, Rimoin DL . Prenatal diagnosis of the skeletal dysplasias. Am J Obstet Gynecol 1993; 169: 668–675.
Donnenfeld AE, Mennuti MT . Second trimester diagnosis of fetal skeletal dysplasias. Obstet Gynecol Surv 1987; 42: 199–217.
Krakow D, Williams J, Poehl M, Rimoin DL, Platt LD . Use of three-dimensional ultrasound imaging in the diagnosis of prenatal-onset skeletal dysplasias. Ultrasound Obstet Gynecol 2003; 21: 467–472.
Unger S, Korkko J, Krakow D, Lachman RS, Rimoin DL, Cohn DH . Double heterozygosity for pseudoachondroplasia and spondyloepiphyseal dysplasia congenita. Am J Med Genet 2001; 104: 140–146.
Allanson JE, Hall JG . Obstetric and gynecologic problems in women with chondrodystrophies. Obstet Gynecol 1986; 67: 74–78.
Acknowledgements
D.L.R., R.S.L., and D.K. are supported by NIH HD22657 and the NIH-sponsored General Clinical Research Center Grant (M01-RR00425). D.K. is also supported by the Joseph Drown Foundation and is a Winnick Family Scholar awardee. The authors thank both the patients and their physicians who provided us with the experience necessary to develop recommendations.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclosure: The authors declare no conflict of interest.
Disclaimer: This guideline is designed primarily as an educational resource for health care providers to help them provide qualitymedical genetic services. Adherence to this guideline does not necessarily assure a successful medical outcome. This guideline should not be considered inclusive of all proper procedures and tests or exclusive of other procedures and tests that are reasonably directed to obtaining the same results. In determining the propriety of any specific procedure or test, the geneticist should apply his or her own professional judgment to the specific clinical circumstances presented by the individual patient or specimen. It may be prudent, however, to document in the patient's record the rationale for any significant deviation from this guideline.
Rights and permissions
About this article
Cite this article
Krakow, D., Lachman, R. & Rimoin, D. Guidelines for the prenatal diagnosis of fetal skeletal dysplasias. Genet Med 11, 127–133 (2009). https://doi.org/10.1097/GIM.0b013e3181971ccb
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1097/GIM.0b013e3181971ccb
Keywords
This article is cited by
-
Using MRI-derived observed-to-expected total fetal lung volume to predict lethality in fetal skeletal dysplasia
Pediatric Radiology (2023)
-
A case report of achondrogenesis type II (Langer-Saldino achondrogenesis)
Egyptian Journal of Radiology and Nuclear Medicine (2021)
-
Prenatal diagnosis of a novel pathogenic variation in the ACAN gene presenting with isolated shortening of fetal long bones in the second trimester of gestation: a case report
BMC Pregnancy and Childbirth (2021)
-
Development and validation of an expanded targeted sequencing panel for non-invasive prenatal diagnosis of sporadic skeletal dysplasia
BMC Medical Genomics (2021)
-
Prenatal diagnosis of fetal skeletal dysplasia using 3-dimensional computed tomography: a prospective study
BMC Musculoskeletal Disorders (2020)