Abstract
Purpose: To elucidate whether thrombocytopenia in 22q11.2 deletion syndrome patients is associated with the hemizygosity of glycoprotein Ib-β and to clarify the correlation of phenotype and genotype of this gene in 22q11.2 deletion syndrome patients with thrombocytopenia.
Methods: Platelet number, mean platelet volume, platelet agglutination, and the protein level of glycoprotein Ib-β were measured in 22q11.2 deletion syndrome patients and controls. Phenotypes other than that of thrombocytopenia were also analyzed in these patients.
Results: The 22q11.2 deletion syndrome patients with thrombocytopenia had a larger mean platelet volume, lower agglutination to ristocetin, and lower protein level of glycoprotein Ib-β than control patients. The 22q11.2 deletion syndrome patients with thrombocytopenia showed an increased risk of developing schizophrenia.
Conclusions: Thrombocytopenia in 22q11.2 deletion syndrome patients is associated with decreased expression of glycoprotein Ib-β because of the hemizygosity. 22q11.2 deletion syndrome patients with thrombocytopenia require total management, especially for schizophrenia.
Similar content being viewed by others
Main
The 22q11.2 deletion syndrome is the most common microdeletion syndrome, occurring with a frequency of 1/4000 in Europe1 and 1/5000 in Japan, as estimated from previous reports.2 The terminology for this condition is confusing as a number of other names have been used to describe this pattern of malformation, including DiGeorge syndrome,3,4 conotruncal anomaly face syndrome (CAFS),5–7 and velocardiofacial syndrome (VCFS).8–10 Patients with 22q11.2 deletion syndrome show a wide spectrum of anomalies, among which are typical faces (short palpebral fissures, puffy eyelids, a low nasal bridge, small mouth, and minor ear anomalies), velopharyngeal insufficiency with or without an overt cleft, thymic abnormalities, hypocalcemia, mental retardation, and scoliosis.11–13 Conotruncal cardiac anomalies are common and include tetralogy of Fallot with or without pulmonary atresia, interruption of the aortic arch, truncus arteriosus communis, and associated cardiovascular anomalies, including aortic arch anomalies.11–13 In our previous studies, a significant number of affected individuals appeared to have thrombocytopenia and/or schizophrenia.11
To date, an analysis of the deleted region of chromosome 22q11.2, elucidating more than 30 genes in this region, has been reported. However, association of these genes with the cause of this syndrome is not yet clearly understood. Glycoprotein Ib-β (GPIb-β) (of which the transcript is a subunit of GPIb, a major component of the platelet membrane receptor for von Willebrand factor, designated GPIb-IX-V complex), has been located in the deleted region.14–16 Homozygous mutation of GPIb-β causes Bernard-Soulier syndrome (BSS), a rare congenital bleeding disorder with autosomal recessive inheritance.17 BSS is characterized by prolonged bleeding time, thrombocytopenia with morphologically enlarged platelets, and an isolated defect in ristocetin-induced agglutination. Patients with BSS usually show bleeding tendencies from early childhood. This syndrome is genetically heterogeneous and can also be caused by mutations of GPIb-α and GPIX. These genes are located on chromosome 17pter-p1218 and on chromosome 3,19 respectively.
It is not known whether the thrombocytopenia in the 22q11.2 deletion syndrome is associated with hemizygosity of GPIb-β or congenital heart disease. We explored this issue by comparing various examinations of platelet structure and function among a group of 22q11.2 deletion syndrome patients with cardiac defects and a control population without the deletion but matched for cardiac abnormalities. The status of GPIb-β was determined in a subset of patients using fluorescence in situ hybridization (FISH), sequencing, and Western blot analysis to quantitate protein levels. Furthermore, we clarified the correlation between the phenotype and genotype of this gene in 22q11.2 deletion syndrome patients with thrombocytopenia.
PATIENTS AND METHODS
Clinical characteristics
Among patients who visited our institution for evaluation of cardiac disease between April 1994 and March 2000, we studied 99 patients with 22q11.2 deletion syndrome (del+) and 124 control patients with similar conotruncal heart anomalies but without the deletion (del−). All 99 del+ patients had conotruncal anomaly face, which is the typical phenotype of 22q11.2 deletion syndrome, and 97 of them had cardiovascular disease. The presence of the syndrome was confirmed by FISH analysis with the N25 probe (Fig. 1b). All 223 patients gave informed consent for enrollment in our study. The study was approved by the internal ethics committee of Tokyo Women’s Medical University. The characteristics of the patients are shown in Table 1. Patients with schizophrenia were diagnosed by psychiatrists according to the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV).20 Patients with decreased platelets (<150 × 109/L) were considered to have thrombocytopenia (del+: 38 patients, del−: 25 patients).
a: Markers in chromosome 22q11.2. N72H9 (D22S181), sc11.1a, N25DGCR (D22S75), C443 (D22S941), sc4.1 (D22S134), sc11.1b (D22S139), N19B3 (D22S264), N122B5 (D22S934), and N77F7(D22S939) are probes for FISH.11 The location of GPIb-β is also indicated. b: The type of deletion in chromosome 22q11.2. The type of deletion was classified according to telomeric deletion breakpoints. Bars with open ellipses indicate the deleted region in each type. Open ellipses are aligned with the loci on the bar with closed ellipses, which illustrates the chromosome schematically. The location of GPIb-β is indicated with an arrow.
Examination of bleeding time, platelet volume, and platelet function
Twelve of the 38 del+ patients and 5 of the 25 del− patients with thrombocytopenia who gave informed consent were examined with Ivy’s bleeding time, mean platelet volume (MPV) and platelet agglutination induced by ADP (2.0, 4.0, 8.0 μM), collagen (0.5, 1.0, 5.0 μg/mL), arachidonic acid (280, 420, 1090 μM), and ristocetin (0.5, 1.0, 1.2, 1.42 mg/mL). The results of the platelet agglutination were obtained by percent aggregation according to Kunishima et al.21 When it lasted more than 9 minutes, bleeding time was considered to be prolonged.
FISH analysis
Human metaphase chromosome slides were prepared from Epstein-Barr virus–transformed lymphoblastoid cell lines or peripheral blood by standard methods.22,23 FISH analysis was then performed to determine the deletion size in the 99 del+ patients, using the same probes as previously described (Fig. 1, a and b).11 According to the telomeric deletion breakpoints, we classified the deletion type as shown in Figure 1b.11 We also determined whether the 38 del+ patients with thrombocytopenia were hemizygous for GPIb-β by FISH analysis using a plasmid which contained normal GPIb-β as the probe. At least 20 metaphases from each patient were analyzed.
Sequence analysis
In the 38 del+ patients with thrombocytopenia, the GPIb-β gene in the remaining allele was sequenced. Genomic DNA samples were extracted from Epstein-Barr virus–transformed lymphoblastoid cell lines. Polymerase chain reaction (PCR) primers were designed as follows to amplify the GATA-binding site and all the exons of GPIb-β on the basis of the genomic sequence reported by Yagi et al.15 (GenBank accession number AF006988): GPIbβ-F3: 5′-GAA TGC CGC GTC CTG TCC TGG TGA-3′ and GPIb-β-R3: 5′-AGG TGG GGT GGG TCT GAG AGA TTG G-3′. The DNA fragment was amplified by PCR followed by sequence analysis with a Thermo Sequence Sequencing Kit (Amersham Pharmacia, Buckinghamshire, England) and A.L.F. DNA Sequencer II (Amersham Pharmacia).
Western blotting
Platelets from 12 del+ patients and 5 del− patients with thrombocytopenia were prepared and solubilized as described previously.24 The lysates were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). Increasing amounts of platelet lysates from normal individuals without a deletion in chromosome 22q11.2, cardiovascular disease, or thrombocytopenia were also subjected to SDS-PAGE. After blocking, the nitrocellulose membrane (Schleicher & Schuell, Keene, NH) was incubated with goat polyclonal antibody to human platelet GPIb-β (Santa Cruz Biotechnology, Santa Cruz, CA) and then incubated with a horseradish peroxidase–conjugated donkey antibody to goat IgG (Santa Cruz Biotechnology). As control, we also performed Western blotting for platelet factor 4 (PF4), a protein packaged in α granules of platelets. Rabbit polyclonal antibody to human PF4 (Chemicon, Temecula, CA) and a horseradish peroxidase–conjugated goat antibody to rabbit IgG (IBL, Fujioka, Japan) were used as the primary and the secondary antibody, respectively. The membrane was treated with ECL plus (Amersham Pharmacia) and then was analyzed quantitatively using the public domain NIH image program. The intensity of the hybridization signal was adjusted according to the PF4 intensity of each lane.
Statistics
Dichotomous variables were compared using the Fisher exact probability test. The Mann-Whitney U test was used to compare two mean values of variables. Data are shown as mean ± SD. The level of significance was set at 0.05.
RESULTS
Clinical characteristics
Thirty-eight del+ patients and 25 del− patients showed thrombocytopenia (Fig. 2). Del+ patients were at a significantly increased risk of thrombocytopenia compared with del− patients [relative risk (RR) = 1.90, 95% confidence interval (CI) = 1.24–2.93, P < 0.05, Table 2]. Age did not differ significantly between the del+ patients with thrombocytopenia and the del− patients with thrombocytopenia [19.7 ± 8.5 vs. 20.4 ± 8.8, not significant (NS)]. One del+ patient with thrombocytopenia was diagnosed with idiopathic thrombocytopenic purpura. Fourteen del+ patients with thrombocytopenia and 12 del− patients with thrombocytopenia did not receive surgical repair for their cardiovascular anomalies because they were inoperable cases or palliative operation cases (37% vs. 48%, NS). Four del+ patients with thrombocytopenia and 2 del− patients with thrombocytopenia had heart failure (11% vs. 8%, NS). Three del+ patients with thrombocytopenia and 7 del− patients with thrombocytopenia had infectious disease (8% vs. 28%, P < 0.05). Four of the 38 del+ patients with thrombocytopenia showed mild bleeding tendencies. Their symptoms included paranasal sinus hematoma, hemarthrosis induced by minor trauma, and epistaxis.
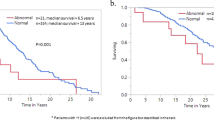
Ten of the 99 del+ patients had schizophrenia (Fig. 2). The clinical characteristics of these 10 patients are described in Table 3. Of interest, 8 of these 10 patients had thrombocytopenia and the 2 other patients had mild thrombocytopenia (Patients 9 and 10 had platelet numbers of 178 × 109/L and 187 × 109/L, respectively). The thrombocytopenia was not drug-induced in these 10 patients. Eight of them were reported previously.25 The mean age at onset of the schizophrenia was 23.1 ± 5.9 years. In 5 of the 8 del+ patients with thrombocytopenia and schizophrenia, the thrombocytopenia appeared earlier than the schizophrenia. Patients 1, 5, and 7 did not have a blood examination around the onset of the schizophrenia, so we could not determine whether it was preceded by thrombocytopenia in these three patients. Del+ patients with thrombocytopenia showed a significantly increased risk of developing schizophrenia compared with del+ patients without thrombocytopenia (RR = 6.42, 95% CI = 1.44–28.66, P < 0.05, Table 4). Two patients with schizophrenia had no cardiovascular anomalies. In del− patients, only one female patient had a conversion disorder and her age at onset was 25 years. She had truncus arteriosus communis and did not have thrombocytopenia.
Examination of bleeding time, platelet volume, and platelet function
In 12 of the 38 del+ patients and 5 of the 25 del− patients with thrombocytopenia, Ivy’s bleeding time was measured and platelets obtained from these 17 patients were subjected to analysis of platelet volume and platelet function. Six of the 12 del+ patients and 1 of the 5 del− patients had prolonged bleeding time of more than 9 minutes (50% vs. 20%, NS). MPV was greater in the del+ patients than in the del− patients (10.2 ± 0.5 fL vs. 9.3 ± 0.3 fL, P < 0.05). No patient had defective platelet agglutination to ristocetin as seen in BSS patients. However, the del+ patients showed a lower percent agglutination to 1.0 mg/mL of ristocetin than the del− patients (14.1 ± 25.3% vs. 54.4 ± 40.1%, P < 0.05). The del+ patients showed a higher percent agglutination to 0.5 μg/mL of collagen than the del− patients (62.8 ± 28.1% vs. 28.6 ± 33.4%, P < 0.05, Table 5). Platelet agglutination induced by ADP and arachidonic acid did not show a significant difference between the del+ and del− patients (data not shown).
FISH analysis
We determined the deletion size of chromosome 22q11.2 in the 99 del+ patients (Fig. 3). Among the 38 del+ patients with thrombocytopenia, 34 patients (89%) had a distal breakpoint (type A), 2 (5%) had an intermediate breakpoint (type B), and 2 (5%) had a proximal breakpoint (type C). The region which encodes GPIb-β was included in the deleted region in these patients. Ten patients with schizophrenia had a distal breakpoint (type A). Nine of these patients had a type A1 deletion and the other patient had a type A2 deletion (Table 3, Fig. 3).
Type of deletion in the 22q11.2 deletion syndrome patients. Bars with open ellipses indicate the deleted region. Open ellipses are aligned with the loci on the bar with closed ellipses, which indicates the chromosome schematically. Numbers of del+ patients (n = 99), del+ patients with thrombocytopenia (n = 38), and del+ patients with schizophrenia (n = 10) for each type of deletion are shown. The location of GPIb-β is indicated. The potential involvement of COMT,34PRODH,35 and CDCrel-1 in psychotic disorders has been suggested, so the location of these genes is also shown.
In the 38 del+ patients with thrombocytopenia, we performed FISH analysis using a plasmid containing GPIb-βcDNA as the probe. All these patients had hemizygosity for GPIb-β (Fig. 4).
A metaphase spread from a patient with 22q11.2 deletion syndrome, showing FISH mapping of the control probe to 22q13.3 (red arrow) and plasmid including GPIb-β to 22q11.2 (green arrow). On the normal chromosome, a green signal (green arrow) can be seen at 22q11.2 and a red signal (red arrow) can be seen at 22q13.3. On the deleted chromatic chromosome, only a red signal (red arrow) can be seen at 22q13.3.
Sequence analysis
After we determined that the 38 del+ patients with thrombocytopenia had hemizygosity for GPIb-β, we sequenced the GPIb-β gene in the homologous allele in these patients. Our sequence analysis covered all the exons and the GATA-binding site of GPIb-β. No mutation was found in GPIb-β in the homologous allele of these patients.
Western blotting
We quantified the protein levels of GPIb-β in the platelets of 12 of the 38 del+ patients with thrombocytopenia and 5 of the 25 del− patients with thrombocytopenia who were analyzed by Ivy’s bleeding time, MPV, and platelet function. As control, the protein levels of PF4 in the platelets of the same patients were quantified (Fig. 5). The protein levels of GPIb-β in the del+ patients was about half of that in del− patients.
Western blotting analysis of GPIb-β and PF4 from the platelets of del+ and del− patients. The molecular weight of GPIb-β and PF4 was 25 kD and 7.8 kD, respectively. Top: Platelet lysates (5 μg) from del+ and del− patients were subjected to SDS-PAGE, transferred to a nitrocellulose membrane, and probed with antibody to human platelet GPIb-β. Increasing amounts (2 μg, 5 μg, and 8 μg) of platelet lysates from a normal control were also subjected to Western blotting analysis. Bottom: Platelet lysates (0.5 μg) from del+ and del− patients were also subjected to Western blotting analysis using antibody to human PF4. The intensity of the hybridization signal of GPIb-β was adjusted according to the intensity of the PF4 signal.
DISCUSSION
Our results revealed that 22q11.2 deletion syndrome patients tended to show thrombocytopenia and enlarged platelets and slightly decreased platelet agglutination to ristocetin. Prolonged bleeding time was also more frequently seen in del+ patients than in del− patients. Van Geet et al.26 reported that patients with 22q11.2 deletion syndrome had an increase in platelet size and a mild decrease in platelet number. However, their results did not show whether thrombocytopenia in 22q11.2 deletion syndrome was due to hemizygosity of GPIb-β or conotruncal heart anomalies. It has been reported that patients with congenital heart disease sometimes show thrombocytopenia.27 There is an inverse relationship between platelet number and hemoglobin in patients with congenital heart disease.27 However, our results showed no significant difference in hemoglobin between the del+ and del− patients (Table 1). In addition, clinical episodes which can affect platelet number, such as heart failure, were not significantly different between the del+ and del− patients with thrombocytopenia. Infectious disease, which can cause thrombocytopenia, was more common in the del− patients with thrombocytopenia than in the del+ patients with thrombocytopenia. These results suggest that the greater frequency of thrombocytopenia in 22q11.2 deletion syndrome patients than in del− patients with a similar conotruncal heart anomaly is not due to a conotruncal heart anomaly.
The platelet membrane GPIb-IX-V complex acts as a receptor on the platelet surface for binding von Willebrand factor and plays a major role in primary hemostasis. This complex is composed of four kinds of subunits: GPIb-α, GPIb-β, GPIX, and GPV. Homozygous mutation of the GPIb-α, GPIb-β, or GPIX subunits results in BSS, characterized by moderate to severe thrombocytopenia, giant platelets, and defective platelet agglutination to ristocetin.17GPIb-β was mapped to chromosome 22q11.2.14–16 Among reported cases of mutation in GPIb-β, patients with associated 22q11.2 deletion syndrome were reported.14,28,29 Budarf et al.14 reported a BSS patient with 22q11.2 deletion syndrome. This patient was a compound heterozygote with the deletion of one allele of GPIb-β and a mutation in a GATA-binding site of GPIb-β in the remaining allele.28 Kenny et al.29 reported a single nucleotide deletion which leads to premature truncation of GPIb-β by a translational frameshift.
In the 38 del+ patients with thrombocytopenia, we confirmed hemizygosity for GPIb-β by FISH analysis. We also sequenced their GPIb-β, including a GATA-binding site in the homologous allele, and no mutation was detected. In 12 del+ patients with thrombocytopenia and 5 del− patients with thrombocytopenia, we quantified the protein levels of GPIb-β by Western blotting. In the 12 del+ patients with thrombocytopenia, the protein levels of GPIb-β were not defective but reduced. The laboratory data showed that 38% of the 22q11.2 deletion syndrome patients had thrombocytopenia (Table 2), and in the del+ patients with thrombocytopenia, a significantly greater MPV and a lower agglutination to ristocetin than in del− patients with thrombocytopenia were detected (Table 5). However, a patient with a homozygous mutation for the GPIb-IX-V complex who does not carry a phenotype of BSS, such as thrombocytopenia, has been reported.30 Therefore, we believe that hemizygosity for GPIb-β may not show full penetrance. The degree of thrombocytopenia may also be influenced by another mechanism besides reduced levels of GPIb-β. In this study, examinations concerning platelet phenotype were performed to the extent of the patient’s informed consent. Our results of genetic analysis for GPIb-β and laboratory data suggest that the characteristics of 22q11.2 deletion syndrome patients are associated with a reduced level of GPIb-β. The 22q11.2 deletion syndrome should be considered as a differential diagnosis of thrombocytopenia.
Previous studies have suggested that patients with 22q11.2 deletion syndrome were at an increased risk for developing a psychotic disorder.11–13,24,31–33 Pulver et al.32 reported that the prevalence of schizophrenia among their CAFS/VCFS patients was 29%, which was much higher than in the general population (1%). Bassett et al.33 reported that 5 of 10 adults with schizophrenia or schizoaffective disorder who had 22q11.2 deletion syndrome showed either mild thrombocytopenia or recurrent epistaxis. In our study, the del+ patients with thrombocytopenia were at increased risk of developing schizophrenia. In these patients, the onset of schizophrenia was observed during adulthood (23.1 ± 5.9 years) and it tended to be preceded by thrombocytopenia. Loebel et al.34 suggested that the duration of psychotic symptoms before treatment significantly affects the prognosis of schizophrenic patients. Therefore, early intervention is important for 22q11.2 deletion syndrome patients with schizophrenia and the presence of thrombocytopenia in such patients may be helpful for early recognition of impending schizophrenia.
The deleted region in chromosome 22q11.2 contains candidate genes for schizophrenia, including catechol-O-methyl-transferase (COMT) and proline dehydrogenase (PRODH) (Fig. 3), but the association of these genes with schizophrenia has not been proven.35,36 In our classification, the A1 type but not the A2 type of the deleted region includes PRODH. However, patients with schizophrenia were seen in both A1 and A2 types. To analyze whether COMT and PRODH play a role in inducing schizophrenia, further investigation is needed. In addition, CDCrel-1 is located adjacent to the locus of GPIb-β (Fig. 3) and its isoforms include a readthrough transcript containing the GPIb-β coding region.37,38CDCrel-1 has been reported to be associated with synaptic vesicles and the inhibition of exocytosis by means of binding to syntaxin.39 Whether CDCrel-1 is involved in the pathogenesis of schizophrenia also requires further investigation.
In conclusion, del+ patients were at a significantly increased risk for thrombocytopenia compared with del− patients. Our results suggest that their phenotype, such as giant platelets, thrombocytopenia and slightly impaired platelet agglutination to ristocetin, is associated with the reduced level of GPIb-β protein due to the hemizygosity. The 22q11.2 deletion syndrome should be considered as a cause of thrombocytopenia. Del+ patients with thrombocytopenia were at a significantly increased risk for developing schizophrenia compared with del+ patients without thrombocytopenia. When 22q11.2 deletion syndrome patients show thrombocytopenia, total management is needed, especially for those with increased risk of developing schizophrenia.
References
Burn J, Wilson DI, Cross I, Atif U, Scambler P, Takao A et al. The clinical significance of 22q11 deletion. In: Clark EB, Markwald RR, Takao A, editors. Developmental mechanisms of heart disease. New York: Futura, 1995: 559–567.
Kinouchi A, Mori K, Ando M, Takao A . Facial appearance of patients with conotruncal anomalies. Pediatr Jpn 1976; 17: 84–84.
DiGeorge AM . Discussion on a new concept of the cellular basis of immunity. J Pediatr 1965; 67: 907–908.
de la Chapelle A, Herva R, Koivisto M, Aula P . A deletion in chromosome 22 can cause DiGeorge syndrome. Hum Genet 1981; 57: 253–256.
Takao A, Ando M, Cho K, Kinouchi A, Murakami Y . Etiologic categorization of common congenital heart disease. In: Van Praagh R, Takao A, editors. Etiology and morphogenesis of congenital heart disease. New York: Futura, 1980: 253–269.
Burn J, Takao A, Wilson D, Cross I, Momma K, Wadey R . Conotruncal anomaly face syndrome is associated with a deletion within chromosome 22q11. J Med Genet 1993; 30: 822–824.
Matsuoka R, Takao A, Kimura M, Imamura S, Kondo C, Joh-o K . et al. Confirmation that the conotruncal anomaly face syndrome is associated with a deletion within 22q11.2. Am J Med Genet 1994; 53: 285–289.
Shprintzen RJ, Goldberg RB, Lewin ML, Sidoti EJ, Berkman MD, Argamaso RV et al. A new syndrome involving cleft palate, cardiac anomalies, typical faces and learning disabilities: velo-cardio-facial syndrome. Cleft Palate J 1978; 5: 56–62.
Driscoll DA, Spinner NB, Budarf ML, McDonald-McGinn DM, Zackai EH, Goldberg RB et al. Deletions and microdeletions of 22q11.2 in velo-cardio-facial syndrome. Am J Med Genet 1992; 44: 261–268.
Scambler PJ, Kelly D, Lindsay E, Williamson R, Goldberg R, Shprintzen R et al. Velo-cardio-facial syndrome associated with chromosome 22 deletions encompassing the DiGeorge locus. Lancet 1992; 339: 1138–1139.
Matsuoka R, Kimura M, Scambler PJ, Morrow BE, Imamura S, Minoshima S et al. Molecular and clinical study of 183 patients with conotruncal anomaly face syndrome. Hum Genet 1998; 103: 70–80.
Goldberg R, Motzkin B, Marion R, Scambler PJ, Shprintzen RJ . Velo-cardio-facial syndrome: a review of 120 patients. Am J Med Genet 1993; 45: 313–319.
Ryan AK, Goodship JA, Wilson DI, Philip N, Levy A, Seidel H et al. Spectrum of clinical features associated with interstitial chromosome 22q11 deletions: a European collaborative study. J Med Genet 1997; 34: 798–804.
Budarf ML, Konkle BA, Ludlow LB, Michaud D, Li M, Yamashiro DJ et al. Identification of a patient with Bernard-Soulier syndrome and a deletion in the DiGeorge/velo-cardio-facial chromosomal region in 22q11.2. Hum Mol Genet 1995; 4: 763–766.
Yagi M, Edelhoff S, Disteche CM, Roth GJ . Structural characterization and chromosomal location of the gene encoding human platelet glycoprotein Ibβ. J Biol Chem 1994; 269: 17424–17427.
Kelly MD, Essex DW, Shapiro SS, Meloni FJ, Druck T, Huebner K et al. Complementary DNA cloning of the alternatively expressed endothelial cell glycoprotein Ibβ (GPIbβ) and localization of the GPIbβ gene to chromosome 22. J Clin Invest 1994; 93: 2417–2424.
Lopez JA, Andrews RK, Afshar-Kharghan V, Berndt MC . Bernard-Soulier syndrome. Blood 1998; 91: 4397–4418.
Wenger RH, Wicki AN, Kieffer N, Adolph S, Hameister H, Clemetson KJ . The 5′ flanking region and chromosomal localization of the gene encoding human platelet membrane glycoprotein Ib alpha. Gene 1989; 85: 517–524.
Hickey MJ, Deaven LL, Roth GJ . Human platelet glycoprotein IX: characterization of cDNA and localization of the gene to chromosome 3. FEBS Lett 1990; 274: 189–192.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed. Washington, DC: American Psychiatric Association, 1994.
Kunishima S, Lopez JA, Kobayashi S, Imai N, Kamiya T, Saito H et al. Missense mutations of the glycoprotein (GP) Ibβ gene impairing the GPIb α/β disulfide linkage in a family with giant platelet disorder. Blood 1997; 89: 2404–2412.
Fukushima Y, Ohashi H, Wakui K, Nishida T, Oh-ishi T . A rapid method for starting a culture for the establishment of Epstein-Barr virus–transformed human lymphoblastoid cell lines. Jpn J Hum Genet 1992; 37: 149–150.
Yoshida MC, Sasaki M, Mise K, Semba K, Nishizawa M, Yamamoto T et al. Regional mapping of the human proto-oncogene c-yes-1 to chromosome 18 at band q21.3. Jpn J Cancer Res 1985; 76: 559–562.
Thiagarajan P, Shapiro SS, Levine E, DeMarco L, Yalcin A . A monoclonal antibody to human platelet glycoprotein IIIa detects a related protein in cultured human endothelial cells. J Clin Invest 1985; 75: 896–901.
Iwai K, Matsuki H, Omori M, Tsuchibuchi S, Ishihara S, Tamura A et al. Mental disorders of del22q11.2 syndrome (CATCH22): cross-sectional and longitudinal observation of 8 cases. J Tokyo Wom Med Univ 1998; 68: 338–345.
Van Geet C, Devriendt K, Eyskens B, Vermylen J, Hoylaerts MF . Velocardiofacial syndrome patients with a heterozygous chromosome 22q11 deletion have giant platelets. Pediatr Res 1998; 44: 607–611.
Corrigan JJ . Hematologic aspects of cardiology. In: Emmanouilides GC, Riemenschneidew TA, Allen HD, Gutgesell HP, editors. Heart disease in infants, children, and adolescents. Baltimore: Williams & Wilkins, 1995: 1786–1797.
Ludlow LB, Schick BP, Budarf ML, Driscoll DA, Zackai EH, Cohen A et al. Identification of a mutation in a GATA binding site of the platelet glycoprotein Ibβ promoter resulting in the Bernard-Soulier syndrome. J Biol Chem 1996; 271: 22076–22080.
Kenny D, Morateck PA, Gill JC, Montgomery RR . The critical interaction of glycoprotein (GP) Ibβ with GPIX: a genetic cause of Bernard-Soulier syndrome. Blood 1999; 93: 2968–2975.
Li C, Martin SE, Roth GJ . The genetic defect in two well-studied cases of Bernard-Soulier syndrome: a point mutation in the fifth leucine-rich repeat of platelet glycoprotein Ibα. Blood 1995; 86: 3805–3814.
Shprintzen RJ, Goldberg R, Golding-Kushner KJ, Marion RW . Late-onset psychosis in the velo-cardio-facial syndrome [letter]. Am J Med Genet 1992; 42: 141–142.
Pulver AE, Nestadt G, Goldberg R, Shprintzen RJ, Lamacz M, Wolyniec PS et al. Psychotic illness in patients diagnosed with velo-cardio-facial syndrome and their relatives. J Nerv Ment Dis 1994; 182: 476–478.
Bassett AS, Hodgkinson K, Chow EWC, Correia S, Scutt LE, Weksberg R . 22q11 deletion syndrome in adults with schizophrenia. Am J Med Genet 1998; 81: 328–337.
Loebel AD, Lieberman JA, Alvir JMJ, Mayerhoff DI, Geisler SH, Szymanski SR . Duration of psychosis and outcome in first-episode schizophrenia. Am J Psychiatry 1992; 149: 1183–1188.
Karayiorgou M, Gogos JA, Galke BL, Wolyniec PS, Nestadt G, Antonarakis SE et al. Identification of sequence variants and analysis of the role of the catechol-o-methyl-transferase gene in schizophrenia susceptibility. Biol Psychiatry 1998; 43: 425–431.
Gogos JA, Santha M, Takacs Z, Beck KD, Luine V, Lucas LR et al. The gene encoding proline dehydrogenase modulates sensorimotor gating in mice. Nat Genet 1999; 21: 434–439.
Yagi M, Zieger B, Roth GJ, Ware J . Structure and expression of the human septin gene HCDCREL-1. Gene 1998; 212: 229–236.
Toda S, Kajii Y, Sato M, Nishikawa T . Reciprocal expression of infant- and adult-preferring transcripts of CDCrel-1 septin gene in the rat neocortex. Biochem Biophys Res Commun 2000; 273: 723–728.
Beites CL, Xie H, Bowser R, Trimble WS . The septin CDCrel-1 binds syntaxin and inhibits exocytosis. Nat Neurosci 1999; 2: 434–439.
Acknowledgements
This research was supported by an Open Research Grant (2000, 2001) from the Japan Research Promotion Society for Cardiovascular Diseases and by a grant for the Promotion of the Advancement of Education and Research in Graduate Schools (1999, 2000, 2001). The authors thank S. Shimizu, PhD (Department of Hygiene and Public Health, Tokyo Women’s Medical University, Tokyo, Japan), for statistical analysis and Ms. B. Levene for reading the manuscript.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kato, T., Kosaka, K., Kimura, M. et al. Thrombocytopenia in patients with 22q11.2 deletion syndrome and its association with glycoprotein Ib-β. Genet Med 5, 113–119 (2003). https://doi.org/10.1097/00125817-200303000-00010
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1097/00125817-200303000-00010