Abstract
Purpose
To describe and correlate the morphological and clinical features of focal choroidal excavation (FCE).
Methods
This is a consecutive case series from the review of the 4436 optical coherence tomography scans performed by Kowloon East Cluster Ophthalmic Service from 1 August 2014–31 January 2016. Statistical analysis was performed on SPSS 18.0 (SPSS, Chicago, IL, USA). A significance level of P<0.05 was taken.
Results
All 16 patients with FCE had unilateral involvement. The mean age of diagnosis was 52.56±14.00. The mean greatest linear dimension (GLD) of FCE was 636.25±265.11 μm. The mean choroidal thickness was 183.63±52.39 μm. Fourteen FCEs (87.5%) were conforming and two were non-conforming (12.5%). In the eyes with FCE, concurrent macular pathology was present in four cases (25.0%). Tractional pathologies of macular pucker and macular scar corresponded to the two non-conforming FCEs in the series. Polypoidal choroidal vasculopathy (PCV) and lacquer crack had a close topographic relationship with the FCE. The mean GLD was significantly larger in eyes with concurrent macular pathology than those without (878.00 vs 555.67 μm, P=0.029). In the fellow eyes, concurrent macular pathology was present in 5 cases (31.3%): PCV in 3 cases and chronic central serous chorioretinopathy in 2 cases.
Conclusion
As a significant proportion of FCE is associated with concurrent macular pathology in the involved or fellow eye, angiography for both eyes is recommended even for asymptomatic cases. The GLD of FCE may have clinical value in risk stratification.
Similar content being viewed by others
Introduction
Focal choroidal excavation (FCE)—an excavation of the choroid detected on optical coherence tomography (OCT) in patients with good visual acuity and normal-appearing overlying retina, was first described by Jampol et al1 in 2006. The aetiology of this type of lesion is uncertain. FCE was once postulated to be congenital posterior segment malformation representing ‘microstaphyloma’. It was also proposed that FCEs could be acquired lesions from choroidal atrophy and scarring following choroiditis.2, 3
Morphologically, FCEs could be classified on the basis of the relationship between the outer retina and the choroid. In the conforming type, the outer retina conforms to the choroidal excavation with preservation of the junction between the photoreceptor tips and the retinal pigmented epithelium (RPE). The non-conforming type corresponds to FCEs with a separation between the photoreceptor tips and RPE forming a subretinal space.4
Although many cases of FCE have a relatively stable course and remain asymptomatic, associations with choroidal neovascularisation (CNV),2, 5 polypoidal choroidal vasculopathy (PCV),6, 7 and central serous chorioretinopathy (CSCR) were found.8, 9, 10 There still exist controversies in the clinical significance of FCE. It is also uncertain whether there is any clinical correlation with the morphological features of FCE.
In this study, therefore, we aimed to describe and correlate the morphological and clinical features of FCE in the context of concurrent macular pathologies.
Materials and methods
This is a consecutive case series of all patients with FCE attending the Kowloon East Cluster Ophthalmic Service from 1 August 2014–31 January 2016. The ophthalmic service in this cluster is provided by United Christian Hospital and Tseung Kwan O Hospital, serving a population of close to 1 million. The study was approved by the Research Ethics Committee of the Hospital Authority (ref: KC/KE-16-0119/ER-2) and adherent to the tenets of the Declaration of Helsinki. The cases were retrieved through a review of the 4436 OCT scans of the macula in the specified period.
Spectral-domain OCT (SD-OCT) was performed with the Heidelberg Retinal Angiography and Optical Coherence Tomography System (Spectralis, HRA-OCT; Heidelberg Engineering, Heidelberg, Germany) by a single operator, using the macular line scan and volume map. Measurements of the various dimensions of the FCEs were performed using the caliper feature on the macular line scan of SD-OCT. The greatest linear dimension (GLD) of the FCE was measured as the maximal horizontal distance between the two points where the RPE layer bulges outward, using a cross-sectional line scan directly across the FCE. The choroidal thickness under the excavation was defined as the distance between the outer border of the RPE and the inner scleral border, using the enhanced depth imaging mode of SD-OCT. Fluorescein angiography (FA) and indocyanine green angiography (ICG) were arranged where appropriate.
The diagnosis of concurrent macular pathologies was made on the basis of clinical examination, SD-OCT and angiography. CNV was diagnosed based on SD-OCT, clinical, or angiographic signs of leakage. CSCR was diagnosed in case of serous retinal detachment in the macular region as observed on fundoscopic examination and SD-OCT, together with leakage points observed on angiography. PCV was diagnosed if early hyperfluorescent hot spot(s) was observed with orange subretinal nodule on clinical examination or fundus photos, massive submacular haemorrhage, pulsatile polyp, hypofluorescent halo, or branching vascular network.
All statistical analysis with the OCT measurements (GLD and choroidal thickness) was performed on the SPSS 18.0 software (SPCC Inc., Chicago, IL, USA). For t-test, equal variances between the groups were checked. A significant level of P>0.05 was taken.
Results
The patient demographics, morphological, and clinical features of FCEs in this study are shown in Table 1.
Patient demographics
All 16 patients with FCE had unilateral involvement. The right to left eye ratio was 10 : 6. Nine patients (56.3%) were females and 7 patients (43.6%) were males. The mean age of diagnosis was 52.56±14.00 (range 31–78). The mean spherical equivalent refraction of the involved eyes was −3.28±3.90 D (range −10.00 to +2.38 D), while that of the fellow eyes was −3.13±3.55 D (range −10.50 to +2.00 D). The mean duration of follow-up was 12.8 months, in which 11 patients were followed up for 6 months or more.
OCT findings
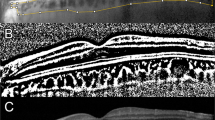
The mean GLD of FCE was 636.25±265.11 μm (range 144–1205 μm). The mean choroidal thickness at the FCE was 183.63±52.39 μm (range 98–268 μm). Out of the 16 cases, 6 FCEs had a subfoveal location (37.5%), while the remaining 10 FCEs were extrafoveal (62.5%). Morphologically, 14 FCEs (87.5%) were conforming type, while the other 2 (12.5%) were non-conforming. The corresponding OCTs of the patients are shown in Figure 1.
Concurrent macular pathology
In the eyes with FCE, concurrent macular pathology was present in four cases (25.0%). One patient had a macular pucker, and another had a parafoveal scar with no clinical or angiographic leakage activity—both corresponding to non-conforming FCEs. The other two patients had conforming FCEs: one associated with PCV and the other with macular haemorrhage from lacquer crack formation due to high myopia. In the latter case, FA did not show any CNV, and the bleeding was self-limiting, leaving no chorioretinal atrophy. There was a close topographic relationship between the FCE and the PCV and lacquer crack, respectively, as shown in Figure 2 and Figure 3.
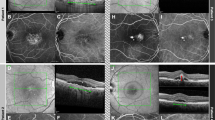
Patient 10: a 70-year-old lady with bilateral polypoidal choroidal vasculopathy. The indocyanine green angiograph (a) shows the polyps on the left eye as indicated by the arrow. The spectral-domain optical coherence tomograph shows that the conforming focal choroidal excavation (b) is in close proximity to the polyps, as indicated by the location of the macular line scan.
Patient 15: a 33-year-old highly myopic man with left eye macular haemorrhage. The fluorescein angiography (a) shows the lacquer crack as indicated by the arrow. The spectral-domain optical coherence tomography shows that the conforming focal choroidal excavation (b) is in close proximity to the lacquer crack, as indicated by the location of the macular line scan.
The remaining 12 cases were not associated with concurrent macular pathology (75.0%). Among these cases, half of them appeared as focal RPE changes on ophthalmoscopic examination; the rest were not discernible clinically.
In fellow eyes of these patients, concurrent macular pathology was present in 5 cases (31.3%): PCV in 3 cases (18.8%) and chronic CSCR in 2 cases.
Visual acuity and clinical course
Out of the 16 eyes with FCE, 11 had a presenting BCVA of 6/12 or above, 9 had a BCVA of 6/9 or above, and only 1 patient had a BCVA below 6/60.
In the presence of concurrent macular pathology, the presenting VA was 6/18 or below. The other 12 cases of FCE without concurrent macular pathology, all conforming in morphologically, were not associated with visual symptoms. They were associated with BCVA of 6/9.5 or above, except in 1 eye where the VA was 6/18 due to a history of retinal detachment.
During the follow-up period, the eyes with FCE followed a stable course of VA that was within one line difference from the presenting VA, except in the highly myopic eye with self-limiting macular haemorrhage whose VA improved from 6/30 to 6/12 without treatment.
Size of FCE and choroidal thickness
The mean GLD of the FCE was significantly larger in eyes with concurrent macular pathology than those without (878.00 vs 555.67 μm, P=0.029). There was no significant difference in the mean choroidal thickness under the excavation in eyes with or without concurrent macular pathology (199.50 vs 178.33 μm, P=0.503). No significant correlation was found between the size of the FCE, as measured by GLD, and the choroidal thickness under the excavation (Pearson correlation 0.108, P=0.69).
Discussion
In this study, FCE was diagnosed in 16 Chinese patients among the 4436 SD-OCT scans performed in the 18 months of study period. The mean age of diagnosis was 52.56, ranging from 31 to 78 years. There was no gender predilection. The patient demographics were similar to previous reports, which were mainly done in Asian populations.4, 6, 11, 12, 13 The mean refraction of both the involved eyes and the fellow eyes was in the −3D myopic range, consistent with the FCE case series by Margolis et al.2
Factors contributing to the formation of FCEs into conforming or non-conforming morphologies are uncertain. However, the two cases of non-conforming FCE in this study were associated with a tractional macular pathology: macular pucker and scar, respectively. We speculate that traction may be a potential factor in the formation of different morphologies of FCE lesions. The non-conforming morphology of FCE may have formed when the photoreceptor tips under traction detach from the underlying RPE.
Consistent with the study by Obata et al,6 the clinical course of FCE in this study was generally stable. No involved eye had a change VA more than one line from baseline during the follow-up period. However, 25.0% of the eyes with FCE were associated with concurrent macular pathology. It remains uncertain whether FCE has an aetiological role, but the observation of choroidal hyperpermeability and focal loss of large choroidal vessels associated with FCE has suggested that mechanical changes in the RPE/Bruch membrane in the FCE resulting from inflammation or ischaemia may predispose to choroidal vessel outgrowth.8, 13, 14, 15, 16, 17 This is supported by the close topographic relationship between FCE and CNV demonstrated in previous studies.5, 14 Consistent with this clinical observation, in this study the FCE and polyp/lacquer crack were also found in close proximity. It is possible that the focal weak point in the RPE/Bruch membrane complex in FCE may predispose in polyp outgrowth in PCV, and lacquer crack formation in a highly myopic patient.
With a mean GLD of 636.25 μm and mean choroidal thickness of 183.63 μm, the FCEs of this case series have physical parameters that are similar to those reported in other series, as shown in Table 2.11, 12, 13 To our knowledge, there has not been any study to date attempting to establish the clinical correlation of different physical parameters of FCE. Our results showed that the mean size of the FCE is significantly larger in eyes with concurrent macular pathology (mean GLD 878.00 vs 555.67 μm, P=0.029). Therefore, the size of the FCE, exemplified by the GLD, may have potential clinical value in risk stratification regarding the occurrence of concurrent macular pathology.
While the current literature mainly focuses on the association between FCE and concurrent macular pathologies in the involved eye, our case series has shown the presence of concurrent macular pathologies in even a higher proportion of the fellow eyes (31.3%) than the FCE-involved eyes (25.0%). In the fellow eyes of patients with FCE, PCV was found in three cases and CSCR in two cases. The association of FCE with macular pathology in the fellow eye, however, is not well reported in the literature. The only relevant study to date was the cohort by Lim et al,15 in which FCE was found in 2.9% of the fellow eyes of patients with PCV and 1.2% of the fellow eyes of patients with CSCR.
On the basis of this finding, we recommend that FA and ICG be arranged for both eyes in any patient with FCE, even for with good VA and no visual symptoms. The finding of abnormal choroidal hyperpermeability or asymptomatic vascular lesions such as PCV or CNV should warrant close observation and early treatment if necessary.
This study is limited by its retrospective nature and moderate case number. The follow-up duration was limited to demonstrate the clinical course of the FCE in some patients, especially among those who were asymptomatic. The occurrence of asymptomatic FCE is probably higher in the general population. Those with other macular pathologies are subject to more OCT examinations and they may be over-represented in this case series.

References
Jampol LM, Shankle J, Schoroeder R, Tornambe P, Spaide RF, Hee MR et al. Diagnostic and therapeutic challenges. Retina 2006; 26 (9): 1072–1076.
Margolis R, Mukkamala SK, Jampol LM, Spaide RF, Ober MD, Sorenson JA et al. The expanded spectrum of focal choroidal excavation. Arch Ophthalmol 2011; 129 (10): 1320–1325.
Hashimoto Y, Saito W, Noda K, Ishida S . Acquired focal choroidal excavation associated with multiple evanescent white dot syndrome: observations at onset and a pathogenic hypothesis. BMC Ophthalmol 2014; 14: 135.
Wakabayashi Y, Nishimura A, Higashide T, Ijiri S, Sugiyama K . Unilateral choroidal excavation in the macula detected by spectral-domain optical coherence tomography. Acta Ophthalmol 2010; 88 (3): e87–e91.
Lee J, Lee W . Choroidal neovascularization associated with focal choroidal neovascularization. Am J Ophthalmol 2014; 157: 710–718.e1.
Obata R, Takahashi H, Ueta T, Yuda K, Kure K, Yanagi Y . Tomographic and angiographic characteristics of eyes with macular focal choroidal excavation. Retina 2013; 33: 1201–1210.
Kobayashi W, Abe T, Tamai H, Nakazawa T . Choroidal excavation with polypoidal choroidal vasculopathy: a case report. Clin Ophthalmol 2012; 6: 1373–1376.
Ellabban AA, Tsujikawa A, Ooto S, Yamashiro K, Oishi A, Nakata I et al. Focal choroidal excavation in eyes with central serous chorioretinopathy. Am J Ophthalmol 2013; 156: 673–683.
Suzuki M, Gomi F, Hara C, Sawa M, Nishida K . Characteristics of central serous chorioretinopathy complicated by focal choroidal excavation. Retina 2014; 34: 1216–1222.
Luk FO, Fok AC, Lee A, Liu AT, Lai TY . Focal choroidal excavation in patients with central serous chorioretinopathy. Eye 2015; 29 (4): 453–459.
Lee CS, Woo SJ, Kim YK, Hwang DJ, Kang HM, Kim H et al. Clinical and spectral-domain optical coherence tomography findings in patients with focal choroidal excavation. Ophthalmology 2014; 121: 1029–1035.
Guo J, Zhong L, Jiang C, Zhou X, Xu G, Wang W et al. Clinical and optic coherence tomography findings of focal choroidal excavation in Chinese patients. BMC Ophthalmol 2014; 14: 63.
Shinojima A, Kawamura A, Mori R, Yuzawa M . Morphologic features of focal choroidal excavation on spectral domain optical coherence tomography with simultaneous angiography. Retina 2014; 34: 1407–1414.
Xu H, Zeng F, Shi D, Sun X, Chen X, Bai Y . Focal choroidal excavation complicated by choroidal neovascularization. Ophthalmology 2014; 121: 246–50.
Lim FP, Loh BK, Cheung CM, Lim LS, Chan CM, Wong DW . Evaluation of focal choroidal excavation in the macula using swept source optical coherence tomography. Eye 2014; 28: 1088–1094.
Kuroda Y, Tsujikawa A, Ooto S, Yamashiro K, Oishi A, Nakanishi H et al. Association of focal choroidal excavation with age related macular degeneration. Invest Ophthalmol Vis Sci 2014; 55: 6046–6054.
Cheung CM, Bhargava M, Laude A, Koh ACh, Xiang L, Wong D et al. Asian age-related macular degeneration phenotyping study: rationale, design and protocol of a prospective cohort study. Clin Exp Ophthalmol 2012; 40: 727–735.
Acknowledgements
Special thanks to Mr Raymond M Wu for all the OCT images of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Part of the contents of the manuscript was presented in the Hong Kong Ophthalmological Symposium in November, 2016.
Rights and permissions
About this article
Cite this article
Chung, C., Li, S. & Li, K. Focal choroidal excavation—morphological features and clinical correlation. Eye 31, 1373–1379 (2017). https://doi.org/10.1038/eye.2017.71
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2017.71
This article is cited by
-
Long-term follow-up demonstrates change in conformation shape of the focal choroidal excavation lesions
BMC Ophthalmology (2024)
-
Pathophysiology of central serous chorioretinopathy: a literature review with quality assessment
Eye (2022)
-
Intercalary membrane break and detachment causes intrachoroidal cavitation in macular coloboma
International Ophthalmology (2022)