Abstract
Purpose
To evaluate the visual outcomes of choroidal neovascularization (CNV) secondary to pathological myopia in eyes treated with photodynamic therapy (PDT), and to determine the effect of lesion location and foveal involvement on visual prognosis.
Methods
Interventional case series of 24 consecutive patients with myopic CNV treated with PDT. The main outcome measure was final LogMAR visual acuity (VA).
Results
Of 24 eyes, the CNV lesion was subfoveal in 11 and extrafoveal in 13. Overall, the mean LogMAR VA at 24 months was 0.72. Extrafoveal CNV lesions achieved significantly better final VA compared with subfoveal CNV (LogMAR 0.45 vs 1.05, P=0.012). Eyes with extrafoveal CNV lesions were subdivided into foveal-sparing PDT (where the PDT laser spot did not involve the foveal center) and foveal-involved PDT (where the PDT laser covered the fovea). At all time points, the group with foveal-sparing PDT had significantly better VA compared with the foveal-involved group. The final LogMAR VA for the foveal-sparing PDT group was 0.26 compared with 1.00 for the foveal-involved PDT group (P=0.003). At 24 months, 77.8% of foveal-sparing PDT cases achieved VA of ≥20/40, compared with 25% of foveal-involved PDT cases and 9.1% of subfoveal CNV lesions (P=0.006).
Conclusion
For patients with myopic CNV, foveal-sparing PDT results in significantly better long-term visual outcomes compared with those with foveal-involved PDT. Foveal-sparing PDT may be of value for treatment of myopic CNV patients who are not suitable for treatment with anti-vascular endothelial growth factor injections.
Similar content being viewed by others
Introduction
The prevalence of myopia is increasing among the younger age groups and also varies by population, with higher rates of myopia reported among populations of Asian descent. Pathologic myopia is a major cause of irreversible vision loss1, 2 and choroidal neovascularization (CNV) secondary to pathological myopia is the leading cause of CNV in people aged younger than 50.3
Among patients with myopic CNV, between 50 and 60% of eyes experience deterioration of vision within 2 years.4 The long-term visual outcome for myopic CNV is poor, and many patients have visual acuity (VA) of 20/200 or worse within 5−10 years.3
The Verteporfin in Photodynamic Therapy (VIP) study5 evaluated the efficacy of photodynamic therapy (PDT) in patients with subfoveal CNV secondary to pathologic myopia, and reported that VA stabilized in 72% of patients at 1 year. More recently, studies on treatment of myopic CNV with intravitreal injections of anti-vascular endothelial growth factor (anti-VEGF) agents have reported good clinical outcomes.6, 7, 8, 9, 10, 11, 12 However, anti-VEGF agents are associated with systemic risks such as cerebrovascular accidents and other arterial thromboembolic events, especially for patients with pre-existing disease.13, 14, 15, 16 In addition, intravitreal injections carry the risk of infectious endophthalmitis.17 Therefore, PDT may still have a role in the management of patients with myopic CNV where anti-VEGF agents are contraindicated due to the systemic risks or where patients are unwilling to accept the systemic or ocular risks associated with this treatment.
We aimed to evaluate the visual outcome of patients with myopic CNV and to determine the role of lesion location and foveal involvement by the PDT laser spot in determining the visual prognosis.
Materials and methods
This was an interventional case series of 24 consecutive patients with pathologic myopia treated for myopic CNV at the Ophthalmology Clinic at Tan Tock Seng Hospital, National Healthcare Group Eye Institute, Singapore. This study was approved by the Institutional Review Board of the National Healthcare Group, Singapore, and complied with the Tenets set forth in the Declaration of Helsinki.
The inclusion criteria were spherical equivalent of −6 D or worse or features of pathological myopia on retinal examination. Patients with co-existing ocular pathology, especially CNV secondary to age-related macular degeneration, were excluded.
Confocal scanning laser ophthalmoscopy flurorescein angiography (FA) and indocyanine green angiography (ICGA) were performed for all patients using the Heidelberg HRA2 (Heidelberg Engineering, Heidelberg, Germany) using a standardized imaging protocol.
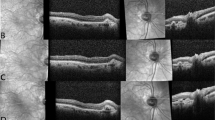
The angiograms were independently reviewed by two ophthalmologists to confirm the diagnosis of myopic CNV and its location. ICGA was used to exclude polypoidal choroidal vasculopathy, which is more common among Asian populations, and retinal angiomatous proliferation. The CNV lesions were classified according to their location on FA (subfoveal, juxta-foveal, or extra foveal).5 The non-subfoveal CNV lesions were further analyzed according to whether or not the laser spot used during PDT treatment covered the foveal center. If the PDT laser spot did not cover the foveal center, this was termed foveal-sparing PDT (Group 1) (Figures 1a and b), whereas if the laser spot covered the foveal center, this was classified as foveal-involved PDT (Group 2). Patients with subfoveal CNV lesions (Figures 1c and d), where the PDT laser spot would definitely cover the foveal center, were classified as Group 3.
During the period these patients initially presented, PDT was the standard of care for patients with myopic CNV and anti-VEGF injections were not commonly in use in our institution. Patients were treated by a single ophthalmologist (THL) and received full-fluence PDT with Verteprofin following the guidelines of the VIP study and Treatment of Age-Related Macular Degeneration with Photodynamic Therapy study.5, 18 All patients were followed up for 2 years and the main outcome measure was LogMAR VA.
Statistical analysis was performed using SPSS for Windows version 16 (SPSS Inc., Chicago, IL, USA), with P values <0.05 taken as significant.
Results
There were 11 males (45.8%) and 13 females (54.2%), with a mean age of 58.7 years (SD±13.7) and mean spherical equivalent of −11.3 D (SD±4.8 D). In 11 patients (45.8%), the right eye was affected.
In 11 patients (45.8%), the CNV lesion was subfoveal. Of the remaining 13 (54.2%) with extrafoveal CNV lesions, 4 had foveal involvement by PDT laser whereas the remaining 9 were foveal-sparing cases. The mean number of PDT treatments for all patients was 1.7 (range, 1−4 treatments) and there was no significant difference in mean number of PDT among the three groups of patients.
The mean best-corrected logMAR VA (BCVA) was 0.67 (±0.51) on presentation and 0.72 (±0.62) at the final review. At all follow-up visits up to 24 months, mean VA was better for extrafoveal lesions compared with subfoveal CNV lesions (all P<0.05 except for 3 and 18 months) (Table 1). At the final visit, the mean LogMAR VA was 0.45 for extrafoveal lesions compared with 1.05 for subfoveal CNV lesions (P=0.012).
Visual outcomes were not associated with gender, initial BCVA, number of PDT treatments, or severity of myopia.
Of the 24 patients, 20 had subsequent FA performed at a mean of 12.9 months (range, 4–24 months). Among eyes with extrafoveal CNV lesions on presentation, 4 of 13 (30.8%) developed expansion of the CNV lesion to involve the fovea, whereas the remainder were either quiescent or had lesions that remained non-subfoveal. Of the four eyes with subsequent subfoveal extension, all experienced worsening of VA after involvement of the fovea by the CNV lesion.
Analysis of visual outcomes based on foveal involvement by PDT laser spot
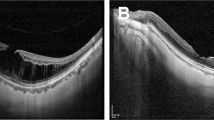
Upon further analysis of patients according to foveal involvement by the PDT laser spot (Figure 2), we found that among patients with foveal-sparing PDT (Group 1), the mean LogMAR VA varied from 0.22 to 0.30 at the various follow-up time points and did not worsen over time. In contrast, the mean LogMAR VA of patients with foveal-involved PDT (Groups 2 and 3 combined) ranged from 0.73 to 1.05, and gradually worsened over the follow-up period. The differences between the two groups was statistically significant at all time points (all P<0.05).
At the final visit (24 months), mean LogMAR VA was 0.26 for the foveal-sparing PDT group compared with 1.00 for the foveal-involved group (P=0.003), and seven of nine patients in Group 1 had VA 20/40 or better, compared with one patient each in Groups 2 and 3 (77.8% vs 25.0% vs 9.1% for groups 1, 2, and 3, respectively, P=0.006).
Discussion
In this study, we have demonstrated that among patients with myopic CNV, those treated with foveal-sparing PDT had significantly better VA compared with those treated with foveal-involving PDT.
The mean LogMAR VA for myopic CNV lesions treated with PDT ranges from 0.25 to 0.72 in the literature (Table 2),19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30 which is comparable to the overall results in this series (mean LogMAR VA 0.72 at 2 years). In studies that have analyzed visual outcomes based on the location of the CNV lesion, final VA for subfoveal myopic CNV ranged from 0.43 to 0.92,20, 22, 24, 29 whereas the final VA for extrafoveal myopic CNV was slightly better, ranging from 0.25 to 0.72.19, 20, 23 Similar to these earlier studies, our results showed a significant difference in visual outcomes based on the location of the CNV lesion, with extrafoveal CNV having better outcomes compared with subfoveal CNV (LogMAR VA 0.45 vs 1.05 at 24 months).
With the advent of anti-VEGF drugs, ophthalmologists now have a newer modality to treat patients with myopic CNV.6, 8, 9, 10, 11, 12 The mean final VA in patients treated with anti-VEGF drugs reported in the literature ranges from 0.23 to 0.55 (Table 2).6, 7, 8, 9, 10, 11, 12,31, 32,33,34,35,36 More importantly, in studies which have compared anti-VEGF with PDT treatment for myopic CNV, patients treated with anti-VEGF generally achieved better visual outcomes compared with the group treated with PDT.7, 24, 25 It is important to note, however, that in these studies, patients were not randomized to the two treatment groups. To the best of our knowledge, there are currently no results from randomized controlled trials comparing the two treatment modalities.
It has been suggested that PDT may damage the underlying choroid and photoreceptors, causing chorioretinal atrophy,19, 20 which accounts for the long-term visual loss. Hence some authors have suggested that anti-VEGF therapy is advantageous compared with PDT. However, anti-VEGF injections are associated with both ocular and systemic risks to patients.13, 14, 15, 16, 17 Some patients with previous history of thromboembolic events may not be willing to accept the higher risk of cerebrovascular accidents or other arterial thromboembolic events. In addition, the risk of endophthalmitis associated with intravitreal injections,17 although rare, is an ever-present consideration each time the injections are performed. Some patients are not keen to take this risk, especially if the fellow eye has already lost functional vision. For this group of patients who are unwilling to accept the risks of anti-VEGF therapy, PDT may offer an alternative treatment modality. We believe that PDT may still have a useful role in the management of myopic CNV, if patients are carefully selected.
Our results have shown that among patients with extrafoveal CNV in which the PDT laser spot does not involve the center of the fovea, the visual outcomes are good, with a mean LogMAR VA better than 0.26. Also, 78% of patients with foveal-sparing PDT had VA 20/40 or better at 2 years. These visual outcomes are comparable to, or in some cases better than, those reported from studies using anti-VEGF drugs. We believe that PDT may be useful in treating extrafoveal CNV lesions, especially if the laser spot can be adjusted to avoid the foveal center.
Patients with foveal-involving PDT included those with subfoveal and extrafoveal CNV lesions. As this is a heterogeneous group, it remains unknown whether the poorer visual outcome in this group is due to the CNV location or involvement of the foveal center by the PDT laser spot. It is likely that both factors contributed to the final VA, and further studies are required to evaluate this in greater detail.
The strengths of this study include a long duration of follow up and treatment of all patients by a single experienced ophthalmologist, thus ensuring that PDT technique does not confound the results. The limitations of this study include the lack of a control group for comparison of visual outcomes. Currently, we are not aware of any published results of randomized controlled trials comparing PDT with anti-VEGF drugs.
The results of earlier randomized studies on myopic CNV have focused on subfoveal lesions, whereas it has been shown that extrafoveal CNV lesions occur in between 18.5 and 32% of myopic CNV patients.3, 4 We believe that our results may provide the impetus to perform randomized controlled trials not only for subfoveal CNV lesions, but also to examine the role of foveal-sparing PDT in the management of extrafoveal myopic CNV.
In summary, this study demonstrates that in patients where foveal-sparing PDT can be performed, patients can have good visual outcomes and PDT may be an alternative treatment for patients who are unwilling to accept the risk of anti-VEGF therapy.
Contributorship statement
Each author (CS Tan, MC Chew, and TH Lim) certifies that they have met all of the following criteria: (1) substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; (2) drafting the article or revising it critically for important intellectual content; and (3) final approval of the version to be published.

References
Xu L, Wang Y, Li Y, Wang Y, Cui T, Li J et al. Causes of blindness and visual impairment in urban and rural areas in Beijing: the Beijing eye study. Ophthalmology 2006; 113 (7): 1134.e1–1134.e11.
Iwase A, Araie M, Tomidokoro A, Yamamoto T, Shimizu H, Kitazawa Y et al. Prevalence and causes of low vision and blindness in a Japanese adult population: the Tajimi study. Ophthalmology 2006; 113 (8): 1354–1362.
Yoshida T, Ohno-Matsui K, Yasuzumi K, Kojima A, Shimada N, Futagami S et al. Myopic choroidal neovascularization: a 10-year follow-up. Ophthalmology 2003; 110 (7): 1297–1305.
Hampton GR, Kohen D, Bird AC . Visual prognosis of disciform degeneration in myopia. Ophthalmology 1983; 90 (8): 923–926.
Verteporfin in Photodynamic Therapy Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in pathologic myopia with verteporfin. 1-year results of a randomized clinical trial--VIP report no. 1. Ophthalmology 2001; 108 (5): 841–852.
Hayashi K, Shimada N, Moriyama M, Hayashi W, Tokoro T, Ohno-Matsui K et al. Two-year outcomes of intravitreal bevacizumab for choroidal neovascularization in Japanese patients with pathologic myopia. Retina 2012; 32 (4): 687–695.
Hayashi K, Ohno-Matsui K, Teramukai S, Shimada N, Moriyama M, Hayashi W et al. Comparison of visual outcome and regression pattern of myopic choroidal neovascularization after intravitreal bevacizumab or after photodynamic therapy. Am J Ophthalmol 2009; 148 (3): 396–408.
Yoon J, Byun Y, Koh H . Intravitreal anti-VEGF versus photodynamic therapy with verteporfin for treatment of myopic choroidal neovascularization. Retina 2010; 30 (3): 418–424.
Ruiz-Moreno J, Montero J, Amat-Peral P . Myopic choroidal neovascularization treated by intravitreal bevacizumab: comparison of two different initial doses. Graefes Arch Clin Exp Ophthalmol 2011; 249 (4): 595–599.
Nakanishi H, Tsujikawa A, Yodoi Y, Ojima Y, Otani A, Tamura H et al. Prognostic factors for visual outcomes 2-years after intravitreal bevacizumab for myopic choroidal neovascularization. Eye (Lond) 2011; 25 (3): 375–381.
Ikuno Y, Sayanagi K, Soga K, Sawa M, Tsujikawa M, Gomi F et al. Intravitreal bevacizumab for choroidal neovascularization attributable to pathological myopia: one-year results. Am J Ophthalmol 2009; 147 (1): 94–100.
Lai T, Chan W, Liu D, Lam D . Intravitreal ranibizumab for the primary treatment of choroidal neovascularization secondary to pathologic myopia. Retina 2009; 29 (6): 750–756.
Day S, Acquah K, Mruthyunjaya P, Grossman DS, Lee PP, Sloan FA et al. Ocular complications after anti-vascular endothelial growth factor therapy in Medicare patients with age-related macular degeneration. Am J Ophthalmol 2011; 152 (2): 266–272.
Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group, Martin DF, Maguire MG, Fine SL, Ying GS, Jaffe GJ, Grunwald JE et al. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology 2012; 119 (7): 1388–1398.
IVAN Study Investigators, Chakravarthy U, Harding SP, Rogers CA, Downes SM, Lotery AJ, Wordsworth S et al. Ranibizumab versus bevacizumab to treat neovascular age-related macular degeneration: one-year findings from the IVAN randomized trial. Ophthalmology 2012; 119 (7): 1399–1411.
Wu L, Martínez-Castellanos MA, Quiroz-Mercado H, Arevalo JF, Berrocal MH, Farah ME et al. Twelve-month safety of intravitreal injections of bevacizumab (Avastin): results of the Pan-American Collaborative Retina Study Group (PACORES). Graefes Arch Clin Exp Ophthalmol 2008; 246 (1): 81–87.
Pilli S, Kotsolis A, Spaide RF, Slakter J, Freund KB, Sorenson J et al. Endophthalmitis associated with intravitreal anti-vascular endothelial growth factor therapy injections in an office setting. Am J Ophthalmol 2008; 145 (5): 879–882.
TAP Study Group. Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin: one-year results of 2 randomized clinical trials—TAP report. Treatment of age-related macular degeneration with photodynamic therapy (TAP) Study Group. Arch Ophthalmol 1999; 117 (10): 1329–1345.
Hayashi K, Ohno-Matsui K, Teramukai S, Shimada N, Moriyama M, Hara W et al. Photodynamic therapy with verteporfin for choroidal neovascularization of pathologic myopia in Japanese patients: comparison with nontreated controls. Am J Ophthalmol 2008; 145 (3): 518–526.
Hayashi K, Ohno-Matsui K, Shimada N, Moriyama M, Hayashi W, Wang J et al. Long-term results of photodynamic therapy for choroidal neovascularization in Japanese patients with pathologic myopia. Am J Ophthalmol 2010; 151 (1): 137–147.
Coutinho AM, Silva RM, Nunes SG, Cachulo ML, Figueira JP, Murta JN et al. Photodynamic therapy in highly myopic eyes with choroidal neovascularization: 5 years of follow-up. Retina 2011; 31 (6): 1089–1094.
Lam D, Chan W, Liu D, Fan D, Lai W, Chong K et al. Photodynamic therapy with verteporfin for subfoveal choroidal neovascularisation of pathologic myopia in Chinese eyes: a prospective series of 1 and 2 year follow up. Br J Ophthalmol 2004; 88 (10): 1315–1319.
Lam D, Liu D, Fan D, Lai W, So S, Chan W et al. Photodynamic therapy with verteporfin for juxtafoveal choroidal neovascularization secondary to pathologic myopia-1-year results of a prospective series. Eye (Lond) 2005; 19 (8): 834–840.
Ikuno Y, Nagai Y, Matsuda S, Arisawa A, Sho K, Oshita T et al. Two-year visual results for older Asian women treated with photodynamic therapy or bevacizumab for myopic choroidal neovascularization. Am J Ophthalmol 2010; 149 (1): 140–146.
Baba T, Kubota-Taniai M, Kitahashi M, Okada K, Mitamura Y, Yamamoto S et al. Two-year comparison of photodynamic therapy and intravitreal bevacizumab for treatment of myopic choroidal neovascularisation. Br J Ophthalmol 2010; 94 (7): 864–870.
Pece A, Vadalà M, Isola V, Matranga D . Photodynamic therapy with verteporfin for juxtafoveal choroidal neovascularization in pathologic myopia: a long-term follow-up study. Am J Ophthalmol 2007; 143 (3): 449–454.
Parodi MB, Iacono P, Papayannis A, Sheth S, Bandello F . Laser photocoagulation, photodynamic therapy, and intravitreal bevacizumab for the treatment of juxtafoveal choroidal neovascularization secondary to pathologic myopia. Arch Ophthalmol 2010; 128 (4): 437–442.
Ruiz-Moreno J, Amat P, Montero J, Lugo F . Photodynamic therapy to treat choroidal neovascularisation in highly myopic patients: 4 years' outcome. Br J Ophthalmol 2008; 92 (6): 792–794.
Giansanti F, Virgili G, Donati MC, Giuntoli M, Pieretti G, Abbruzzese G et al. Long-term results of photodynamic therapy for subfoveal choroidal neovascularization with pathologic myopia. Retina 2012; 32 (8): 1547–1552.
Axer-Siegel R, Ehrlich R, Weinberger D, Rosenblatt I, Shani L, Yassur Y et al. Photodynamic therapy of subfoveal choroidal neovascularization in high myopia in a clinical setting: visual outcome in relation to age at treatment. Am J Ophthalmol 2004; 138 (4): 602–607.
Gharbiya M, Cruciani F, Parisi F, Cuozzo G, Altimari S, Abdolrahimzadeh S et al. Long-term results of intravitreal bevacizumab for choroidal neovascularisation in pathological myopia. Br J Ophthalmol 2012; 96 (8): 1068–1072.
Oishi A, Yamashiro K, Tsujikawa A, Ooto S, Tamura H, Nakata I et al. Long-term effect of intravitreal injection of anti-VEGF agent for visual acuity and chorioretinal atrophy progression in myopic choroidal neovascularization. Graefes Arch Clin Exp Ophthalmol 2013; 251 (1): 1–7.
Voykov B, Gelisken F, Inhoffen W, Voelker M, Bartz-Schmidt K, Ziemssen F et al. Bevacizumab for choroidal neovascularization secondary to pathologic myopia: is there a decline of the treatment efficacy after 2 years? Graefes Arch Clin Exp Ophthalmol 2010; 248 (4): 543–550..
Ruiz-Moreno JM, Montero JA, Arias L, Araiz J, Gomez-Ulla F, Silva R et al. Twelve-month outcome after one intravitreal injection of bevacizumab to treat myopic choroidal neovascularization. Retina 2010; 30 (10): 1609–1615.
Vadalà M, Pece A, Cipolla S, Monteleone C, Fasolino G, Casuccio A et al. Is ranibizumab effective in stopping the loss of vision for choroidal neovascularisation in pathologic myopia? A long-term follow-up study. Br J Ophthalmol 2011; 95 (5): 657–661.
Yoon JU, Kim YM, Lee SJ, Byun YJ, Koh HJ . Prognostic factors for visual outcome after intravitreal anti-VEGF injection for naive myopic choroidal neovascularization. Retina 2012; 32 (5): 949–955.
Acknowledgements
Dr Tan receives research support from the National Healthcare Group Clinician Scientist Career Scheme Grant CSCS/12005 and Clinician Leadership in Research Grant CLR-09006.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Tan receives travel support from Novartis and Bayer. Professor Lim receives travel support from Novartis, Bayer, and Heidelberg Engineering.
Rights and permissions
About this article
Cite this article
Tan, C., Chew, M. & Lim, T. Comparison of foveal-sparing with foveal-involving photodynamic therapy for myopic choroidal neovascularization. Eye 28, 17–22 (2014). https://doi.org/10.1038/eye.2013.204
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2013.204