Abstract
Purpose
To study the relationship between macular ischaemia on fluorescein angiography (FA) and pathomorphology at the foveal centre delineated by spectral-domain optical coherence tomography (OCT) in macular oedema (MO) associated with branch retinal vein occlusion (BRVO).
Methods
One hundred and five consecutive eyes of 105 patients with MO (centre point thickness (CPT) ≥300 μm) associated with BRVO in which FA using Heidelberg Retinal Angiography 2 and Spectralis OCT were performed on the same day were retrospectively reviewed. We evaluated the foveal pathomorphology using OCT images and the association with macular ischaemia.
Results
Within 1 year from symptom onset, 94 eyes were classified with perfused macula (34 eyes) or non-perfused macula (60 eyes). Eyes with perfused macula had better visual acuity and less CPT than those with non-perfused macula (P=0.024 and P<0.001, respectively). Fourteen eyes with perfused macula had serous retinal detachment (SRD) alone at the presumed foveal centre (SRD type); seven, a sponge-like swelling at that area (retinal swelling type); 11, foveal cystoid spaces alone (cystoid MO (CMO) type), and 2, with both SRD and foveal cystoid spaces (SRD+CMO type). However, 58 eyes with non-perfused macula had foveal cystoid spaces (42 of CMO type and 16 of SRD+CMO type), with a significant association between them (P<0.001). Among 11 eyes with symptoms exceeding 1 year, 6 eyes had perfused macula, and none had the SRD type.
Conclusion
Most eyes without foveal cystoid spaces have perfused macula in MO associated with BRVO.
Similar content being viewed by others
Introduction
Branch retinal vein occlusion (BRVO) is a common retinal vascular disease that leads to vision-threatening pathogenesis, vascular hyperpermeability, and neovascularization.1, 2 BRVO involving the macula often is associated with macular oedema (MO), which affects the central vision. Although several treatment modalities have been used,1, 3, 4, 5, 6 many patients still have poor visual prognosis. Further investigations into the pathogenesis and interventions remain to be performed.
BRVO often develops in transient or permanent non-perfused areas (NPA)2, 7, 8 in the affected area after the obstructed retinal vein increases the hydrostatic pressure and disrupts the blood flow. Several studies have reported that an enlarged foveal avascular zone (FAZ) is correlated with the visual acuity (VA), and the disrupted macular microcirculation is involved in MO associated with BRVO.9, 10 In contrast, Finkelstein8 reported a better prognosis in eyes with ischaemic macula associated with BRVO. Although it is evident that retinal ischaemia exacerbates dysfunction in the affected retina, the controversy suggests that pathology other than macular ischaemia, such as MO, causes visual dysfunction.11, 12
Recent advances in optical coherence tomography (OCT) imaging have enabled clinicians to obtain useful and detailed information about the various retinal layers.13, 14 Compared with the classic examinations including biomicroscopy, OCT images have shown the detailed pathomorphology in MO associated with retinal vascular diseases including diabetic retinopathy (DR) and retinal vein occlusion (RVO), cystoid spaces, sponge-like swelling, and serous retinal detachments (SRDs).15, 16 The integrity of outer layers of the fovea, the junction of the inner and outer segments, and the external limiting membrane also have been reported to be related to visual function in resolved MO associated with BRVO and DR.17, 18, 19 However, it is unclear how an obstruction of a branch retinal vein results in a great diversity of morphology in the retinal vasculature and parenchyma. It also is not well understood how macular ischaemia affects the pathomorphology in the retinal parenchyma and vice versa.
The current study investigated the association between changes in the perifoveal capillaries and the foveal pathomorphology in MO associated with BRVO, and showed that macular ischaemia is significantly associated with the foveal cystoid spaces.
Materials and methods
Patients
We retrospectively evaluated 105 eyes of 105 patients (mean, 68.7±11.5 years; range, 43–89 years) with MO (centre point thickness (CPT)20 ≥300 μm) associated with BRVO for which fluorescein angiography (FA) and OCT images of sufficient quality were obtained. Twenty-five eyes were omitted because of the poor quality of images due to retinal haemorrhage, media opacities, and so on. The patients visited the Department of Ophthalmology of Kyoto University Hospital from December 2007 to November 2009. All research and measurements adhered to the tenets of the Declaration of Helsinki and were approved by the ethics committee in our institution. Each patient provided informed consent after a detailed explanation of the nature and possible consequences of the study procedures was provided.
Fluorescein angiography
After the best-corrected VA was measured and fundus biomicroscopy was performed, FA images of the macular area were obtained using Heidelberg Retinal Angiography 2 (Heidelberg Engineering, Heidelberg, Germany) and the macular ischaemia was evaluated. Considering previous publications that documented it,8, 10, 21 we classified the macula as either perfused with a capillary plexus around the entire circumference of the FAZ or non-perfused, in which the FAZ was in contact with the NPA.
Optical coherence tomography
The retinal sectional images in the macula were acquired using spectral-domain OCT (Spectralis OCT, Heidelberg Engineering) with infrared fundus imaging.14 Reduced speckle noise enabled us to divide each retinal layer on the Spectralis OCT images. In the vertical and horizontal sections centred on the fovea, the presumed fovea was defined as the central area in the absence of the inner retinal layers, the nerve fibre layer, the ganglion cell layer, the inner plexiform layer, and the inner nuclear layer (INL), whether any foveal pathomorphology was present or absent.22 Briefly, all or any of these layers, which were obvious in the peripheral areas, were traced to the centre until the area where these layers disappeared was clearly determined. We then classified them qualitatively into four types: the SRD type, which had SRD but no cystoid spaces at the presumed foveal centre; the cystoid MO (CMO) type, which had predominantly cystoid spaces at that area and sometimes minimal triangularly shaped SRD; the CMO+SRD type, which had both SRD and cystoid spaces; and the retinal swelling type, which had no SRD or cystoid spaces. We then measured the thickness at the presumed foveal centre, from the innermost of ILM to the retinal pigment epithelium.
Statistical analysis
The results are expressed as the mean±SD. Statistical analysis was performed using the Student’s t-test to compare quantitative data populations with normal distributions and equal variance. Data were analysed using the Mann–Whitney U test for populations with non-normal distributions or unequal variance. Significant differences in the sampling distributions were determined using the χ2 square test or Fisher’s exact test. A P-value <0.05 was considered statistically significant.
Results
Patterns of the perifoveal capillary network (PCN) in MO associated with BRVO
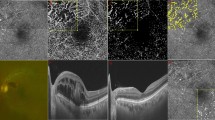
Although the NPA has been reported to be associated with neovascular complications,2, 23 it remains to be evaluated how ischaemic changes affect MO associated with BRVO. We investigated the status of the PCN in 105 consecutive eyes with MO associated with BRVO. We focused on 94 eyes with fresh BRVO (<1 year after the onset of symptoms) and divided them based on their perfusion status; that is, those with a perfused macula (34 eyes; Figure 1a) and those with non-perfused macula (60 eyes; Figures 1b and c). A total of 33 eyes with non-perfused macula had a continuous PCN (Figure 1c), and 27 eyes had disrupted PCN (Figure 1b). When we compared each parameter, we found that the logarithm of the minimum angle of resolution VA was better in eyes with perfused macula, and the CPT was smaller compared with the parameters of the non-perfused macula (P<0.024 and P<0.001, respectively). There were no significant differences with other parameters (Table 1).
Perfused or non-perfused macula in MO associated with BRVO. (a) Perfused macula has a capillary plexus around the entire circumference, whereas non-perfused macula has the FAZ in contact with the NPA, whether the PCN is disrupted (b) or continuous (c) in fresh BRVO. (d) Old BRVO has capillary dilatation in the affected area and transition zone. Left column: colour fundus photographs. Middle column: corresponding FA images. Right column: magnified images of the macula in FA. White arrowheads indicate FAZ, red arrow disrupted PCN, white arrow PCN between the FAZ and contacting NPA.
We also evaluated the status of the PCN in old BRVO (>1 year after the onset of symptoms) and found that all 11 eyes had fusiform or aneurysmal dilatation of the PCN (Figure 1d). Six eyes had perfused macula, whereas five had non-perfused macula (Table 2).
Perfused macula with SRD, sponge-like swelling, or cystoid spaces at the fovea
We recently found that foveal pathomorphology is associated with capillary lesions around the fovea in diabetic MO,22 which prompted us to investigate the relationship between macular ischaemia and foveal pathomorphology in MO associated with BRVO. Among 34 eyes with perfused macula and fresh BRVO, 14 eyes had the SRD type (Figure 2), 7 the retinal swelling type (Supplementary Figure 1), and 11 the CMO type (Supplementary Figure 2). Most of the last type had fusiform or aneurysmal dilatation of the perifoveal capillaries in the affected area compared with the fine capillary plexus in the other two types.
Perfused macula with the SRD type in fresh BRVO. FA in the early phase (a), its magnified image (b) with perfused macula (white arrowheads), and the late phase (c). (e) An OCT image of the retinal section along the vertical arrow in the infrared image (d) shows SRD (arrowheads) at the foveal centre (black arrow).
Non-perfused fovea associated with foveal cystoid spaces
We further analysed the foveal pathomorphology in the 60 eyes with non-perfused macula and found that 42 eyes had foveal cystoid spaces alone (Figure 3). Among them, 20 eyes had a disrupted PCN, and 22 had a continuous irregularly shaped PCN. In all, 16 of 60 eyes with non-perfused macula had the SRD+CMO type (Supplementary Figure 3), 11 eyes had a continuous PCN, and 5 eyes had a disrupted PCN. When we evaluated the association between macular ischaemia and foveal pathomorphology, we found that eyes with non-perfused macula had foveal cystoid spaces more frequently than those with perfused macula (13/34 eyes (38.2%) vs 58/60 eyes (96.7%), respectively; P<0.001) (Table 1).
Fresh BRVO with non-perfused macula shows the C MO type. FA in the early phase (a), a magnified image (b), and the late phase (c) showing non-perfused macula, in which the FAZ (white arrowheads) is in contact with the non-perfused area (white arrow). (e) An OCT image corresponding to the arrow in the infrared image (d) shows the cystoid spaces (arrowheads) at the foveal centre (arrow).
Old CMO
MO can recur or persist in BRVO,24 which prompted us to investigate the status of the parafoveal capillaries and foveal pathomorphology in MO associated with old BRVO (Supplementary Figure 4). In all, 6 out of 11 eyes with old BRVO had perfused macula and 5 had non-perfused macula (Table 2). Regarding the anatomic morphology, 10 eyes (90.9%) had the CMO type, and 23 of 94 eyes (24.5%) with fresh BRVO did not have foveal cystoid spaces. We further assessed qualitatively the cystoid spaces in the parafovea and found that the cystoid spaces in the INL were predominant in cases of old BRVO compared with those in the outer plexiform layer (OPL)/outer nuclear layer in fresh BRVO.
Discussion
Although the MO associated with retinal vascular diseases often leads to severe vision loss, we do not fully understand the pathogenesis, pathophysiology, and molecular mechanisms. Recent advances in OCT technology have demonstrated the morphological diversity of the MO,15 and the association between the vascular hyperpermeability and pathomorphology of the retinal parenchyma has been investigated.25, 26 Among several mechanisms including the breakdown of the blood-retinal barrier (BRB), mechanical forces, inflammation, and hydrostatic pressure in the current study, we focused on retinal ischaemia,7, 8 and showed for the first time a significant relationship between non-perfused macula and the foveal cystoid spaces in MO associated with fresh BRVO. It suggests the clinical relevance that most eyes without foveal cystoid spaces had perfused macula.
We first addressed the question of how an occluded branch retinal vein results in morphological variations in the PCN. In this current study, we found that 13 (38.2%) of 34 eyes with perfused macula presented with foveal cystoid spaces, although most eyes (58 of 60 eyes, 96.7%) with non-perfused macula had cystoid spaces at the foveal centre. This suggested that the development of foveal cystoid spaces is followed by non-perfusion around the fovea. We therefore speculated that foveal cystoid spaces may compress the parafoveal capillaries, contributing to disrupted blood flow and transient non-perfusion in the capillary beds. The hypothesis might be compatible with the FA findings, that is, that eyes with either the SRD type or the retinal swelling type with perfused macula had a fine capillary plexus around the fovea, whereas non-perfused macula had a dilated PCN, suggesting vascular remodelling. In other words, the topological disequilibrium in parenchymal pressure induced by the cystoid spaces may partly explain the differences between functioning and occluded capillaries. However, we could not exclude the possibility that perfused and non-perfused macula could have different pathogenesis, but not at the different time points in the same sequence, because the times from the onset of symptoms to examination did not differ between them. Further longitudinal observation may elucidate their association.
We further considered how the foveal morphology is determined in MO associated with BRVO. MO is exacerbated by mechanical forces, vascular hyperpermeability, ischaemic changes, metabolic alterations, inflammation, and molecular changes such as growth factors and cytokines.27, 28, 29, 30 Among them, we showed an association between the ischaemia and the foveal cystoid spaces in MO associated with BRVO. Occlusion of the retinal veins results in dysfunctional blood flow; that is, relative ischaemia, which might decrease the exchange of nutrients and wastes. Cytotoxic changes including hypertonicity and concomitant parenchymal swelling could develop as in cerebral oedema.8, 31, 32 Specifically, direct ischaemic effects on the neuroglial cells, including the Müller cells, induce intracellular swelling and their degeneration in the retinal parenchyma, resulting in structural fragility,33, 34, 35 and the cellular dysfunction in the BRB might exacerbate the extracellular fluid, causing vasogenic effects. These changes together might cause development or progression of the cystoid spaces. The pathogenesis of the retinal parenchyma further exacerbates the disrupted microcirculation as discussed previously, which would lead to complete non-perfusion in the retinal capillary beds. The resultant ischaemia in the retinal parenchyma increases the expression of vascular endothelial growth factor, which also induces breakdown of the BRB in the retinal vasculature.27, 36 Ischaemia also modulates the inflammatory response, including an increased interaction between the endothelial cells and leukocytes, which induces disruption of the vascular integrity.37 In addition, increased hydrostatic pressure contributes to the vascular hyperpermeability, according to Starling’s law, and the excessive shear stress also changes the paracellular flux in the vascular endothelium.38
SRD, another pathomorphological change seen on OCT, had been documented previously.16, 39 Intriguingly, in the current case series, most eyes with the SRD type had perfused macula with fresh BRVO, which might agree with the remote effects reported previously.40, 41 Marmor 42reported that vitreous hyperosmolarity can cause SRD and speculated that the flow of water from the vitreous to the choroid, which depends on the difference in osmolarity, is reduced by vitreal hyperosmolarity, resulting in pooling of the subretinal fluid. Similarly, vascular hyperpermeability in an area distant from the macula might increase intravitreal osmolarity, and concomitantly SRD can develop in the fovea in MO associated with BRVO. However, care must be taken when considering why most eyes with the SRD type had perfused macula in the current study. The perfused type had an intact retinal structure, which would guarantee the mechanical integrity, whereas the non-perfused macula could be fragile mechanically because of dysfunction of the structural cells.33, 34, 35 We therefore speculated that hyperosmolarity because of breakdown of the BRB increased the extracellular fluid in the intraretinal spaces of the non-perfused macula or in the subretinal spaces of the perfused macula. Recently, breaks in the outer retinas were reported to contribute to the development of SRDs in macroaneurysms.41 Considering that report, another possible explanation might be that the inner retinal layers of the non-perfused macula are fragile and tend to store the extracellular fluid in the intraretinal spaces, whereas the integrity in the inner layers of the perfused macula increased the possibility of disruption of the outer retinal layers and concomitant SRD. Further molecular and cellular studies would elucidate the exact mechanisms of the development of SRD.
We sometimes observed recurrent or persistent MO associated with BRVO,24 although in the current study none of the 11 eyes had foveal retinal detachment. This suggested that damage in the sensory retina over a longer period of time might increase the possibility of development of cystoid spaces as discussed previously rather than development of SRDs. In addition, the cystoid spaces are mainly in the INL in the parafoveal area in cases of old BRVO compared with those in the OPL in cases with fresh BRVO. Old BRVO was characterized by fluorescein leakage and engorged capillaries in the affected area or collateral vessels in the transition zone. The fluidics affecting vascular permeability also is altered in cases of old BRVO. These vascular changes might contribute to the differences in the location of the cystoid spaces between fresh and old BRVO.
In the current study, we demonstrated that most eyes without foveal cystoid spaces presented perfused macula in MO associated with BRVO, suggesting that capillary perfusion affects retinal oedema and vice versa. As many reports have documented the efficacy of several treatment modalities, the pathogenetic classification in this study may shed light on the development of customized treatments.

References
The Branch Vein Occlusion Study Group. Argon laser photocoagulation for macular edema in branch vein occlusion. Am J Ophthalmol 1984; 98 (3): 271–282.
Branch Vein Occlusion Study Group. Argon laser scatter photocoagulation for prevention of neovascularization and vitreous hemorrhage in branch vein occlusion. A randomized clinical trial. Arch Ophthalmol 1986; 104 (1): 34–41.
Jonas JB, Kreissig I, Degenring RF . Intravitreal triamcinolone acetonide as treatment of macular edema in central retinal vein occlusion. Graefes Arch Clin Exp Ophthalmol 2002; 240 (9): 782–783.
Rosenfeld PJ, Fung AE, Puliafito CA . Optical coherence tomography findings after an intravitreal injection of bevacizumab (avastin) for macular edema from central retinal vein occlusion. Ophthalmic Surg Lasers Imaging 2005; 36 (4): 336–339.
Opremcak EM, Bruce RA . Surgical decompression of branch retinal vein occlusion via arteriovenous crossing sheathotomy: a prospective review of 15 cases. Retina 1999; 19 (1): 1–5.
Murakami T, Takagi H, Kita M, Nishiwaki H, Miyamoto K, Ohashi H et al. Intravitreal tissue plasminogen activator to treat macular edema associated with branch retinal vein occlusion. Am J Ophthalmol 2006; 142 (2): 318–320.
Gass JD . A fluorescein angiographic study of macular dysfunction secondary to retinal vascular disease. II. Retinal vein obstruction. Arch Ophthalmol 1968; 80 (5): 550–568.
Finkelstein D . Ischemic macular edema. Recognition and favorable natural history in branch vein occlusion. Arch Ophthalmol 1992; 110 (10): 1427–1434.
Noma H, Funatsu H, Sakata K, Harino S, Nagaoka T, Mimura T et al. Macular microcirculation and macular oedema in branch retinal vein occlusion. Br J Ophthalmol 2009; 93 (5): 630–633.
Parodi MB, Visintin F, Della Rupe P, Ravalico G . Foveal avascular zone in macular branch retinal vein occlusion. Int Ophthalmol 1995; 19 (1): 25–28.
Gutman FA, Zegarra H . The natural course of temporal retinal branch vein occlusion. Trans Am Acad Ophthalmol Otolaryngol 1974; 78 (2): OP178–OP192.
Michels RG, Gass JD . The natural course of retinal branch vein obstruction. Trans Am Acad Ophthalmol Otolaryngol 1974; 78 (2): OP166–OP177.
Drexler W, Morgner U, Ghanta RK, Kartner FX, Schuman JS, Fujimoto JG . Ultrahigh-resolution ophthalmic optical coherence tomography. Nat Med 2001; 7 (4): 502–507.
Wolf-Schnurrbusch UE, Enzmann V, Brinkmann CK, Wolf S . Morphologic changes in patients with geographic atrophy assessed with a novel spectral OCT-SLO combination. Invest Ophthalmol Vis Sci 2008; 49 (7): 3095–3099.
Otani T, Kishi S, Maruyama Y . Patterns of diabetic macular edema with optical coherence tomography. Am J Ophthalmol 1999; 127 (6): 688–693.
Spaide RF, Lee JK, Klancnik JK, Gross NE . Optical coherence tomography of branch retinal vein occlusion. Retina 2003; 23 (3): 343–347.
Murakami T, Tsujikawa A, Ohta M, Miyamoto K, Kita M, Watanabe D et al. Photoreceptor status after resolved macular edema in branch retinal vein occlusion treated with tissue plasminogen activator. Am J Ophthalmol 2007; 143 (1): 171–173.
Sakamoto A, Nishijima K, Kita M, Oh H, Tsujikawa A, Yoshimura N . Association between foveal photoreceptor status and visual acuity after resolution of diabetic macular edema by pars plana vitrectomy. Graefes Arch Clin Exp Ophthalmol 2009; 247 (10): 1325–1330.
Yamaike N, Tsujikawa A, Ota M, Sakamoto A, Kotera Y, Kita M et al. Three-dimensional imaging of cystoid macular edema in retinal vein occlusion. Ophthalmology 2008; 115 (2): 355–362 e2.
Browning DJ, Glassman AR, Aiello LP, Bressler NM, Bressler SB, Danis RP et al. Optical coherence tomography measurements and analysis methods in optical coherence tomography studies of diabetic macular edema. Ophthalmology 2008; 115 (8): 1366–1371 1371 e1.
Remky A, Wolf S, Knabben H, Arend O, Reim M . Perifoveal capillary network in patients with acute central retinal vein occlusion. Ophthalmology 1997; 104 (1): 33–37.
Murakami T, Nishijima K, Sakamoto A, Ota M, Horii T, Yoshimura N . Foveal cystoid spaces are associated with enlarged foveal avascular zone and microaneurysms in diabetic macular edema. Ophthalmology 2011; 118 (2): 359–367.
Hayreh SS, Rojas P, Podhajsky P . Montague P and Woolson RF. Ocular neovascularization with retinal vascular occlusion-III. Incidence of ocular neovascularization with retinal vein occlusion. Ophthalmology 1983; 90 (5): 488–506.
Ota M, Tsujikawa A, Murakami T, Yamaike N, Sakamoto A, Kotera Y et al. Foveal photoreceptor layer in eyes with persistent cystoid macular edema associated with branch retinal vein occlusion. Am J Ophthalmol 2008; 145 (2): 273–280.
Otani T, Kishi S . Correlation between optical coherence tomography and fluorescein angiography findings in diabetic macular edema. Ophthalmology 2007; 114 (1): 104–107.
Yeung L, Lima VC, Garcia P, Landa G, Rosen RB . Correlation between spectral domain optical coherence tomography findings and fluorescein angiography patterns in diabetic macular edema. Ophthalmology 2009; 116 (6): 1158–1167.
Aiello LP, Bursell SE, Clermont A, Duh E, Ishii H, Takagi C et al. Vascular endothelial growth factor-induced retinal permeability is mediated by protein kinase C in vivo and suppressed by an orally effective beta-isoform-selective inhibitor. Diabetes 1997; 46 (9): 1473–1480.
Cunha-Vaz J, Faria de Abreu JR, Campos AJ . Early breakdown of the blood-retinal barrier in diabetes. Br J Ophthalmol 1975; 59 (11): 649–656.
Cunha-Vaz JG, Travassos A . Breakdown of the blood-retinal barriers and cystoid macular edema. Surv Ophthalmol 1984; 28 (Suppl): 485–492.
Nagai N, Noda K, Urano T, Kubota Y, Shinoda H, Koto T et al. Selective suppression of pathologic, but not physiologic, retinal neovascularization by blocking the angiotensin II type 1 receptor. Invest Ophthalmol Vis Sci 2005; 46 (3): 1078–1084.
Klatzo I . Presidental address. Neuropathological aspects of brain edema. J Neuropathol Exp Neurol 1967; 26 (1): 1–14.
O’Brien MD . Ischemic cerebral edema. A review. Stroke 1979; 10 (6): 623–628.
Tso MO . Pathology of cystoid macular edema. Ophthalmology 1982; 89 (8): 902–915.
Fine BS, Brucker AJ . Macular edema and cystoid macular edema. Am J Ophthalmol 1981; 92 (4): 466–481.
Yanoff M, Fine BS, Brucker AJ, Eagle RC . Pathology of human cystoid macular edema. Surv Ophthalmol 1984; 28 (Suppl): 505–511.
Aiello LP, Pierce EA, Foley ED, Takagi H, Chen H, Riddle L et al. Suppression of retinal neovascularization in vivo by inhibition of vascular endothelial growth factor (VEGF) using soluble VEGF-receptor chimeric proteins. Proc Natl Acad Sci USA 1995; 92 (23): 10457–10461.
Tsujikawa A, Ogura Y, Hiroshiba N, Miyamoto K, Kiryu J, Tojo SJ et al. Retinal ischemia-reperfusion injury attenuated by blocking of adhesion molecules of vascular endothelium. Invest Ophthalmol Vis Sci 1999; 40 (6): 1183–1190.
DeMaio L, Chang YS, Gardner TW, Tarbell JM, Antonetti DA . Shear stress regulates occludin content and phosphorylation. Am J Physiol Heart Circ Physiol 2001; 281 (1): H105–H113.
Ohashi H, Oh H, Nishiwaki H, Nonaka A, Takagi H . Delayed absorption of macular edema accompanying serous retinal detachment after grid laser treatment in patients with branch retinal vein occlusion. Ophthalmology 2004; 111 (11): 2050–2056.
Finkelstein D, Patz A . Distant effect of peripheral branch vein occlusion on the macula. Trans Am Ophthalmol Soc 1988; 86: 380–388.
Tsujikawa A, Sakamoto A, Ota M, Oh H, Miyamoto K, Kita M et al. Retinal structural changes associated with retinal arterial macroaneurysm examined with optical coherence tomography. Retina 2009; 29 (6): 782–792.
Marmor MF . Control of subretinal fluid: experimental and clinical studies. Eye (Lond) 1990; 4 (Part 2): 340–344.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Eye website .
Rights and permissions
About this article
Cite this article
Murakami, T., Tsujikawa, A., Miyamoto, K. et al. Relationship between perifoveal capillaries and pathomorphology in macular oedema associated with branch retinal vein occlusion. Eye 26, 771–780 (2012). https://doi.org/10.1038/eye.2012.85
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2012.85
Keywords
This article is cited by
-
Morphological changes of foveal cysts as a predictor for visual response to anti-vascular endothelial growth factor treatments in diabetic macular edema
International Ophthalmology (2023)
-
Treatment responses for branch retinal vein occlusion predicted by semi-automated fluorescein angiography quantification
BMC Ophthalmology (2022)
-
Choroidal thickening in retinal vein occlusion patients with serous retinal detachment
Graefe's Archive for Clinical and Experimental Ophthalmology (2021)
-
Arteriovenous crossing associated with branch retinal vein occlusion
Japanese Journal of Ophthalmology (2019)
-
Extended field imaging using swept-source optical coherence tomography angiography in retinal vein occlusion
Japanese Journal of Ophthalmology (2018)