Abstract
Usher syndrome is an autosomal recessive disorder characterized by congenital hearing impairment, progressive visual loss owing to retinitis pigmentosa and in some cases vestibular dysfunction. Usher syndrome is divided into three subtypes, USH1, USH2 and USH3. Twelve loci and eleven genes have so far been identified. Duplications and deletions in PCDH15 and USH2A that lead to USH1 and USH2, respectively, have previously been identified in patients from United Kingdom, Spain and Italy. In this study, we investigate the proportion of exon deletions and duplications in PCDH15 and USH2A in 20 USH1 and 30 USH2 patients from Denmark using multiplex ligation-dependent probe amplification (MLPA). Two heterozygous deletions were identified in USH2A, but no deletions or duplications were identified in PCDH15. Next-generation mate-pair sequencing was used to identify the exact breakpoints of the two deletions identified in USH2A. Our results suggest that USH2 is caused by USH2A exon deletions in a small fraction of the patients, whereas deletions or duplications in PCDH15 might be rare in Danish Usher patients.
Similar content being viewed by others
Introduction
Usher syndrome (USH) is an autosomal recessive disorder characterized by congenital hearing impairment, progressive visual loss owing to retinitis pigmentosa (RP) and in some cases, vestibular dysfunction. USH accounts for 50% of the deaf–blind cases and the prevalence has been estimated to be 3.5–16.7 per 100.000.1, 2, 3, 4 USH is genetically and clinically a heterogeneous disorder. On the basis of the degree of hearing loss, onset of RP and effect on the vestibular function USH is clinically divided into three subtypes: USH1, USH2 and USH3. Patients with USH1 have severe to profound congenital hearing impairment, prepubertal onset of RP and vestibular dysfunction.5 In USH2 patients, the hearing loss is moderate to severe, the onset of RP is pre- or postpubertal, and the vestibular function is normal. Patients affected with USH3 have a moderate to severe progressive loss of hearing. RP begins at puberty or later and vestibular function may be affected.6, 7
To date, eleven genes for USH have been identified. Six genes have been identified for USH1 (OMIM 276900): MYO7A (OMIM 276903), USH1C (OMIM 605242), PCDH15 (OMIM 605514), USH1G (OMIM 607696), CDH23 (OMIM 605516) and CIB2 (OMIM 605564).8 Three genes have been identified for USH2 (OMIM 276901): USH2A (OMIM 608400), GPR98 (OMIM 602851) and DFNB31 (OMIM 607084).9 For USH3, two genes have been identified: CLRN1 (OMIM 606397) and HARS (OMIM 614504).10 Furthermore, the USH2 modifier gene, PDZD7, has been recognized.11
Sequence variants affecting the function of the PCDH15 gene, located at chromosome 10q21-22, are identified in 11–19% of the USH1 patients.12, 13, 14 Function-affecting variants in PCDH15 may not only lead to USH1, but also to autosomal recessive nonsyndromic profound hearing impairment (DFNB23).12
Most patients with USH2, including patients from Denmark, have function-affecting variants in USH2A,15, 16, 17, 18, 19 which is located on chromosome 1. Sequence variants in USH2A are also responsible for atypical Usher syndrome and recessive nonsyndromic RP.20 Besides the frequent function-affecting variant, c.2299delG18 in USH2A, a large number of private family-specific ‘function-affecting’, sequence variants have been identified in this gene (https://grenada.lumc.nl/LOVD2/Usher_montpellier/home.php?select_db=USH2A).
To date, only two published studies used multiplex ligation-dependent probe amplification (MLPA) to investigate for exon deletions and duplications in USH genes.21, 22 Investigation of PCDH15 revealed two deletions of 55 kb (exon 3 including the flanking introns) and 82 kb (exon 4–6 including the flanking introns), respectively,21 whereas investigation of USH2A revealed one duplication (from exon 4 to exon 13) and five deletions (exon 4, exons 22-23, exon 27, exon 40 or exon 70 respectively).22
Here we used MLPA to screen a cohort of 50 Danish USH patients (20 USH1 and 30 USH2) for exon deletions and duplications in PCDH15 and USH2A.
Materials and methods
Patients
Fifty Danish unrelated probands diagnosed with USH were included in this study: 20 USH1 and 30 USH2 patients. The patients were diagnosed based on the diagnostic criteria for USH.5
The group of patients was unselected. All USH patients with DNA available at the National Eye Clinic for the Visual Impaired at the Kennedy Center were included. The majority of the patients had not previously been investigated for sequence variants in any USH genes: Thus only eight USH2 patients had two variants in USH2A affecting the function, three USH1 patients had one sequence variant in MYO7A affecting the function and four USH2 patients had one function-affecting variant in USH2A at the start of the study. The remaining 35 patients had no function-affecting sequence variants identified before this investigation. The eight patients with two sequence variants in USH2A were included, as patients with three different function-affecting sequence variants located in two different genes19, 23 as well as patients with three different sequence variants located in the same gene, including USH2A, have been published previously.15, 24
The project was approved by the local ethics committee (H-3-2011-070) and carried out in accordance with the Declaration of Helsinki. Consent to genetic testing was obtained from the patients.
USH2A sequencing
Prior to MLPA, patient 1063 and 55555 were analyzed for sequence variants in USH2A by Sanger sequencing, as described previously.17
MLPA analysis
Genomic DNA was extracted from blood samples using standard methods. The MLPA PCDH15 (P292-A2) kit and the USH2A kit (P361-A1, mix 1 and P362-A2, mix 2), respectively, were obtained from MRC-Holland (Amsterdam, Netherlands). The P292-A2 probe-mix contains two probes for exon 1 and 2, and one probe for each of the remaining 34 exons of PCDH15 with the exception of exon 3, 13, 33 and 35. The two USH2A kits, P361-A1 and P362-A2, have overall probes for each of the 72 exons of USH2A. Details of probe sequences can be found at the company’s website (https://www.mrc-holland.com). MLPA was performed according to the manufacturer’s protocol using 50 ng of genomic DNA per reaction and 1 μl of each reaction product was separated on a POP7 polymer ABI 3130xl capillary sequencer (Applied Biosystems, Foster City, CA, USA). The results were analyzed by comparing the peak profiles using GeneMapper (Applied Biosystems).
Targeted next-generation sequencing of a panel of USH genes
Genomic DNA from patient 70353 was analyzed by targeted next-generation sequencing (NGS) of the USH genes MYO7A, PCDH15, CDH23, USH1C, USH1G, USH2A, GPR98, DFNB31, CLRN1 and PDZD7. The targeted NGS analysis and initial bioinformatics data analysis were conducted by Otogenetics Corporation (Norcross, GA, USA) and delivered through DNAnexus.com together with a report of identified sequence variants. Identified variants were classified and annotated using Alamut v2.4 (Interactive Biosoftware, Rouen, France).
Affymetrix CytoScan HD analysis
The Affymetrix CytoScan HD array (AROS Applied Biotechnology AS, Aarhus, Denmark) was used to confirm the results obtained with MLPA. This array can detect 25–50 kb copy number changes with its 750 000 single-nucleotide polymorphisms (SNPs) and 1 900 000 nonpolymorphic (CNV) markers (Affymetrix, Santa Clara, USA). Data analysis was carried out with Affymetrix Chromosome Analysis Suite Software (ChAS). The data were normalized to the reference model (NA32.1) provided by Affymetrix (284 HapMap samples and 96 phenotypically healthy male and female individuals). The hidden Markov model available within the software package was used to determine the copy number states and to estimate the breakpoints. Thresholds of log2 ratio ⩾0.58 and ⩽−1 were used to categorize altered regions as gains and losses, respectively. In order to exclude false-positive CNVs, only alterations that involved at least 10 consecutive probes for gains and 5 consecutive probes for losses were considered in the analysis. The confidence limit was set to 80%.
Next-generation2 mate-pair sequencing
Mate-pair libraries of DNA from patient 70353 and from patient 1063, respectively, were prepared using the Nextera Mate Pair Sample Preparation Kit (Illumina, San Diego, CA, USA) essentially according to the manufacturer’s protocol. Libraries were quantified and ~40 million fragments from each library were paired-end sequenced (2 × 100 bp) on a HiSeq2000 (Illumina).
Molecular characterization of breakpoints identified by next-generation mate-pair sequencing
Read-pairs were aligned to the human genome (Hg19) using Burrows–Wheeler Aligner (http://bio-bwa.sourceforge.net/). Discordant reads were extracted and structural variations were identified using SVDetect.25 The results allowed us to design primers for breakpoint spanning PCRs. In brief, genomic sequences flanking each of the breakpoints indicated by mate-pair analysis were extracted and masked for repetitive sequences using the UCSC Genomic Browser. The sequences were linked and primers were designed to amplify sequences spanning the breakpoints. PCRs were performed on genomic DNA from the respective probands and from normal controls. PCR conditions are available upon request. The primers used to identify the exon 4–10 deletion breakpoints were: 5′-TGATCTTCTCTAAAGCATCATGAGAGG-3′ and 5′-TGGGAGATATCATCACAGCACAAAATGACA-3′, and those used to identify the exon 22–24 deletion breakpoints were: 5′-AGAACTCTGCCAATCCAGAGGTTCCCA-3′ and 5′-CGGTTTTGGAGGACAACTGCAGCTAAA-3′. The resulting breakpoint spanning sequencing data has been submitted to https://grenada.lumc.nl/LOVD2/Usher_montpellier/home.php?select_db=USH2A (patient IDs 1063 and 55555).
In order to search for microhomology and/or small deletions/insertions flanking the breakpoints, the junction sequences were aligned to the genomic sequence using Multalin (http://multalin.toulouse.inra.fr/multalin/). Variants are described according to USH2A accession number NM_206933.2 with +1 as A in the translation start codon. Exons are numbered according to NG_009497.1.
Relatedness mapping
The estimation of relatedness locally on the genome was carried out with ‘identity by descent’ (IBD) mapping based on SNP data from chromosome 1 around USH2A (CytoScan files) as published previously.26 The CytoScan files were sorted and merged. Sites with missing data or annotation were discarded. The alleles were flipped to the plus strand. The data were merged with data from 112 unrelated CEU individuals (Individuals with European ancestry from the CEPH (Centre d'Etude du Polymorphisme Humain) Utah samples) from HapMap3. Non-overlapping sites were removed, leaving 47 7798 sites. Sites with a minor allele frequency below 5% were removed. Linkage disequilibrium (LD) was accommodated by conditioning on one of 50 previous SNPs with the largest amount of LD. The most likely combination of IBD states across the chromosome was estimated using the Viterbi algorithm.
Results
A cohort of 50 Danish unrelated patients with Usher syndrome, 20 USH1 and 30 USH2, was investigated for exon duplications and deletions in USH2A and PCDH15 by MLPA. All patients were investigated by both kits because of the well known clinical overlap between the different subtypes of USH.27
No deletions or duplications were identified in PCDH15. In contrast, there were abnormal relative peak areas at about 0.5 for several exons of USH2A in three USH2 patients, indicating the presence of a heterozygous deletion (Figure 1). According to the MLPA data, two patients had a deletion of exons 4–10 (patients 70353 and 55555) and one patient had a deletion covering exons 22–24 (patient 1063). Patients 55555 and 1063 also had other function-affecting sequence variants in USH2A, found by a previous Sanger sequencing of USH2A. Thus, patient 55555 had the previously published variant c.2299delG, p.(Glu767Serfs*21) in heterozygous state.17 Furthermore, we here identified the variant c.3309C>A, p.(Tyr1103*) in heterozygous state in patient 1063. This variant designated rs397518011 (http://www.ncbi.nlm.nih.gov/snp) has previously been classified as ‘affects function’ (HGMD Professional 2014.2, August 2014, (http://www.hgmd.org/). To search for a second function-affecting variant in the third patient, 70353, targeted NGS of USH genes led to the identification of the USH2A sequence variant c.10684G>T, p.(Glu3562*) in heterozygous state. This variant, also classified as ‘affects function’, has previously been identified as disease causing when present in a specific context in USH2 patients (HGMD Professional 2, August 2014, http://www.hgmd.org/). The results are summarized in Table 1.
CytoScan analysis of the three samples confirmed the deletions in USH2A in all three patients (Figure 1). The deletions in patients 70353, 55555 and 1063 were localized to chr1.hg19: g.216,473,521_216,535,711; chr1.hg19:g.216,473,521_216,536,791; and chr1.hg19:g. 216,260,845_216,326,858, respectively.
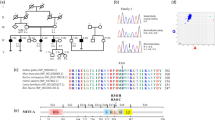
To identify the exact deletion breakpoints, we performed next-generation mate-pair sequencing on genomic DNA from patients 70353 and 1063 (Supplementary Figure 1). We were able to design PCR primers that amplified across the deletion breakpoints using DNA from the two patients. Subsequent sequencing identified the respective breakpoints (Figure 2). At the protein level, the deletion of USH2A exons 22–24 is predicted to be in frame p.(Gly1543_Pro1662del). The deletion of exons 4–10 in patient 70353 partially deleted exon 4 (including the MLPA probe ligation site) by deleting the last 113 bases of exon 4 (and keeping the first 20 bases intact). On the protein level, this deletion is predicted to produce a frameshift from amino acid 224: p.(Ser224Argfs*5).
Mapping of two deletion breakpoints in USH2A. On the basis of the next-generation mate-pair sequencing results, PCR primers were designed to amplify across the deletion breakpoints from genomic DNA from patients 70353 and 1063. (a) Sequence chromatogram showing that patient 70353 (and 55555) have a 74 052-bp deletion (chr1.hg19:g.216464358_216538408del) with breakpoints in exon 4 and in intron 11. (b) Sequence chromatogram showing that patient 1063 has a 63 758-bp deletion (chr.hg19:g.216259403_216323159del) with breakpoints in intron 21 and in intron 24.
To test whether the breakpoint in patient 55555 was located at the same position as the breakpoint in patient 70353, we performed PCR on DNA from patient 55555 using the same primers as used for PCR amplification of the breakpoint in patient 70353. Interestingly, the procedure revealed identical breakpoints. We therefore carried out two different inquiries to substantiate whether the deletion had occurred once in a common ancestor or on two independent occasions. In one approach, we used traditional genealogy. For both individuals, we have established their ancestry back to 1850, 4–6 generations, without finding a common ancestor. In a second approach, we used a statistical method to infer relatedness locally across the genome.26 Individuals who share an inherited (family specific) sequence variant have a high degree of similarity in the region around the variant. Relatedness mapping based on SNPs in the region containing USH2A on chromosome 1 revealed a high probability of relatedness in the region located between 206 and 218 Mb (Figure 3), indicating a common ancestor carrying the deletion. As the similarity (relatedness) was low in the rest of the investigated region, the possible common ancestor of patients 70353 and 55555 probably lived many generations back, which agrees with the genealogy. Note, the deletion in USH2A is located around chr1.hg19:g.216,464,000_216,538,000 (see Figure 3).
Estimation of relatedness between patient 70353 and patient 55555 based on investigation of SNP data from chromosome 1. Plots of relatedness across chromosome 1 indicate that the region from 206 to 218 Mb on one of the two alleles with very high probability is identical by decent, and thus inherited from a common ancestor. The orange line is the probability for being related for one allele and unrelated for the other (here 100% in USH2A). The blue line is the probability for being unrelated (here 0% in USH2A). The green line indicating that both alleles exhibit identity by descent (IBD) has 0% probability across the chromosome and is almost not visible. The most likely IBD states across the chromosome are indicated by a bar above the plot, showing that the two individuals are only related at the USH2A locus (black arrow).
Disussion
MLPA revealed two deletions in USH2A: one deletion spanning exons 4–10 and another deletion spanning exons 22–24. A deletion of exons 22–24 in USH2A has been described previously by Baux et al.28 and deletion breakpoints that differs only by 2 bp (AT) compared with the present case was submitted to https://grenada.lumc.nl/LOVD2/Usher_montpellier/home.php?select_db=USH2A in 2012 (DB-ID USH2A_00095; Patient data: #0000067). In two apparently unrelated patients, 70353 and 55555, we identified a deletion of exons 4–10 with exactly the same breakpoints (c.672_1840+1160del). Interestingly, we found evidence for the existence of a common ancestor in which the deletion had occurred many generations back, as we were not able to identify a common ancestor by traditional genealogy going back to 1850, yet SNP mapping revealed possible relatedness. A deletion of exons 4–10 has—to our knowledge—not been described previously.
Mate-pair NGS was used as a fast and efficient method for confirmation and directed cloning of the MLPA suggested breakpoints. This method is particularly useful in cases of breakpoints in large introns making long range PCR difficult and in cases where no tissue for RT-PCR studies are available for further studies of the consequences of the deletions at the mRNA level.
At both breakpoints, we observed microhomology of 2 bp (Supplementary Figure 2). We speculate whether the mechanism behind these deletions could be non-homologous end-joining (NHEJ). NHEJ does not require the presence of extensive homology but can be facilitated by the presence of microhomology (1–4 bp).29 Another mechanism could be microhomology-mediated end-joining (MMEJ), but normally a longer stretch of microhomology (5–25 bp) is required for MMEJ to occur.30, 31
A total of 435 different sequence variants in USH2A have been registered in HGMD Professional (August, 2014). The majority, >50%, consists of missense or nonsense variants, but gross deletions, duplications or complex rearrangements have also been identified and represent about 5% of the variants. In our patients, we found deletions and duplications of the same magnitude, as we identified deletions in USH2A in three USH2 patients, corresponding to 10% of the USH2 patients (3/30). However, as patient 70353 and 55555 have inherited the same USH2A allele, we only identified a deletion in two different alleles corresponding to 3.33% of the alleles (2/60). No duplications were identified. Steele et al.22 identified also USH2A deletions with similar frequency, as deletions were identified in 5% (7/121) unselected USH2 patients.
It has been reported that large deletions or duplications within PCDH15 are frequent causes for USH1.32 To date, 71 different PCDH15 sequence variants are registered in HGMD Professional (August, 2014). Interestingly, partial gene deletions and duplications account for a relatively large fraction, that is, ~17% of the 71 sequence variants identified in PCDH15. However, we could not identify any deletion or duplication in PCDH15 in this study, but recognize that only 20 USH type 1 patients were analyzed. As the P292 kit for PCDH15 does not include probes for all exons of PCDH15, we cannot exclude that deletions or duplications of any of these exons might be present in our cohort of patients. Aller et al.21 identified deletions/duplications in PCDH15 in 13% of 23 USH1 patients by combining MLPA and CGH procedures. The 23 USH1 patients represent a cohort of selected (remaining) patients, with one or no identified function-affecting sequence variants after screening a larger number of patients by Sanger sequencing for variants in known USH genes. In contrast, our cohort was unselected. Inclusion did not depend on whether the patient had been subjected to molecular genetic tests previously or whether any function-affecting sequence variant was identified, all available patients were included. Our unselected and relatively small USH cohort might, at least partly, explain the absence of PCDH15 deletions in our patients.
Interestingly, we found a MLPA signal of about 0.5 for USH2A exon 12 in one patient and for exon 19 in another patient, indicating a deletion of the exon. However, by sequencing exon 12 and exon 19 in the two patients, we found a function-affecting variant c.2023C>T, p.(Gln675*) in exon 12 and the variant c.4106C>T, p.(Ser1369Leu) (rs201709513) in exon 19, both in heterozygous form, indicating that neither exon 12 nor exon 19 was deleted. Investigation of the location of the MLPA probe in exon 12 and in exon 19 (available from www.mlpa.com) revealed that c.2023C and c.4106C are located in ligation regions, indicating a false-positive MLPA result. Changes in the target sequence have previously been shown to lead to false-positive MLPA results.33 Great care must therefore be exercised, especially when MLPA results indicate the deletion of a single exon.
MLPA would be a useful technique to test for deletions and duplications in other USH genes as well. Exon deletions or duplications in any of the other genes might probably contribute to USH as well. So far, only very few exon deletions/duplications have been identified in MYO7A, USH1C, CDH23, CLRN1 and GPR98, whereas none have been identified in USH1G and DFNB31 according to HGMD Professional. The absence of commercially available MLPA kits that target these genes is probably the main reason for this.
Change history
12 November 2015
This paper has been corrected since online publication and a corrigendum also appears in this issue
References
Boughman JA, Vernon M, Shaver KA : Usher syndrome: definition and estimate of prevalence from two high-risk populations. J Chronic Dis 1983; 36: 595–603.
Rosenberg T, Haim M, Hauch AM, Parving A : The prevalence of Usher syndrome and other retinal dystrophy-hearing impairment associations. Clin Genet 1997; 51: 314–321.
Spandau UH, Rohrschneider K : Prevalence and geographical distribution of Usher syndrome in Germany. Graefes Arch Clin Exp Ophthalmol 2002; 240: 495–498.
Kimberling WJ, Hildebrand MS, Shearer AE et al: Frequency of Usher syndrome in two pediatric populations: Implications for genetic screening of deaf and hard of hearing children. Genet Med 2010; 12: 512–516.
Smith RJ, Berlin CI, Hejtmancik JF et al: Clinical diagnosis of the Usher syndromes. Usher Syndrome Consortium. Am J Med Genet 1994; 50: 32–38.
Millan JM, Aller E, Jaijo T, Blanco-Kelly F, Gimenez-Pardo A, Ayuso C : An update on the genetics of usher syndrome. J Ophthalmol 2011; 2011: 417217.
Saihan Z, Webster AR, Luxon L, Bitner-Glindzicz M : Update on Usher syndrome. Curr Opin Neurol 2009; 22: 19–27.
Riazuddin S, Belyantseva IA, Giese AP et al: Alterations of the CIB2 calcium- and integrin-binding protein cause Usher syndrome type 1 J and nonsyndromic deafness DFNB48. Nat Genet 2012; 44: 1265–1271.
Petit C : Usher syndrome: from genetics to pathogenesis. Annu Rev Genomics Hum Genet 2001; 2: 271–297.
Puffenberger EG, Jinks RN, Sougnez C et al: Genetic mapping and exome sequencing identify variants associated with five novel diseases. PLoS One 2012; 7: e28936.
Ebermann I, Phillips JB, Liebau MC et al: PDZD7 is a modifier of retinal disease and a contributor to digenic Usher syndrome. J Clin Invest 2010; 120: 1812–1823.
Ahmed ZM, Riazuddin S, Ahmad J et al: PCDH15 is expressed in the neurosensory epithelium of the eye and ear and mutant alleles are responsible for both USH1F and DFNB23. Hum Mol Genet 2003; 12: 3215–3223.
Ouyang XM, Yan D, Du LL et al: Characterization of Usher syndrome type I gene mutations in an Usher syndrome patient population. Hum Genet 2005; 116: 292–299.
Roux AF, Faugere V, Le GS et al: Survey of the frequency of USH1 gene mutations in a cohort of Usher patients shows the importance of cadherin 23 a nd protocadherin 15 genes and establishes a detection rate of above 90%. J Med Genet 2006; 43: 763–768.
Dreyer B, Tranebjaerg L, Rosenberg T, Weston MD, Kimberling WJ, Nilssen O : Identification of novel USH2A mutations: implications for the structure of USH2A protein. Eur J Hum Genet 2000; 8: 500–506.
Dreyer B, Tranebjaerg L, Brox V et al: A common ancestral origin of the frequent and widespread 2299delG USH2A mutation. Am J Hum Genet 2001; 69: 228–234.
Dreyer B, Brox V, Tranebjaerg L et al: Spectrum of USH2A mutations in Scandinavian patients with Usher syndrome type II. Hum Mutat 2008; 29: 451.
Le Quesne SP, Saihan Z, Rangesh N et al: Comprehensive sequence analysis of nine Usher syndrome genes in the UK National Collaborative Usher Study. J Med Genet 2012; 49: 27–36.
Besnard T, Vache C, Baux D et al: Non-USH2A mutations in USH2 patients. Hum Mutat 2012; 33: 504–510.
Rivolta C, Sweklo EA, Berson EL, Dryja TP : Missense mutation in the USH2A gene: association with recessive retinitis pigmentosa without hearing loss. Am J Hum Genet 2000; 66: 1975–1978.
Aller E, Jaijo T, Garcia-Garcia G et al: Identification of large rearrangements of the PCDH15 gene by combined MLPA and a CGH: large duplications are responsible for Usher syndrome. Invest Ophthalmol Vis Sci 2010; 51: 5480–5485.
Steele-Stallard HB, Le Quesne SP, Lenassi E et al: Screening for duplications, deletions and a common intronic mutation detects 35% of second mutations in patients with USH2A monoallelic mutations on Sanger sequencing. Orphanet J Rare Dis 2013; 8: 122.
Yoshimura H, Iwasaki S, Nishio SY et al: Massively parallel DNA sequencing facilitates diagnosis of patients with Usher syndrome type 1. PLoS One 2014; 9: e90688.
Huang XF, Xiang P, Chen J et al: Targeted exome sequencing identified novel USH2A mutations in Usher syndrome families. PLoS One 2013; 8: e63832.
Zeitouni B, Boeva V, Janoueix-Lerosey I et al: SVDetect: a tool to identify genomic structural variations from paired-end and mate-pair sequencing data. Bioinformatics 2010; 26: 1895–1896.
Albrechtsen A, Sand KT, Moltke I, van Overseem HT, Nielsen FC, Nielsen R : Relatedness mapping and tracts of relatedness for genome-wide data in the presence of linkage disequilibrium. Genet Epidemiol 2009; 33: 266–274.
Jaijo T, Aller E, Garcia-Garcia G et al: Microarray-based mutation analysis of 183 Spanish families with Usher syndrome. Invest Ophthalmol Vis Sci 2010; 51: 1311–1317.
Baux D, Larrieu L, Blanchet C et al: Molecular and in silico analyses of the full-length isoform of usherin identify new pathogenic alleles in Usher type II patients. Hum Mutat 2007; 28: 781–789.
Lieber MR : The mechanism of human nonhomologous DNA end joining. J Biol Chem 2008; 283: 1–5.
McVey M, Lee SE : MMEJ repair of double-strand breaks (director's cut): deleted sequences and alternative endings. Trends Genet 2008; 24: 529–538.
Ma JL, Kim EM, Haber JE, Lee SE : Yeast Mre11 and Rad1 proteins define a Ku-independent mechanism to repair double-strand breaks lacking overlapping end sequences. Mol Cell Biol 2003; 23: 8820–8828.
Le GS, Faugere V, Malcolm S, Claustres M, Roux AF : Large genomic rearrangements within the PCDH15 gene are a significant cause of USH1F syndrome. Mol Vis 2007; 13: 102–107.
Moller LB, Ott P, Lund C, Horn N : Homozygosity for a gross partial gene deletion of the C-terminal end of ATP7B in a Wilson patient with hepatic and no neurological manifestations. Am J Med Genet A 2005; 138: 340–343.
Acknowledgements
We thank the families for their participation. Jette Bune Rasmussen is acknowledged for the photographic assistance. Lone Sandbjerg Hindbæk is thanked for the excellent technical help and Susan Peters for proofreading the manuscript. The NGS mate-pair analysis took place at ICMM. This work was supported by the Oticon Foundation, the Jascha Foundation, the Dag Lenard Foundation, the Augustinus Foundation, and the Director Jacob Madsen and wife Olga Madsen Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on European Journal of Human Genetics website
Supplementary information
Rights and permissions
About this article
Cite this article
Dad, S., Rendtorff, N., Kann, E. et al. Partial USH2A deletions contribute to Usher syndrome in Denmark. Eur J Hum Genet 23, 1646–1651 (2015). https://doi.org/10.1038/ejhg.2015.54
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejhg.2015.54
This article is cited by
-
The role of CNVs in the etiology of rare autosomal recessive disorders: the example of TRAPPC9-associated intellectual disability
European Journal of Human Genetics (2018)