Abstract
Polyglutamine (polyQ) disorders are inherited neurodegenerative conditions defined by a common pathogenic CAG repeat expansion leading to a toxic gain-of-function of the mutant protein. Consequences of this toxicity include activation of heat-shock proteins (HSPs), impairment of the ubiquitin-proteasome pathway and transcriptional dysregulation. Several studies in animal models have shown that reducing levels of toxic protein using small RNAs would be an ideal therapeutic approach for such disorders, including spinocerebellar ataxia-7 (SCA7). However, testing such RNA interference (RNAi) effectors in genetically appropriate patient cell lines with a disease-relevant phenotype has yet to be explored. Here, we have used primary adult dermal fibroblasts from SCA7 patients and controls to assess the endogenous allele-specific silencing of ataxin-7 by two distinct siRNAs. We further identified altered expression of two disease-relevant transcripts in SCA7 patient cells: a twofold increase in levels of the HSP DNAJA1 and a twofold decrease in levels of the de-ubiquitinating enzyme, UCHL1. After siRNA treatment, the expression of both genes was restored towards normal levels. To our knowledge, this is the first time that allele-specific silencing of mutant ataxin-7, targeting a common SNP, has been demonstrated in patient cells. These findings highlight the advantage of an allele-specific RNAi-based therapeutic approach, and indicate the value of primary patient-derived cells as useful models for mechanistic studies and for measuring efficacy of RNAi effectors on a patient-to-patient basis in the polyQ diseases.
Similar content being viewed by others
Introduction
Polyglutamine (polyQ) disorders are a group of neurodegenerative diseases caused by a common CAG repeat expansion mutation, which confers a toxic gain-of-function on the translated protein.1 They are characterised by the late onset of clinical symptoms, and a progressive, unrelenting phenotype caused by specific neuronal degeneration. To date, nine polyQ disorders have been identified, including Huntington disease (HD), dentatorubralpallidoluysian atrophy, spinal bulbar muscular atrophy and spinocerebellar ataxias (SCA) 1, 2, 3, 6, 7 and 17.
Mutant polyQ proteins have been shown to cause a multitude of downstream pathogenic events resulting in neuronal degeneration including aggregation of mutant proteins, aberrant protein interactions, impairment of misfolded protein clearance pathways and transcriptional dysregulation.1 Pre-empting these downstream events by inhibiting formation of the toxic mutant protein would thus be a favourable therapeutic strategy.
The selection of appropriate models of disease which are easy to access and manipulate, and which accurately represent the genetic context of the mutation, remains one of the major challenges in the development of effective therapies for neurodegenerative conditions. Several studies have demonstrated the potential of RNA interference (RNAi) based silencing as a therapeutic approach for HD, SCA1, SCA3, SCA6 and SCA7.2, 3, 4, 5, 6, 7, 8, 9, 10, 11 However, most of these reports have used overexpression models or transgenic animal models to demonstrate efficacy—models which fail to mimic the precise genomic context of mutation found in patients. These models have also failed to provide answers to important questions regarding the necessity and feasibility of allele-specific silencing of the mutant transcript alone, without concomitant silencing of the wild type. Thus, genetically accurate patient-derived cell lines would provide ideal models—both for therapeutic screening and for addressing the requirement for allele specificity. The demonstration of a disease-relevant phenotype in easily accessible peripheral cells, such as fibroblasts, which could be used to measure the efficacy of therapeutic molecules, would further enhance the value of such a model.
SCA7 occurs at an unusually high prevalence in the South African population, the result of a founder effect in patients of Black African ethnic origin.12 Consequently, South African SCA7 patients display a unique phenomenon—a SNP (rs3774729) within the causative gene, ataxin-7, is linked to the mutation in all patients studied to date, with approximately 43% of these individuals heterozygous for the polymorphism (genotype data publically available in the next build of dbSNP, B140, at https://www.ncbi.nlm.nih.gov/SNP/).13 This SNP provides an ideal target for developing an allele-specific silencing therapy. We have previously shown that allele-specific RNAi treatment removes polyQ aggregations in an over-expression cell model of SCA7.10 Here we have developed a method for measuring endogenous allele-specific silencing and show selective knockdown of the mutant ataxin-7 transcript using siRNAs targeting a SNP linked to the polyQ expansion in South African SCA7 patients. We further identified two transcripts, differentially expressed in SCA7 patient dermal fibroblasts compared with controls, whose expression could be restored towards normal levels following administration of RNAi—providing preliminary evidence of the restoration of a disease-relevant phenotype in patient cells by targeting a common SNP.
These results demonstrate that SCA7 patient-derived fibroblast cell lines are a valuable tool for screening therapeutic molecules, and further support the superiority of an allele-specific silencing approach.
Materials and methods
Sizing of ataxin-7 CAG repeat
PCR primers were designed to flank the CAG repeat region in the ataxin-7 gene (ENSG00000163635) and used in the following 10 μl reaction: 1 × Failsafe Premix buffer J (Epicentre Biotechnologies, Madison, WI, USA), 0.4 μ M primers (forward and reverse), 0.6U GoTaq DNA polymerase (Promega, Madison, WI, USA), 100 ng DNA. Primer sequences: forward primer 5′-HEX-ATTGTAGGAGCGGAAAGAATG-3′; reverse primer 5′-CCAGCATCACTTCAGGAC-3′. Cycling conditions: 95 °C for 5 min; 30 cycles of 95 °C for 30 s, 53 °C for 6 s and 72 °C for 40 s; single cycle of 72 °C for 7 min. All PCR products were analysed by capillary electrophoresis on the ABI 3100 Genetic Analyzer (Applied Biosystems, Carlsbad, CA, USA). The GeneScan 500 Rox Size Standard (Applied Biosystems) was used and results were analysed using GeneMapper software (version 3.0, Applied Biosystems). Repeat sizes were calculated using the formula y=0.3063x−76.475 (adapted from Dorschner et al14).
Genotyping of SNP rs3774729
The PCR was carried out in a 25-μl reaction mixture with 1 × GoTaq Buffer (Promega), 200 μ M dNTPs (Bioline, London, UK), 0.4 μ M of each primer, 0.5U GoTaq Polymerase (Promega) and 100 ng DNA. Cycling conditions were 95 °C for 5 min; 30 cycles of 95 °C for 30 s, 61 °C for 30 s and 72 °C for 40 s; single cycle of 72 °C for 7 min. Forward primer 5′-AATGAACTGCCTGTCAACTC-3′, reverse primer 5′-GCTCACAGTCCATTTCCTAC-3′. PCR products were sequenced using a standard cycle sequencing reaction and conditions. In a 10-μl reaction mixture: 1 × BigDye Terminator Sequencing Buffer (Applied Biosystems), 1 μl BigDye Terminator v3.1 Cycle Sequencing RR-100 (Applied Biosystems), 0.4 μ M forward primer, 2 μl PCR product. Cycling: 98 °C for 5 min, followed by 25 cycles of 96 °C for 10 s, 50 °C for 15 s, 60 °C for 4 min. Sequencing products were precipitated with ethanol and analysed on the ABI 3100 Genetic Analyzer. Sequence alignments were performed using the BioEdit Sequence Alignment Editor version 7.0.9.0 (Ibis Biosciences, Carlsbad, CA, USA).
Cell culture and transfections
Written informed consent was obtained from patients before acquiring skin biopsies (University of Cape Town Human Research Ethics Committee reference number 460/2010, renewed annually). Control lines were obtained from the Coriell Institute for Medical Research. Patient fibroblasts were isolated by sterile 4 mm punch biopsy using standard procedures.15 The resulting cells were dissociated manually and by enzymatic treatment and grown in DMEM with 10% fetal calf serum (Invitrogen, Carlsbad, CA, USA) to establish fibroblast cell lines. These lines were cultured at 37 °C with 5% CO2. Cells were transfected with one of two siRNAs, allele-specific siR-P16 or non-allele-specific siR-atxn7. SiR-P16 was designed to mismatch the SNP at position 16 with position 1 designated as the first nucleotide on the 5′ end of the guide strand, as previously published.10 siR-atxn7 was designed by Juerg Hunziker, Novartis. Both siRNAs were supplied by Novartis (Basel, Switzerland).
Transfection was performed using the Neon Transfection System (Invitrogen) according to manufacturer’s instructions and under the following modified electroporation conditions: pulse width=20, voltage=1700, pulse number=1. Cells were harvested for RNA extraction 4 or 7 days after electroporation.
RNA isolation and quantitative PCR (qPCR)
RNA was isolated using the High Pure RNA Isolation kit (Roche Applied Sciences, Penzberg, Germany) and cDNA synthesised using the High Capacity cDNA reverse transcription kit (Applied Biosystems). qPCR assays were completed using the Rotor-Gene 6000 real-time rotary analyser (Qiagen, Venlo, The Netherlands) and the SensiMix SYBR kit (Bioline). Primers for ataxin-7, DNAJA1, Hsp27, Hsp70, Hsp150, UCHL1 and β-actin were designed and validated by PrimerDesign (Southampton, UK). In each case, gene expression was determined relative to β-actin. Allele-specific primers were designed and validated to specifically amplify the A or G allele of the ataxin-7 SNP (rs3774729). For optimisation purposes, cDNA samples of all three genotypes were used to validate the specificity of the qPCR. Template quality was confirmed before cDNA synthesis; cDNA synthesis per data set took place in the same reaction set up to minimise variation. All primers were optimised before use. Each qPCR experiment was performed in experimental duplicate relative to β-actin; a standard curve for both the gene of interest and β-actin was performed for every set of results to take into account the small changes in experimental efficiency between data sets, including variations in cDNA synthesis quality. Primer reproducibility was optimised by ensuring the PCR efficiency demonstrated by each standard curve approached 100%, with an r2 value >0.998, and a gradient of −3.4. Data from experimental samples were only used if the values fell within the range of the standard curve.
Immunostaining
Fibroblasts grown on coverslips were washed three times with 1 × PBS and fixed with 4% paraformaldehyde for 30 min at room temperature. Thereafter, cells were washed three times with 1 × PBS and incubated with blocking/permeabilisation solution (5% fetal bovine serum, 0.01% Triton-X in 1 × PBS) for 1 h at room temperature. The primary antibody (Ataxin-7 polyclonal antibody, Thermo Scientific, Waltham, MA, USA) was added at a 1:400 dilution in blocking solution and incubated overnight at 4 °C. The following day, cells were washed three times with 1 × PBS and incubated with secondary antibody (1:500 dilution, Cy3 Donkey anti Rabbit IgG, Jackson ImmunoResearch, West Grove, PA, USA) for 2 h at room temperature. Finally, cells were stained with Hoechst solution (0.5 μg/ml) for 5 min and washed three times with 1 × PBS. Coverslips were mounted with Mowiol and observed with an Axiovert 200M microscope (Zeiss, Oberkochen, Germany). Images were analysed using AxioVision 4.8 software (Zeiss).
Statistical analysis
Transfections were carried out in triplicate. Each qPCR reaction was performed in duplicate. All P-values were calculated using a one- or two-tailed t-test where appropriate.
Results
Allele-specific silencing of endogenous ataxin-7
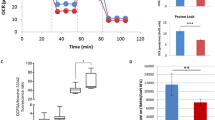
A critical question in an RNAi-based therapy is whether allele-specific or non-allele-specific approaches should be adopted. We have previously shown that an siRNA targeting mutant ataxin-7 (siR-P16) is allele specific in an over-expression cellular model,10 targeting a common SNP linked to the mutation.13 The patient cell line used in this study is heterozygous for the same SNP present in the South African population (Supplementary Figure 1). To assess allele-specific silencing in primary cells, we therefore designed and validated qPCR primers capable of specifically amplifying each allele of the SNP. A common reverse primer, placed 3′ to the SNP, was used together with a forward primer matching either the A or G allele of the SNP to amplify the mutant and wild-type transcripts, respectively (Supplementary Figure 2). The qPCR was validated to be specific in both reactions, that is, no product was detected using G allele primers on homozygous AA samples, nor did A allele primers detect a product using homozygous GG samples (Supplementary Figure 2). siR-atxn7 is a non-allele-specific siRNA, which targets the 3′ UTR of both mutant and wild-type ataxin-7, whereas siR-P16 has been designed to specifically silence the A allele associated with the mutant transcript, with reduced activity against the G allele associated with the wild-type transcript (Figure 1a). Allele-specific qPCR showed that siR-atxn7 silences both the mutant and wild-type transcripts with similar efficacy across all three doses (Figure 1b). In contrast, siR-P16 has no silencing effect on the wild-type transcript at 1 nM, yet silences the mutant transcript by 34% (P<0.05; Figure 1c). This allele specificity is maintained at 5 nM; silencing the mutant transcript by 49% and the wild-type by only 25%. Allele specificity is only ablated at the highest dose of 20 nM, although saturating effects at higher doses are well established.
Allele-specific silencing in patient fibroblasts. (a) Schematic showing the allele-specific design of siR-P16 targeting the A allele of SNP rs3774729, and a non-allele-specific siRNA, siR-atxn7, targeting the 3′ UTR. The CAG repeat expansion ((CAG)ex) and the normal sized repeat ((CAG)n) are linked to the A and G allele of the SNP, respectively. (b) Levels of wild-type allele are not significantly different from levels of mutant allele transcript following treatment with siR-atxn7. (c) Treatment with siR-P16 reduces levels of mutant allele at a similar rate to that of siR-atxn7. Levels of wild-type allele are significantly unaffected by siR-P16 over low doses. All qPCR values are relative to that of a non-specific siRNA at the equivalent concentrations. Results are compiled from an n=2 (experimental).
Identification of transcriptional differences between SCA7 and control fibroblasts
SCA7 patient fibroblasts show no sign of gross morphological abnormalities, including morphology, doubling time and disease protein aggregation, consistent with the notion that these cells do not contribute to the patient disease phenotype (Supplementary Figure 4). However, given the ubiquitous expression of Ataxin-716 and its role in gene expression regulation through association with the widely expressed SPT3/TAF9/GCN5 acetyltransferase (STAGA) histone acetylation complex,17 it is likely that many cell types (including those of unaffected tissues) may show some sign of the transcriptional alterations, considered early manifestations of disease.18
We thus sought to identify gene expression differences between SCA7 patient and control dermal fibroblasts, which might act as markers of disease progression and therapeutic efficacy. We chose six candidate transcripts previously shown to be differentially expressed in SCA7 models of disease and linked to general, rather than neuronal- or retinal-specific, pathways involved in polyQ pathogenesis.19, 20 These included proteins involved in cellular stress, such as the HSPs-27, -70, -105 and DNAJ/HSP40 homologue, subfamily A, member 1 (DNAJA1) as well as an RNA chaperone, RNA-Binding Motif protein 3 (RBM3); and the ubiquitin carboxy-terminal hydrolase L1 (UCHL1), a protein involved in de-ubiquitination. qPCR analysis was performed to compare transcript levels of these six genes between multiple control (n=2) and SCA7 patient (n=3) fibroblast cell lines (Figure 2). Consistent with the SCA7 models previously described,19 we observed a twofold increase in DNAJA1 (corresponding to an early upregulation in the heat-shock response observed in 6-month-old SCA7 transgenic mice) and a twofold decrease in UCHL1 in SCA7 patient fibroblasts relative to control lines. We observed no significant changes in levels of Hsp27, -70, -105 and RBM3.
Transcriptional changes in SCA7 fibroblasts. qPCR analysis of endogenous transcripts is shown relative to β-actin. Control fibroblasts, n=2; SCA7 fibroblasts, n=3; these numbers represent individual cell lines. UCHL1, ubiquitin carboxy-terminal hydrolase LI; Hsp-, heat-shock protein-27, -70, -105; DNAJA1 (HSP40), DNAJ homologue, subfamily A, member 1; RBM3, RNA-binding motif protein 3. *P value<0.05.
Effect of ataxin-7 silencing on DNAJA1 and UCHL1 expression
To assess whether the above-mentioned transcriptional changes were affected by inhibiting production of mutant polyQ protein, fibroblast cell lines were transfected with siR-atxn7 or siR-P16. For comparative purposes, total ataxin-7 levels were used as a measure of gene silencing efficiency, as mutant allele levels could not be assessed in control fibroblasts. Both siRNAs were found to silence total ataxin-7 in a dose-dependent manner 7 days after treatment, ranging from 21 to 77% knockdown for siR-P16, and 29 to 68% for siR-atxn7, as measured by qPCR (Figure 3a).
Ataxin-7 silencing normalises dysregulated transcripts in SCA7 patient cells. (a) Two siRNAs (siR-atxn7 and siR-P16) silence total ataxin-7 transcript levels in a dose-dependent manner relative to a non-specific siRNA (siR-NS). (b) Corresponding levels of DNAJA1 expression after treatment with increasing doses of siR-P16 and siR-atxn7. (c) Corresponding levels of UCHL1 expression after treatment with increasing doses of siRP16 and siR-atxn7. Concentrations of siRNAs are shown on the x axis. *P value<0.05. Measurements of expression levels are taken from the same sample cohort as those demonstrating allele- or non-allele-specific silencing in Figure 1. Results are compiled from one primary cell line, experiment performed in duplicate.
We then measured expression levels of DNAJA1 and UCHL1 7 days post ataxin-7 silencing. At low levels of silencing (less than 50%), there was no significant change in DNAJA1 levels (Figure 3b). However, when silencing of over 50% was achieved, expression of DNAJA1 was significantly reduced (P<0.05) by 30% (siR-P16) and 41% (siR-atxn7)—approaching the levels observed in control fibroblasts. Interestingly, robust silencing by both siRNAs did not normalise levels of DNAJA1 if assessed at a shorter time point of 96 h (Supplementary Figure 3). This may indicate that sustained inhibition of ataxin-7 expression is required to achieve a reduction in mutant polyQ protein, sufficient to alleviate the cell stress response as measured by DNAJA1 HSP induction.
We further measured transcript levels of UCHL1 (Figure 3c). At low levels of ataxin-7 knockdown (21–48%), UCHL1 levels were elevated towards that observed in control samples with 30–59% increased expression of UCHL1. However, when the non-allele-specific siRNA, siR-atxn7, reduced total ataxin-7 expression by 68%, UCHL1 expression reverted to that observed in the absence of ataxin-7 silencing. In contrast, the allele-specific siR-P16, which demonstrated noticeably greater total ataxin-7 silencing (77%), sustained a 25% increase in UCHL1 expression.
To validate that normalisation of these transcripts was associated with removal of mutant ataxin-7, we performed parallel silencing experiments in control fibroblasts. Dose-dependent silencing by both siRNAs was highly similar to that observed in patient fibroblasts ranging from 30 to 64% knockdown (Figure 4a). However, no significant correlative decrease in DNAJA1 was observed (Figure 4b). Furthermore, at low doses of siRNA, no elevation of UCHL1 was observed (Figure 4c). We did, however, note a 30% decrease in UCHL1 expression (P<0.05) at high doses of siR-atxn7 in control cells, consistent with the reduced expression in patient cells at the same levels of silencing.
Effects of ataxin-7 silencing in control fibroblasts. (a) Two siRNAs (siR-atxn7 and siR-P16) silence ataxin-7 transcript levels in a dose-dependent manner relative to a non-specific siRNA (siR-NS). (b) Corresponding levels of DNAJA1 expression after treatment with increasing doses of siR-P16 and siR-atxn7. (c) Corresponding levels of UCHL1 expression after treatment with increasing doses of siR-P16 and siR-atxn7. Concentrations of siRNAs are shown on the x axis. *P value<0.05. Measurements of expression levels are taken from the same sample cohort as those demonstrating allele- or non-allele-specific silencing in Figure 1. Results are compiled from a one primary cell line, experiment performed in duplicate.
Discussion
RNAi-based therapies have shown great potential for treating several neurodegenerative diseases where the disease-causing gene has been identified and shown to have a toxic gain-of-function, as is the case with the polyQ disorders.6, 11, 21 However, only a few studies have assessed silencing in patient-derived cells, which exhibit physiologically relevant levels of endogenous expression.3, 22, 23, 24, 25 We sought to address whether polyQ patient-derived fibroblasts might be a useful model for assessing therapeutic strategies, such as comparing allele-specific and non-allele-specific silencing approaches in genetically accurate cells. All the polyQ patient-derived cell line studies to date3, 22, 23, 24, 25, 26 have focused on the development of allele-specific silencing approaches targeting SNPs linked to the mutation and/or the mutation itself, and show increasingly promising results. We used an siRNA designed to target a common SNP in the ataxin-7 transcript in the South African SCA7 population, in an allele-specific manner. A criticism of targeting SNPs is the low frequency of such a target, requiring multiple SNPs to be identified to incorporate the entire patient population.27 However, this SNP is linked to the polyQ expansion in 100% of the South African SCA7 patients and is heterozygous in 43% of this population,10, 13 making it a promising target for therapies aimed at treating large numbers of patients. Here, we compared the efficacy of two siRNAs, one allele-specific, one non-allele-specific, by measuring the expression of endogenous mutant and wild-type transcripts in patient cells. We show that the allele-specific siRNA silences the mutant transcript more efficiently than the wild-type transcript in comparison to the consistent silencing of both alleles by siR-atxn7. Although allele selectivity is lost at the highest dose of siRNA, the likelihood of saturation of the endogenous RNAi pathway at such doses28 suggests that our allele-specific silencing approach may still have significant clinical relevance.
Having established the allele specificity of our two siRNAs, we then investigated whether there might be transcriptional differences between patient and control fibroblast cell lines, which could be used to measure the efficacy of each silencing approach, as transcriptional dysregulation is considered a key feature of SCA7 pathogenesis.29, 30 Data from a SCA7 mouse model19 and patient-derived lymphoblasts20 have revealed a spectrum of transcriptional dysregulation. From these reports, we selected a subset of six transcripts in which robust changes had been published, and could be linked to a more general cell response that might present in fibroblasts, which are not considered to be a disease-susceptible cell type. Of these six, the HSP, DNAJA1, and the de-ubiquitinating enzyme, UCHL1, were found to be differentially expressed in patient cell lines compared with control lines, corresponding to changes observed in a SCA7 mouse model.19
DNAJA1 is elevated in the brains of the SCA7 mouse model in the early stages of disease, but is later decreased compared with control mouse brains. This phenomenon, in which an initially robust heat-shock response becomes progressively impaired in polyQ diseased animals of advanced age, is thought to arise from histone hypoacetylation, resulting in a decrease in heat-shock gene transcription over time.31 The initial increase in mouse brain and the observed increase in our patient-derived fibroblasts may represent a general response to cellular stress not specific to neurons. Cellular stress from accumulation of mutant polyQ protein is well established (reviewed in Huen et al32). Thus, it was not surprising to observe restoration of transcript levels to that of the control lines after suppression of mutant polyQ protein expression. We further observed that this restoration only occurred after sustained mutant ataxin-7 silencing. This may be indicative of the reported stability of mutant polyQ protein;33 such that improvements were noted only after sufficient silencing of the ataxin-7 transcript allowing for degradation of residual protein.
It should be noted that restoration of DNAJA1 expression was achieved only at non-allele-specific doses of siRNA. Nevertheless, the fact that silencing of ataxin-7 at equivalent doses in control fibroblasts had no effect on DNAJA1 expression suggests that normalisation of DNAJA1 expression in patient cells results from removal of mutant Ataxin-7 protein (ATXN7), rather than loss of normal ATXN7 function. That this effect is observed only at the concentrations at which siRNA is no longer allele specific is a small concern. However, the dosage effects in this in vitro assay may not equate to those in vivo. It is therefore possible that sustained allele-specific silencing in vivo would restore DNAJA1 over time, while maintaining allele specificity.
We observed decreased UCHL1 expression in patient cells relative to controls as previously reported.19 UCHL1 is a neuronal transcript, which recycles ubiquitin thereby maintaining protein degradation.34 Decreases in its expression, resulting in impairment of the ubiquitin proteasome pathway, have been linked to several neurodegenerative disorders, including Parkinson disease, Alzheimer’s disease and HD.35, 36, 37 Furthermore, a mouse model with a loss-of-function UCHL1 deletion displays an ataxic phenotype,38, 39 validating the changes seen in SCA7 patient cells.
Ataxin-7 silencing in patient cells resulted in an initial restoration of UCHL1 levels towards that observed in control lines. This is encouraging given that boosting UCHL1 levels has been suggested as a therapeutic strategy for Alzheimer’s disease.39 However, this initial increase was mitigated at higher levels of ataxin-7 knockdown after treatment with the non-allele-specific siRNA. In contrast, the allele-specific siRNA maintained elevated levels of UCHL1, implying that loss of UCHL1 expression at high doses of non-allele-specific silencing may be caused by loss of normal function of the wild-type ATXN7 protein. Although only significant after treatment with siR-atxn7, we observed a similar loss of UCHL1 expression in control fibroblasts. This suggests that in the context of SCA7 pathogenesis, loss of UCHL1 expression may be caused by both the toxic gain-of-function of the mutant polyQ protein as well as by loss of wild-type ATXN7 function. ATXN7 functions as a critical component of the STAGA transcriptional co-activator complex17 and polyQ-expanded ATXN7 has been shown to inhibit this function.40 Therefore, reduced UCHL1 expression as a result of decreased ATXN7 function fits well with our understanding of polyQ pathogenesis including aspects of both loss and gain of function.
We acknowledge that quantification of endogenous levels of ATXN7 protein would have served to enhance our understanding of the transcriptional dysregulation observed. Although we were able to successfully measure over-expression of co-transfected exogenous ATXN7 protein in HEK293 cells (data not shown), this was not reliably transferred to a robust measurement of equivalent endogenous protein. Nevertheless, we believe our novel allele-specific transcript quantification has yielded valuable information for South African patient-specific treatment.
We have shown that SCA7 patient-derived cell lines, which are generated and manipulated with relative ease, are highly suitable for studies of therapeutic efficacy (in this case, allele-specific vs non-allele specific siRNAs). To the best of our knowledge, this is the first time the superiority of an allele-specific silencing approach targeting a common SNP has been demonstrated in patient-derived cells. This suggests that although fibroblasts are not susceptible to the neuronal-specific degeneration exhibited in polyQ disorders, they may be a valuable tool for screening therapeutic molecules as well as providing insight into the mechanisms of polyQ disease.
References
La Spada AR, Taylor JP : Repeat expansion disease: progress and puzzles in disease pathogenesis. Nat Rev Genet 2010; 11: 247–258.
Alves S, Nascimento-Ferreira I, Auregan G et al: Allele-specific RNA silencing of mutant ataxin-3 mediates neuroprotection in a rat model of Machado-Joseph disease. PLoS One 2008; 3: e3341.
Fiszer A, Mykowska A, Krzyzosiak WJ : Inhibition of mutant huntingtin expression by RNA duplex targeting expanded CAG repeats. Nucleic Acids Res 2011; 39: 5578–5585.
Franich NR, Fitzsimons HL, Fong DM, Klugmann M, During MJ, Young D : AAV Vector–mediated RNAi of mutant Huntingtin expression is neuroprotective in a novel genetic rat model of Huntington's disease. Mol Ther 2008; 16: 947–956.
Gao Y, Zu T, Low WC, Orr HT, McIvor RS, Antisense RNA : sequences modulating the ataxin-1 message: molecular model of gene therapy for spinocerebellar ataxia type 1, a dominant-acting unstable trinucleotide repeat disease. Cell Transplant 2008; 17: 723–734.
Harper SQ, Staber PD, He X et al: RNA interference improves motor and neuropathological abnormalities in a Huntington's disease mouse model. Proc Natl Acad Sci USA 2005; 102: 5820–5825.
Kubodera T, Yokota T, Ishikawa K, Mizusawa H : New RNAi strategy for selective suppression of a mutant allele in polyglutamine disease. Oligonucleotides 2005; 15: 298–302.
Miller VM, Xia H, Marrs GL et al: Allele-specific silencing of dominant disease genes. Proc Natl Acad Sci USA 2003; 100: 7195–7200.
Rodriguez-Lebron E, Denovan-Wright EM, Nash K, Lewin AS, Mandel RJ : Intrastriatal rAAV-mediated delivery of anti-huntingtin shRNAs induces partial reversal of disease progression in R6/1 Huntington's disease transgenic mice. Mol Ther 2005; 12: 618–633.
Scholefield J, Greenberg LJ, Weinberg MS, Arbuthnot PB, Abdelgany A, Wood MJA : Design of RNAi hairpins for mutation-specific silencing of ataxin-7 and correction of a SCA7 phenotype. PLoS One 2009; 4: e7232.
Xia H, Mao Q, Eliason SL et al: RNAi suppresses polyglutamine-induced neurodegeneration in a model of spinocerebellar ataxia. Nat Med 2004; 10: 816–820.
Smith D, Bryer A, Watson LM, Greenberg LJ : Inherited polyglutamine spinocerebellar ataxias in South Africa. S Afr Med J 2012; 102: 683–686.
Greenberg J, Solomon G, Vorster A, Heckmann J, Bryer A : Origin of the SCA7 gene mutation in South Africa: implications for molecular diagnostics. Clin Genet 2006; 70: 415–417.
Dorschner MO, Barden D, Stephens K : Diagnosis of five spinocerebellar ataxia disorders by multiplex amplification and capillary electrophoresis. J Mol Diagn 2002; 4: 108–113.
Sly WS, Grubb J : Isolation of fibroblasts from patients. Meth Enzymol 1979; 58: 444–450.
Einum DD, Townsend JJ, Ptcek LJ, Fu Y : Ataxin-7 expression analysis in controls and spinocerebellar ataxia type 7 patients. Neurogenet 2001; 3: 83–90.
Helmlinger D, Hardy S, Sasorith S et al: Ataxin-7 is a subunit of GCN5 histone acetyltransferase-containing complexes. Hum Mol Genet 2004; 13: 1257.
Luthi-Carter R, Strand A, Peters NL et al: Decreased expression of striatal signaling genes in a mouse model of Huntington’s disease. Hum Mol Genet 2000; 9: 1259–1271.
Chou A, Chen C, Chen S et al: Polyglutamine-expanded ataxin-7 causes cerebellar dysfunction by inducing transcriptional dysregulation. Neurochem Int 2010; 56: 329–339.
Tsai HF, Lin SJ, Li C, Hsieh M : Decreased expression of Hsp27 and Hsp70 in transformed lymphoblastoid cells from patients with spinocerebellar ataxia type 7. Biochem Biophys Res Commun 2005; 334: 1279–1286.
Wang H, Ghosh A, Baigude H et al: Therapeutic gene silencing delivered by a chemically modified small interfering RNA against mutant SOD1 slows amyotrophic lateral sclerosis progression. J Biol Chem 2008; 283: 15845–15852.
Bilsen Pv, Jaspers L, Lombardi M, Odekerken J, Burright E, Kaemmerer W : Identification and allele-specific silencing of the mutant huntingtin allele in Huntington's disease patient-derived fibroblasts. Hum Gene Ther 2008; 19: 710–718.
Hu J, Matsui M, Gagnon KT et al: Allele-specific silencing of mutant huntingtin and ataxin-3 genes by targeting expanded CAG repeats in mRNAs. Nat Biotechnol 2009; 27: 478–484.
Hu J, Liu J, Corey DR : Allele-selective inhibition of huntingtin expression by switching to an miRNA-like RNAi mechanism. Chem Biol 2010; 17: 1183–1188.
Hu J, Gagnon KT, Liu J et al: Allele-selective inhibition of ataxin-3 (ATX3) expression by antisense oligomers and duplex RNAs. Biol Chem 2011; 392: 315–325.
Lombardi MS, Jaspers L, Spronkmans C et al: A majority of Huntington's disease patients may be treatable by individualized allele-specific RNA interference. Exp Neurol 2009; 217: 312–319.
Pfister EL, Kennington L, Straubhaar J et al: Five siRNAs targeting three SNPs may provide therapy for three-quarters of Huntington's disease patients. Curr Biol 2009; 19: 774–778.
Grimm D, Streetz KL, Jopling CL et al: Fatality in mice due to oversaturation of cellular microRNA/short hairpin RNA pathways. Nature 2006; 441: 537–541.
Ström A, Forsgren L, Holmberg M : A role for both wild-type and expanded ataxin-7 in transcriptional regulation. Neurobiol Dis 2005; 20: 646–655.
Helmlinger D, Tora L, Devys D : Transcriptional alterations and chromatin remodeling in polyglutamine diseases. Trends Genet 2006; 22: 562–570.
Labbadia J, Cunliffe H, Weiss A et al: Altered chromatin architecture underlies progressive impairment of the heat shock response in mouse models of Huntington disease. J Clin Invest 2011; 121: 3306–3319.
Huen NYM, Wong SLA, Chan HYE : Transcriptional malfunctioning of heat shock protein gene expression in spinocerebellar ataxias. Cerebellum 2007; 6: 111–117.
Yvert G, Lindenberg KS, Devys D, Helmlinger D, Landwehrmeyer GB, Mandel J : SCA7 mouse models show selective stabilization of mutant ataxin-7 and similar cellular responses in different neuronal cell types. Hum Mol Genet 2001; 10: 1679–1692.
Setsuie R, Wada K : The functions of UCH-L1 and its relation to neurodegenerative diseases. Neurochem Int 2007; 51: 105–111.
Metzger S, Bauer P, Tomiuk J et al: The S18Y polymorphism in the UCHL1 gene is a genetic modifier in Huntington’s disease. Neurogenet 2006; 7: 27–30.
Ardley HC, Scott GB, Rose SA, Tan NG, Robinson PA : UCH-L1 aggresome formation in response to proteasome impairment indicates a role in inclusion formation in Parkinson's disease. J Neurochem 2004; 90: 379–391.
Gong B, Cao Z, Zheng P et al: Ubiquitin hydrolase Uch-L1 rescues β-amyloid-induced decreases in synaptic function and contextual memory. Cell 2006; 126: 775–788.
Yamazaki K, Wakasugi N, Tomita T, Kikuchi T, Mukoyama M, Ando K : Gracile axonal dystrophy (GAD), a new neurological mutant in the mouse. Proc Soc Exp Biol Med 1988; 187: 209–215.
Saigoh K, Wang Y, Suh J et al: Intragenic deletion in the gene encoding ubiquitin carboxy-terminal hydrolase in gad mice. Nat Genet 1999; 23: 47–51.
Palhan VB, Chen S, Peng GH et al: Polyglutamine-expanded ataxin-7 inhibits STAGA histone acetyltransferase activity to produce retinal degeneration. Proc Natl Acad Sci USA 2005; 102: 8472–8477.
Acknowledgements
We are grateful to Juerg Hunziker for the design of siRNAs and to Novartis for providing the siRNAs, as well as Ingrid Baumgarten, who established patient fibroblast cultures. This project was funded by the National Ataxia Foundation (UK), the John Fell OUP Research Fund (University of Oxford), the National Research Foundation of South Africa and the University of Cape Town Research Council. At the time of writing, JS was a Nuffield Medical Fellow and LW received funding from the Commonwealth Scholarships Commission (UK), the National Research Foundation and the Harry Crossley Foundation. DS received funding from the German Academic Exchange Service (DAAD-NRF).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on European Journal of Human Genetics website
Rights and permissions
About this article
Cite this article
Scholefield, J., Watson, L., Smith, D. et al. Allele-specific silencing of mutant Ataxin-7 in SCA7 patient-derived fibroblasts. Eur J Hum Genet 22, 1369–1375 (2014). https://doi.org/10.1038/ejhg.2014.39
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejhg.2014.39
This article is cited by
-
Chemical engineering of therapeutic siRNAs for allele-specific gene silencing in Huntington’s disease models
Nature Communications (2022)
-
Exploring the Potential of Small Molecule-Based Therapeutic Approaches for Targeting Trinucleotide Repeat Disorders
Molecular Neurobiology (2020)
-
Ophthalmological and Neurologic Manifestations in Pre-clinical and Clinical Phases of Spinocerebellar Ataxia Type 7
The Cerebellum (2019)
-
Genetics, Mechanisms, and Therapeutic Progress in Polyglutamine Spinocerebellar Ataxias
Neurotherapeutics (2019)
-
Molecular Targets and Therapeutic Strategies in Spinocerebellar Ataxia Type 7
Neurotherapeutics (2019)