Abstract
Background/Objectives:
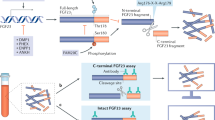
Iron and phosphate are both vital to many biological cellular processes with central roles in energy metabolism, cellular proliferation and nucleic acid synthesis. Regulatory pathways in some of these metabolic pathways may intersect at fibroblast growth factor 23 (FGF23), a major phosphate regulatory hormone. Iron is reported to induce hypophosphataemia in rare cases, and recent reports suggest that iron deficiency may upregulate FGF23 synthesis by mechanisms involving hypoxia-inducible factor 1α (HIF1α). Our objective was to evaluate the effect of administration of intravenous iron polymaltose on intact and c-terminal FGF23 (i:cFGF23) ratios in two independent cohorts of patients, iron-deficient but non-inflamed patients and haemodialysis (HD)-dependent patients, and to examine the balance of synthesis and degradation.
Subjects/Methods:
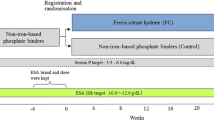
We studied biochemical effects of intravenous iron polymaltose on both iFGF23 and cFGF23 fragments and their ratios in two patient groups: iron-deficient patients with normal renal function (ID-norm) and HD patients receiving iron supplementation (HD-ESKD) at a single institution. Patients were tested at baseline, day 4 and day 12 post iron administration.
Results:
Parenteral iron polymaltose resulted in increased i:cFGF23 ratios in ID-norm patients where circulating cFGF23 levels decreased with no appreciable effect on iFGF23, whereas no significant changes in i:cFGF23 ratios were observed in HD-ESKD patients following intravenous administration of 100mg iron polymaltose.
Conclusions:
Dysregulation of intracellular FGF23-processing mechanisms may be related to iron deficiency per se rather than iron repletion with iron polymaltose. In ID-norm, i:cFGF23 ratios altered with iron administration without significant clinical alterations in mineral parameters, implying that other regulatory mechanisms may be important. Finally, iron supplementation in HD-ESKD patients does not appear to significantly affect i:cFGF23 ratios already disturbed by a chronic inflammatory or functionally iron-deficient state.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Camaschella C . Iron-deficiency anemia. N Engl J Med 2015; 372: 1832–1843.
Hentze MW, Muckenthaler MU, Galy B, Camaschella C . Two to tango: regulation of Mammalian iron metabolism. Cell 2010; 142: 24–38.
Nissenson AR, Strobos J . Iron deficiency in patients with renal failure. Kidney Int Suppl 1999; 69: S18–S21.
Kidney Disease: Improving Global Outcomes Anemia Work Group. KDIGO Clinical Practice Guideline for Anemia in Chronic Kidney Disease. Kidney Int 2012 (Suppl 2), 279–335.
Schouten BJ, Doogue MP, Soule SG, Hunt PJ . Iron polymaltose-induced FGF23 elevation complicated by hypophosphataemic osteomalacia. Ann Clin Biochem 2009; 46 (Pt 2), 167–169.
Feng S, Wang J, Zhang Y, Creighton CJ, Ittmann M . FGF23 promotes prostate cancer progression. Oncotarget 2015; 6: 17291–17301.
Suvannasankha A, Tompkins DR, Edwards DF, Petyaykina KV, Crean CD, Fournier PG et al. FGF23 is elevated in multiple myeloma and increases heparanase expression by tumor cells. Oncotarget 2015; 6: 19647–19660.
Smith ER, McMahon LP, Holt SG . Fibroblast growth factor 23. Ann Clin Biochem 2014; 51 (Pt 2), 203–227.
David V, Martin A, Isakova T, Spaulding C, Qi L, Ramirez V et al. Inflammation and functional iron deficiency regulate fibroblast growth factor 23 production. Kidney Int 2015; 89: 135–146.
Walton RJ, Bijvoet OL . Nomogram for derivation of renal threshold phosphate concentration. Lancet 1975; 2: 309–310.
Kenny AP, Glen AC . Tests of phosphate reabsorption. Lancet 1973; 2: 158.
Barth JH, Jones RG, Payne RB . Calculation of renal tubular reabsorption of phosphate: the algorithm performs better than the nomogram. Ann Clin Biochem 2000; 37 (Pt 1), 79–81.
Wolf M, Koch TA, Bregman DB . Effects of iron deficiency anemia and its treatment on fibroblast growth factor 23 and phosphate homeostasis in women. J Bone Miner Res 2013; 28: 1793–1803.
Schouten BJ, Hunt PJ, Livesey JH, Frampton CM, Soule SG . FGF23 elevation and hypophosphatemia after intravenous iron polymaltose: a prospective study. J Clin Endocrinol Metab 2009; 94: 2332–2337.
Goetz R, Nakada Y, Hu MC, Kurosu H, Wang L, Nakatani T et al. Isolated C-terminal tail of FGF23 alleviates hypophosphatemia by inhibiting FGF23-FGFR-Klotho complex formation. Proc Natl Acad Sci USA 2010; 107: 407–412.
Stenvinkel P . Inflammation in end-stage renal disease: the hidden enemy. Nephrology 2006; 11: 36–41.
Hryszko T, Rydzewska-Rosolowska A, Brzosko S, Koc-Zorawska E, Mysliwiec M . Low molecular weight iron dextran increases fibroblast growth factor-23 concentration, together with parathyroid hormone decrease in hemodialyzed patients. Ther Apher Dial 2012; 16: 146–151.
Takeda Y, Komaba H, Goto S, Fujii H, Umezu M, Hasegawa H et al. Effect of intravenous saccharated ferric oxide on serum FGF23 and mineral metabolism in hemodialysis patients. Am J Nephrol 2011; 33: 421–426.
Farrow EG, Yu X, Summers LJ, Davis SI, Fleet JC, Allen MR et al. Iron deficiency drives an autosomal dominant hypophosphatemic rickets (ADHR) phenotype in fibroblast growth factor-23 (Fgf23) knock-in mice. Proc Natl Acad Sci USA 2011; 108: E1146–E1155.
Peyssonnaux C, Zinkernagel AS, Schuepbach RA, Rankin E, Vaulont S, Haase VH et al. Regulation of iron homeostasis by the hypoxia-inducible transcription factors (HIFs). J Clin Invest 2007; 117: 1926–1932.
Knowles HJ, Raval RR, Harris AL, Ratcliffe PJ . Effect of ascorbate on the activity of hypoxia-inducible factor in cancer cells. Cancer Res 2003; 63: 1764–1768.
Gravesen E, Hofman-Bang J, Mace ML, Lewin E, Olgaard K . High dose intravenous iron, mineral homeostasis and intact FGF23 in normal and uremic rats. BMC Nephrol 2013; 14: 281.
Danielson B . Structure, Chemistry, and Pharmacokinetics of Intravenous Iron Agents. J Am Soc Nephrol 2004; 15 (Suppl 2), S93–S98.
Dillon R, Momoh I, Francis Y, Cameron L, Harrison CN, Radia D . Comparative efficacy of three forms of parenteral iron. J Blood Transfus 2012; 2012: 473514.
Smith ER, Cai MM, McMahon LP, Holt SG . Biological variability of plasma intact and C-terminal FGF23 measurements. J Clin Endocrinol Metab 2012; 97: 3357–3365.
Acknowledgements
SJT has received speaking honoraria from Shire. ERS has received research funding from Amgen and Baxter, honoraria from Shire and served as a consultant for Vifor Pharma. NDT has received consultancy fees, honoraria and research funding from Amgen and Shire Pharmaceuticals. SGH has received research funding or honoraria from Amgen, Baxter, Gilead, Novartis and Shire.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Tan, SJ., Satake, S., Smith, E. et al. Parenteral iron polymaltose changes i:c-terminal FGF23 ratios in iron deficiency, but not in dialysis patients. Eur J Clin Nutr 71, 180–184 (2017). https://doi.org/10.1038/ejcn.2016.217
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2016.217
This article is cited by
-
The Effect of Iron Supplementation on FGF23 in Chronic Kidney Disease Patients: a Systematic Review and Time-Response Meta-Analysis
Biological Trace Element Research (2021)