Abstract
Background/Objectives:
New methods to measure visceral adipose tissue (VAT) by dual-energy X-ray absorptiometry (DXA) may help discern sex, race and phenotype differences in the role of VAT in cardiometabolic risk. This study was designed (1) to compare relationships of DXA-VAT, anthropometric and body composition variables with cardiometabolic risk factors in obese women; (2) to determine which variables most robustly predict impaired glucose tolerance (IGT) and metabolic syndrome (MetSx); and (3) to determine thresholds for DXA-VAT by race.
Subjects/Methods:
VAT mass (g) and volume (cm3) were measured in 229 obese (body mass index (BMI), 30–49.9) women aged 21–69 years of European-American (EA=123) and African-American (AA=106) descent using the CoreScan algorithm on a Lunar iDXA scanner. Linear regression modeling and areas under the curve (AUC of ROC (receiver operating characteristic) curves) compared relationships with cardiometabolic risk. Bootstrapping with LASSO (least absolute shrinkage and selection operator) regression modeling determined thresholds and predictors of IGT and MetSx.
Results:
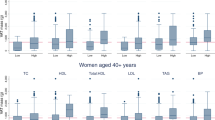
DXA-VAT explained more of the variance in triglycerides, blood pressure, glucose and homeostatic model assessment-insulin resistance (HOMA-IR) compared with anthropometric and other body composition variables. DXA-VAT also had the highest AUC for IGT (0.767) and MetSx (0.749). Including race as a variable and the interaction between VAT and race in modeling did not significantly change the results. Thresholds at which the probability of developing IGT or MetSx was⩾50% were determined separately for AA women (IGT: 2120 cm3; MetSx: 1320 cm3) and EA women (IGT: 2550 cm3; MetSx: 1713 cm3). The odds for IGT or MetSx were fourfold greater with each standard deviation increase in DXA-VAT.
Conclusions:
DXA-VAT provides robust clinical information regarding cardiometabolic risk in AA and EA obese women and offers potential utility in the risk reduction interventions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Lebovitz HE . The relationship of obesity to the metabolic syndrome. Int J Clin Pract Suppl 134: 18–27.
Canoy D . Distribution of body fat and risk of coronary heart disease in men and women. Curr Opin Cardiol 23: 591–598.
Lee CM, Huxley RR, Wildman RP, Woodward M . Indices of abdominal obesity are better discriminators of cardiovascular risk factors than BMI: a meta-analysis. J Clin Epidemiol 2008; 61: 646–653.
Girard J, Lafontan M . Impact of visceral adipose tissue on liver metabolism and insulin resistance. Part II: Visceral adipose tissue production and liver metabolism. Diabetes Metab 34: 439–445.
Bjorntorp P . "Portal" adipose tissue as a generator of risk factors for cardiovascular disease and diabetes. Arteriosclerosis 1990; 10: 493–496.
Liu J, Coady S, Carr JJ, Hoffmann U, Taylor HA, Fox CS . Differential associations of abdominal visceral, subcutaneous adipose tissue with cardiometabolic risk factors between African and European Americans. Obesity (Silver Spring) 2014; 22: 811–818.
Fox CS, Massaro JM, Hoffmann U, Pou KM, Maurovich-Horvat P, Liu CY et al. Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the Framingham Heart Study. Circulation 2007; 116: 39–48.
Neeland IJ, Ayers CR, Rohatgi AK, Turer AT, Berry JD, Das SR et al. Associations of visceral and abdominal subcutaneous adipose tissue with markers of cardiac and metabolic risk in obese adults. Obesity (Silver Spring) 2013; 21: E439–E447.
Kuk JL, Katzmarzyk PT, Nichaman MZ, Church TS, Blair SN, Ross R . Visceral fat is an independent predictor of all-cause mortality in men. Obesity (Silver Spring) 2006; 14: 336–341.
Patel P, Abate N . Body fat distribution and insulin resistance. Nutrients 2013; 5: 2019–2027.
Smith SR, Lovejoy JC, Greenway F, Ryan D, deJonge L, de la Bretonne J et al. Contributions of total body fat, abdominal subcutaneous adipose tissue compartments, and visceral adipose tissue to the metabolic complications of obesity. Metabolism 2001; 50: 425–435.
Ogden CL, Carroll MD, Kit BK, Flegal KM . Prevalence of childhood and adult obesity in the United States, 2011-2012. JAMA 2014; 311: 806–814.
Demerath EW, Sun SS, Rogers N, Lee M, Reed D, Choh AC et al. Anatomical patterning of visceral adipose tissue: race, sex, and age variation. Obesity (Silver Spring) 2007; 15: 2984–2993.
Ross R, Shaw KD, Rissanen J, Martel Y, de Guise J, Avruch L . Sex differences in lean and adipose tissue distribution by magnetic resonance imaging: anthropometric relationships. Am J Clin Nutr 1994; 59: 1277–1285.
Okosun IS, Chandra KM, Boev A, Boltri JM, Choi ST, Parish DC et al. Abdominal adiposity in U.S. adults: prevalence and trends, 1960-2000. Prev Med 39: 197–206.
Pascot A, Lemieux I, Prud'homme D, Tremblay A, Nadeau A, Couillard C et al. Reduced HDL particle size as an additional feature of the atherogenic dyslipidemia of abdominal obesity. J Lipid Res 2001; 42: 2007–2014.
Anjana M, Sandeep S, Deepa R, Vimaleswaran KS, Farooq S, Mohan V . Visceral and central abdominal fat and anthropometry in relation to diabetes in Asian Indians. Diabetes Care 2004; 27: 2948–2953.
Lear SA, Chockalingam A, Kohli S, Richardson CG, Humphries KH . Elevation in cardiovascular disease risk in South Asians is mediated by differences in visceral adipose tissue. Obesity (Silver Spring) 2012; 20: 1293–1300.
Katzmarzyk PT, Bray GA, Greenway FL, Johnson WD, Newton RL Jr., Ravussin E et al. Racial differences in abdominal depot-specific adiposity in white and African American adults. Am J Clin Nutr 2010; 91: 7–15.
Liu J, Coady S, Carr JJ, Md UH, Taylor HA, Fox CS . Differential associations of abdominal visceral, subcutaneous adipose tissue with cardiometabolic risk factors between African and European Americans. Obesity (Silver Spring) 2013; 22: 14.
Tchernof A, Despres JP . Pathophysiology of human visceral obesity: an update. Physiol Rev 93: 359–404.
Black HR . The burden of cardiovascular disease: following the link from hypertension to myocardial infarction and heart failure. Am J Hypertens 16: 4S–6S.
Carroll JF, Chiapa AL, Rodriquez M, Phelps DR, Cardarelli KM, Vishwanatha JK et al. Visceral fat, waist circumference, and BMI: impact of race/ethnicity. Obesity (Silver Spring) 2008; 16: 600–607.
Barreira TV, Staiano AE, Harrington DM, Heymsfield SB, Smith SR, Bouchard C et al. Anthropometric correlates of total body fat, abdominal adiposity, and cardiovascular disease risk factors in a biracial sample of men and women. Mayo Clin Proc 2012; 87: 452–460.
Janssen I, Katzmarzyk PT, Ross R . Waist circumference and not body mass index explains obesity-related health risk. Am J Clin Nutr 2004; 79: 379–384.
Karelis AD . Metabolically healthy but obese individuals. Lancet 2008; 372: 1281–1283.
Primeau V, Coderre L, Karelis AD, Brochu M, Lavoie ME, Messier V et al. Characterizing the profile of obese patients who are metabolically healthy. Int J Obes (Lond) 2011; 35: 971–981.
Wildman RP, Muntner P, Reynolds K, McGinn AP, Rajpathak S, Wylie-Rosett J et al. The obese without cardiometabolic risk factor clustering and the normal weight with cardiometabolic risk factor clustering: prevalence and correlates of 2 phenotypes among the US population (NHANES 1999-2004). Arch Intern Med 2008; 168: 1617–1624.
Wang J, Thornton JC, Bari S, Williamson B, Gallagher D, Heymsfield SB et al. Comparisons of waist circumferences measured at 4 sites. Am J Clin Nutr 2003; 77: 379–384.
Maurovich-Horvat P, Massaro J, Fox CS, Moselewski F, O'Donnell CJ, Hoffmann U . Comparison of anthropometric, area- and volume-based assessment of abdominal subcutaneous and visceral adipose tissue volumes using multi-detector computed tomography. Int J Obes 31: 500–506.
Liou TH, Chan WP, Pan LC, Lin PW, Chou P, Chen CH . Fully automated large-scale assessment of visceral and subcutaneous abdominal adipose tissue by magnetic resonance imaging. Int J Obes (Lond) 2006; 30: 844–852.
Silver HJ, Welch EB, Avison MJ, Niswender KD . Imaging body composition in obesity and weight loss: challenges and opportunities. Diabetes Metab Synd Obes 3: 337–347.
Micklesfield LK, Goedecke JH, Punyanitya M, Wilson KE, Kelly TL . Dual-energy X-ray performs as well as clinical computed tomography for the measurement of visceral fat. Obesity (Silver Spring) 2012; 20: 1109–1114.
Treuth MS, Hunter GR, Kekes-Szabo T . Estimating intraabdominal adipose tissue in women by dual-energy X-ray absorptiometry. Am J Clin Nutr 1995; 62: 527–532.
Park YW, Heymsfield SB, Gallagher D . Are dual-energy X-ray absorptiometry regional estimates associated with visceral adipose tissue mass? Int J Obes Relat Metab Disord 2002; 26: 978–983.
Hill AM, LaForgia J, Coates AM, Buckley JD, Howe PR . Estimating abdominal adipose tissue with DXA and anthropometry. Obesity (Silver Spring) 2007; 15: 504–510.
Scherzer R, Shen W, Bacchetti P, Kotler D, Lewis CE, Shlipak MG et al. Comparison of dual-energy X-ray absorptiometry and magnetic resonance imaging-measured adipose tissue depots in HIV-infected and control subjects. Am J Clin Nutr 2008; 88: 1088–1096.
Kamel EG, McNeill G, Han TS, Smith FW, Avenell A, Davidson L et al. Measurement of abdominal fat by magnetic resonance imaging, dual-energy X-ray absorptiometry and anthropometry in non-obese men and women. Int J Obes Relat Metab Disord 1999; 23: 686–692.
Silver HJ, Niswender KD, Kullberg J, Berglund J, Johansson L, Bruvold M et al. Comparison of gross body fat-water magnetic resonance imaging at 3 Tesla to dual-energy X-ray absorptiometry in obese women. Obesity (Silver Spring) 2013; 21: 765–774.
Kaul S, Rothney MP, Peters DM, Wacker WK, Davis CE, Shapiro MD et al. Dual-energy X-ray absorptiometry for quantification of visceral fat. Obesity (Silver Spring) 2012; 20: 1313–1318.
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG . Research electronic data capture (REDCap)–a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 42: 377–381.
Martin AD, Daniel MZ, Drinkwater DT, Clarys JP . Adipose tissue density, estimated adipose lipid fraction and whole body adiposity in male cadavers. Int J Obes Relat Metab Disord 1994; 18: 79–83.
Wallace TM, Levy JC, Matthews DR . Use and abuse of HOMA modeling. Diabetes Care 2004; 27: 1487–1495.
Pacini G, Bergman RN . MINMOD: a computer program to calculate insulin sensitivity and pancreatic responsivity from the frequently sampled intravenous glucose tolerance test. Comput Methods Programs Biomed 23: 113–122.
Avignon A, Boegner C, Mariano-Goulart D, Colette C, Monnier L . Assessment of insulin sensitivity from plasma insulin and glucose in the fasting or post oral glucose-load state. Int J Obes Relat Metab Disord 1999; 23: 512–517.
American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2013; 36: S67–S74.
Kahn SE, Prigeon RL, McCulloch DK, Boyko EJ, Bergman RN, Schwartz MW et al. The contribution of insulin-dependent and insulin-independent glucose uptake to intravenous glucose tolerance in healthy human subjects. Diabetes 1994; 43: 587–592.
American Heart Association; National Heart, Lung, and Blood Institute, American Heart Association; National Heart, Lung, and Blood Institute, Grundy SM, American Heart Association; National Heart, Lung, and Blood Institute, Cleeman JI, American Heart Association; National Heart, Lung, and Blood Institute, Daniels SR et al. Diagnosis and management of the metabolic syndrome. An American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Executive summary. Cardiol Rev 13: 322–327.
Harford KA, Reynolds CM, McGillicuddy FC, Roche HM . Fats inflammation and insulin resistance: insights to the role of macrophage and T-cell accumulation in adipose tissue. Proc Nutr Soc 70: 408–417.
Bastard JP, Maachi M, Lagathu C, Kim MJ, Caron M, Vidal H et al. Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur Cytokine Netw 2006; 17: 4–12.
Galic S, Oakhill JS, Steinberg GR . Adipose tissue as an endocrine organ. Mol Cell Endocrinol 2010; 316: 129–139.
Alvehus M, Buren J, Sjostrom M, Goedecke J, Olsson T . The human visceral fat depot has a unique inflammatory profile. Obesity (Silver Spring) 2010; 18: 879–883.
Fontana L, Eagon JC, Trujillo ME, Scherer PE, Klein S . Visceral fat adipokine secretion is associated with systemic inflammation in obese humans. Diabetes 2007; 56: 1010–1013.
Hyatt TC, Phadke RP, Hunter GR, Bush NC, Munoz AJ, Gower BA . Insulin sensitivity in African-American and white women: association with inflammation. Obesity (Silver Spring) 2009; 17: 276–282.
Albert MA, Glynn RJ, Buring J, Ridker PM . C-reactive protein levels among women of various ethnic groups living in the United States (from the Women's Health Study). Am J Cardiol 2004; 93: 1238–1242.
Coll T, Eyre E, Rodriguez-Calvo R, Palomer X, Sanchez RM, Merlos M et al. Oleate reverses palmitate-induced insulin resistance and inflammation in skeletal muscle cells. J Biol Chem 2008; 283: 11107–11116.
Coll T, Palomer X, Blanco-Vaca F, Escola-Gil JC, Sanchez RM, Laguna JC et al. Cyclooxygenase 2 inhibition exacerbates palmitate-induced inflammation and insulin resistance in skeletal muscle cells. Endocrinology 2010; 151: 537–548.
Qi L, Hu FB . Dietary glycemic load, whole grains, and systemic inflammation in diabetes: the epidemiological evidence. Curr Opin Lipidol 2007; 18: 3–8.
Liu S, Manson JE, Buring JE, Stampfer MJ, Willett WC, Ridker PM . Relation between a diet with a high glycemic load and plasma concentrations of high-sensitivity C-reactive protein in middle-aged women. Am J Clin Nutr 2002; 75: 492–498.
Shibao C, Gamboa A, Diedrich A, Ertl AC, Chen KY, Byrne DW et al. Autonomic contribution to blood pressure and metabolism in obesity. Hypertension 2007; 49: 27–33.
Abate NI, Mansour YH, Tuncel M, Arbique D, Chavoshan B, Kizilbash A et al. Overweight and sympathetic overactivity in black Americans. Hypertension 2001; 38: 379–383.
Tybor DJ, Lichtenstein AH, Dallal GE, Daniels SR, Must A . Independent effects of age-related changes in waist circumference and BMI z scores in predicting cardiovascular disease risk factors in a prospective cohort of adolescent females. Am J Clin Nutr 2011; 93: 392–401.
Brummett BH, Babyak MB, Siegler IC, Surwit R, Georgiades A, Boyle SH et al. Systolic blood pressure and adiposity: examination by race and gender in a nationally representative sample of young adults. Am J Hypertens 2012; 25: 140–144.
Hajjar I, Kotchen JM, Kotchen TA . Hypertension: trends in prevalence, incidence, and control. Annu Rev Public Health 2006; 27: 465–490.
Cowie CC, Rust KF, Byrd-Holt DD, Eberhardt MS, Flegal KM, Engelgau MM et al. Prevalence of diabetes and impaired fasting glucose in adults in the U.S. population: National Health And Nutrition Examination Survey 1999-2002. Diabetes Care 2006; 29: 1263–1268.
Centers for Disease Control and Prevention. Prevalence of diabetes and impaired fasting glucose in adults–United States, 1999-2000. MMWR Morb Mortal Wkly Rep 2003; 52: 833–837.
Katzmarzyk PT, Heymsfield SB, Bouchard C . Clinical utility of visceral adipose tissue for the identification of cardiometabolic risk in white and African American adults. Am J Clin Nutr 2013; 97: 480–486.
Nicklas BJ, Penninx BW, Ryan AS, Berman DM, Lynch NA, Dennis KE . Visceral adipose tissue cutoffs associated with metabolic risk factors for coronary heart disease in women. Diabetes Care 2003; 26: 1413–1420.
Katzmarzyk PT, Greenway FL, Heymsfield SB, Bouchard C . Clinical utility and reproducibility of visceral adipose tissue measurements derived from dual-energy X-ray absorptiometry in white and African American adults. Obesity (Silver Spring) 2013; 21: 2221–2224.
Acknowledgements
This study was supported by a grant from the Dr Robert C and Veronica Atkins Foundation to Dr Silver, NIH K23 HL103976 PhRMA Foundation Career Development Award to Dr Shibao and resources from Vanderbilt CTSA award UL1TR000445 from the NIH National Center for Advancing Translational Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Bi, X., Seabolt, L., Shibao, C. et al. DXA-measured visceral adipose tissue predicts impaired glucose tolerance and metabolic syndrome in obese Caucasian and African-American women. Eur J Clin Nutr 69, 329–336 (2015). https://doi.org/10.1038/ejcn.2014.227
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2014.227
This article is cited by
-
A new approach to quantify visceral fat via bioelectrical impedance analysis and ultrasound compared to MRI
International Journal of Obesity (2024)
-
Visceral Adipose Tissue and Non-alcoholic Fatty Liver Disease in Patients with Type 2 Diabetes
Digestive Diseases and Sciences (2022)
-
Official Position of the Brazilian Association of Bone Assessment and Metabolism (ABRASSO) on the evaluation of body composition by densitometry—part II (clinical aspects): interpretation, reporting, and special situations
Advances in Rheumatology (2022)
-
Age- and sex-specific visceral fat reference cutoffs and their association with cardio-metabolic risk
International Journal of Obesity (2021)
-
Age- and sex-specific reference intervals for visceral fat mass in adults
International Journal of Obesity (2020)