- NEWS FEATURE
How nasal-spray vaccines could change the pandemic
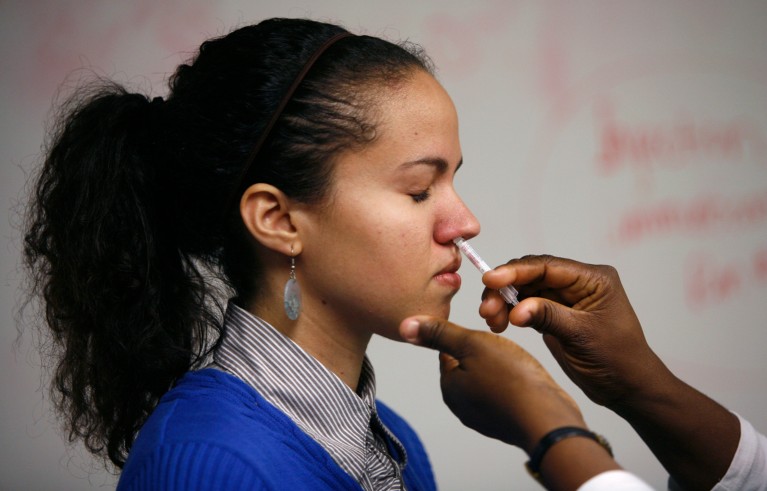
A student in Washington DC receives an influenza nasal-spray vaccine, in 2009. Intranasal and oral COVID-19 vaccines are now in development. Credit: Hyungwon Kang/Reuters
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 609, 240-242 (2022)
doi: https://doi.org/10.1038/d41586-022-02824-3
References
Belshe, R. B. et al. N. Engl. J. Med. 356, 685–696 (2007).
Mao, T. et al. Preprint at bioRxiv https://doi.org/10.1101/2022.01.24.477597 (2022).
Le Nouën, C. et al. Preprint at bioRxiv https://doi.org/10.1101/2022.05.21.492923 (2022).
Li, J.-X. et al. Preprint at SSRN https://doi.org/10.2139/ssrn.4000565 (2022).
Jin, L. et al. Preprint at medRxiv https://doi.org/10.1101/2022.07.26.22278072 (2022).

 China and India approve nasal COVID vaccines — are they a game changer?
China and India approve nasal COVID vaccines — are they a game changer?
 New Omicron-specific vaccines offer similar protection to existing boosters
New Omicron-specific vaccines offer similar protection to existing boosters
 Which COVID boosters to take and when: a guide for the perplexed
Which COVID boosters to take and when: a guide for the perplexed
 Fast-evolving COVID variants complicate vaccine updates
Fast-evolving COVID variants complicate vaccine updates